Research Article
Ariel Poulson
Ariel Poulson
D.
Gary Young Research Institute, Lehi, UT 84043, USA.
Tyler M. Wilson
Tyler M. Wilson
Corresponding Author
D.
Gary Young Research Institute, Lehi, UT 84043, USA.
E
mail: tywilson@youngliving.com, Tel.:
+1-801-899-4350
Reilly E. Sonstrom
Reilly E. Sonstrom
BrightSpec, Inc.,
Charlottesville, VA 22903, USA
Audra Davis
Audra Davis
D.
Gary Young Research Institute, Lehi, UT 84043, USA.
Justin L. Neill
Justin L. Neill
BrightSpec, Inc.,
Charlottesville, VA 22903, USA
Richard E. Carlson
Richard E. Carlson
D. Gary Young Research Institute, Lehi, UT 84043, USA.
E mail: RichCarlson@YoungLiving.com
Received: 2023-05-31 | Revised:2023-06-30 | Accepted: 2023-07-04 | Published: 2023-07-13
Pages: 246-254
DOI: https://doi.org/10.58985/jeopc.2023.v01i03.31
Abstract
Juniperus
communis var depressa (common juniper) is an essential oil-bearing
evergreen shrub native to North America, including the state of Utah. Cones
(berries), leaves, and limb material were collected, and steam distilled
together. The resulting essential oil samples (n = 3) were analyzed, and
both the achiral and chiral profiles established by GC/FID, GC/MS,
enantioselective GC/FID, and molecular rotational resonance (MRR). Prominent
compounds of the achiral profile include (averages) α-pinene (63.9%), β-pinene
(6.2%), myrcene (6.9%), δ-3-carene (6.8%), and limonene (3.3%). Four prominent
chiral pairs (α-pinene, β-pinene, δ-3-carene, limonene) were analyzed to
determine the enantioselective profile. Where enantiopure standards were not
commercially available, (-)-δ-3-carene, MRR was used as a quick and reliable
analytical technique for chiral analysis. This study verifies the achiral
profile for North American common juniper and, for the first time to the
authors’ knowledge, establishes the achiral and chiral profiles for J.
communis var. depressa, specifically from Utah. This study
confirms the utility and practicality of using MRR for determining chiral
profiles in essential oils. Additionally, results provide a foundation for
future research in the flavor and fragrance industries for common juniper of
North American origin.
Keywords
Cupressaceae, enantioselective
profile, essential oil composition, gas chromatography, Juniperus communis var.
depressa, molecular rotational resonance (MRR)
1. Introduction
Common juniper (Juniperus communis L.) is
a small coniferous evergreen shrub or tree in the Cupressaceae family, and the
most widespread juniper species in the world [1, 2]. This
circumboreal species is native to both the Eastern and Western Hemispheres and
is subject to much geographic variation [1,3]. Nine
different varieties of J. communis have been identified worldwide [3].
Species
from the eastern and western hemispheres can easily be distinguished through
genetic investigation [4, 5]. In the western
hemisphere, distinguishing the native varieties of J. communis is more
difficult, but currently there are five accepted varieties of the species based
on DNA and morphology: var. charlottensis, var. depressa, var. jackii,
var. megostocarpa, and var. saxatilis [4,
6]. J.
communis var. depressa is common throughout North
America [2,
7, 8].
The only documented variety of common juniper in Utah is Juniperus communis var. depressa Pursh [1,9-12]. Typically, it is found in alpine regions among aspen and spruce-fir communities, is less than 1 m in height, has awl shaped leaves in whorls of three that are dark green with a white band on the upper portion, and is generally dioecious, although it can be monecious [1,10-12]. The berry-like cones ripen and mature from green to dark blue-black over 2 years [1,10-13].
One of the most common uses of juniper
cones is in making beverages, liqueurs (Borovička and Steinhäger), and in
flavoring gin [1, 2, 11]. During the second
World War, North America was cut off from European sources of commercial
juniper cones (typically var. erecta), and several attempts were made to
replace the demand with domestic sources (var. depressa). While domestic
supplies were mostly considered inferior to European supplies, due to a
turpentine-like off-note, some sources contained a similar aroma and flavor to
var. erecta [2].
Since each volatile compound, and
enantiomer, displays a unique aroma and flavor, the achiral and chiral
essential oil composition are integral to understanding the use of common
juniper essential oil in the flavor and fragrance industry. Many different
varieties exist with established achiral essential oil [14-21]
and chiral (enantioselective) profiles [16-23].
To the author’s best knowledge, the essential oil composition and
enantioselective profile of J. communis var. depressa from Utah
have never been characterized. In this study limb, leaf, and cone material of J.
communis var. depressa were steam distilled together, and the
resulting essential oil analyzed by GC/FID, GC/MS, enantioselective GC, and
molecular rotational resonance (MRR). The achiral, essential oil composition was
determined by gas chromatography and enantioselective profile established
utilizing gas chromatography and MRR, which characterizes volatile compounds in
the gas phase through their characteristic pure rotational momentum transitions
[24]. As MRR is an extremely high-resolution
spectroscopic technique and is highly sensitive to slight changes in a
molecule’s three-dimensional mass distribution, distinct compounds (including
isomers), using the chiral tagging technique, can be identified and quantified
in a mixture without the need for enantiopure reference standards or
chromatographic separation [24-27]. Results
provide a foundation for future research in the flavor and fragrance industries
for common juniper of North American origin.
2. Materials and methods
Juniperus
communis var depressa plant material (cones, leaves, limb) was
collected during the third week of August 2020 from private land in Tabiona,
UT, USA. Plant material was collected from the following location: 40°20’43” N
110°45’10”W (elevation 2350 m) and stored at -20 ± 2 °C until distillation. A representative voucher sample is held in the Young Living Aromatic
Herbarium (YLAH): Juniperus communis var. depressa Pursh, Wilson 2021-01 (YLAH).
Prior to
distillation, the frozen limbs material was cut into 5-10 cm segments and all
plant parts (cones, leaves, limbs) were distilled together/simultaneously.
Laboratory-scale distillation was as follows: 3 L of water was added to the
bottom of a 12 L distillation chamber (Albrigi Luigi S.R.L., Italy), plant
material accurately weighed and added to the distillation chamber, distillation
for 2 hours from pass-over by direct steam, essential oil separated by a cooled
condenser and Florentine flask. Essential oil samples (n = 3) were
filtered and stored in a sealed amber glass bottle until analysis. For
simplicity and consistency, each sample will be referred to by a letter, A-C.
Essential oils were analyzed, and volatile compounds identified, by
GC/MS using an Agilent 7890B GC/5977B MSD (Agilent Technologies, Santa Clara,
CA, USA) and Agilent J&W DB-5, 60 m x 0.25 mm, 0.25 μm film thickness,
fused silica capillary column. Operating conditions: 0.1 μL of sample (20%
soln. for essential oils in ethanol), 150:1 split ratio, initial oven
temperature of 40 °C with an initial hold time of 5 min, oven ramp rate of 4.5
°C per minute to 310 °C with a hold time of 5 min, helium carrier gas. The
electron ionization energy was 70 eV, scan range 35–650 amu, scan rate 2.4
scans per second, source temperature 230 °C, and quadrupole temperature 150 °C.
Volatile compounds were identified using the Adams volatile oil library
(version 4) [28] using Chemstation library
search in conjunction with retention indices. Note that
limonene/β-phellandrene and epi-α-cadinol/epi-α-muurolol elute as unresolved
peaks. Their ratios were determined by the ratio of masses 41, 68, 93
(limonene), 65, 77, 93 (β-phellandrene) and 81, 105, 161 (epi-α-cadinol), 43,
95, 121 (epi-α-muurolol), respectively. Volatile
compounds were quantified and are reported as a relative area percent by GC/FID
using an Agilent 7890B and Agilent J&W DB-5, 60 m x 0.25 mm, 0.25 μm film
thickness, fused silica capillary column. Operating conditions: 0.1 μL of
sample (20% soln. for essential oils in ethanol, 1% for reference compounds in
ethanol, 0.1% soln. for C7–C30 alkanes in hexane), 25:1 split injection,
initial oven temperature at 40 °C with an initial hold time of 2 min, oven ramp
rate of 3.0 °C per minute to 250 °C with a hold time of 3 min, helium carrier
gas. Essential oil samples were analyzed in triplicate by GC/FID to ensure
repeatability (standard deviation < 1 for all compounds). Compounds were
assigned using retention indices coupled with the retention time data of
reference compounds (MilliporeSigma, Sigma-Aldrich, St. Louis, USA).
The percent
yield was calculated as the ratio of the mass of processed plant material
immediately before distillation to the mass of essential oil produced,
multiplied by 100.
Enantioselective analysis was performed on chiral compounds that had an average area > 2% (achiral profile). Essential oils were analyzed, and chiral pairs identified, by GC/MS using an Agilent 7890B GC/5977B MSD (Agilent Technologies, Santa Clara, CA, USA) and Restek Rt-β, 30 m x 0.32 mm, 0.25 μm film thickness, fused silica capillary column. Operating conditions: 0.2 μL of sample (0.5% soln. for essential oils in ethanol), 25:1 split ratio, initial oven temperature of 40 °C with an initial hold time of 20 min, oven ramp rate of 2.0 °C per minute to 140 °C with a hold time of 35 min, second oven ramp rate of 30.0 °C per minute to 230 °C with a hold time of 2 min, helium carrier gas. The electron ionization energy was 70 eV, scan range 35–650 amu, scan rate 2.4 scans per second, source temperature 230 °C, and quadrupole temperature 150 °C. Volatile compounds were identified using the Adams volatile oil library (version 4) [28] using Chemstation library search. Chiral pairs were quantified and are reported as enantiomeric excess (ee %) by GC/FID using an Agilent 7890B and Restek Rt-β, 30 m x 0.32 mm, 0.25 μm film thickness, fused silica capillary column. Operating conditions: 0.2 μL of sample (0.5% or 2% soln. for essential oils in ethanol, 0.1% for enantiopure reference compounds in ethanol), 10:1 split injection, initial oven temperature at 40 °C with an initial hold time of 20 min, oven ramp rate of 2.0 °C per minute to 140 °C with a hold time of 35 min, second oven ramp rate of 30.0 °C per minute to 230 °C with a hold time of 2 min, helium carrier gas. Essential oil samples were analyzed in triplicate by GC/FID to ensure repeatability (standard deviation < 0.5 when calculating ee % for each chiral pair). Enantiopure reference standards were used for (-)-α-pinene, (+)-α-pinene, (+)-β-pinene, (-)-β-pinene, (+)-δ-3-carene, (S)-(-)-limonene, (R)-(+)-limonene (MilliporeSigma, Sigma-Aldrich, St. Louis, USA). Black pepper (Piper nigrum L.) essential oil was used for identification of the (-)-δ-3-carene enantiomer (Young Living Essential Oils, Lehi, USA).
The enantiomeric composition of δ-3-carene and α-pinene in the common
juniper essential oil samples was also measured with MRR, using the chiral
tagging technique. In these measurements, the analytes of interest are mixed
with a small chiral molecule (tag) of known enantiomeric composition to prepare
noncovalently bound diastereomeric complexes in the gas phase. These complexes,
which have different mass distributions, can be resolved using MRR [26]. First, a broadband MRR spectrometer was used
to characterize the MRR spectrum of each compound and to select the appropriate
chiral tag. Racemic δ-3-carene (MilliporeSigma, Sigma-Aldrich, St. Louis, USA)
and a commercial sample of (-)-a-pinene (TCI America, Portland, USA) were used for
this screening. Neon carrier gas, pre-mixed with the chiral tag of interest at
approx. 0.1% concentration, was seeded with the vapor from the analyte sample.
The samples had enough vapor pressure at room temperature to achieve sufficient
analyte concentration in the gas phase. The resulting gas mixture was then
injected into a high vacuum chamber through a pulsed supersonic expansion
nozzle to create a rotationally cold sample for analysis. The structures of the
resulting non-covalent complexes formed between the analyte and chiral tag are
determined by comparison of the experimentally derived rotational constants to
those calculated from quantum chemical calculations using dispersion-corrected
density functional theory (B3LYP-GD3BJ/def2TZVP) [29].
For δ-3-carene, 2,2,2-trifluoroisopropanol (TFIP) was used as the chiral
tag, while propylene oxide (PO) was used for the α-pinene enantiomer
measurement. A racemic tag sample was used in these measurements to generate
the two diastereomeric complexes of analyte and tag at equal concentration.
After this initial characterization, the targeted IsoMRR spectrometer
(BrightSpec, Inc., Charlottesville, USA) was used to measure chiral purity of
these two analytes in the common juniper oil samples [26].
The IsoMRR instrument can run measurements more quickly, and with much
lower sample consumption, due to the use of a cavity to enhance the measurement
sensitivity. In these measurements, 5 μL of the neat oil was injected into the
sample inlet, which was held at 30°C. Strong lines of the two diastereomeric
complexes of the analyte and tag were measured to determine the enantiomeric
composition of each analyte in the sample. The instrument response to the two
complexes was calibrated by additionally measuring the analyte signal levels
using a racemic tag sample. The total measurement time per sample was
approximately 18 minutes for each analyte. Additional details of the analyses
can be found in Supplementary
Table S1 –S8 and Figure S1- S2.
3. Results and
discussion
The aromatic profile of Juniperus
communis var. depressa is detailed in Table 1. Essential oil samples
were analyzed in triplicate to ensure reproducibility (standard deviation <
1 for all compounds). Prominent compounds (defined as averages > 2%) detected included α-pinene (63.9%),
β-pinene (6.2%), myrcene (6.9%), δ-3-carene (6.8%), and limonene (3.3%),
averaged over all samples. The achiral profile established herein shows
similarities to previously established results for J. communis var. depressa
from New Mexico, with α-pinene (53.9%), β-pinene (5.5%), myrcene (4.1%),
δ-3-carene (9.3%), and limonene (2.6%) also comprising most of the essential
oil profile [14]. However, from the same
study, J. communis (common juniper) samples of North American origin
from different taxonomic varieties demonstrated widely varying profiles.
Similar findings were observed when comparing the profiles from the current
study to those of European and North African origin, which sources are of
economic relevance. Previously published literature revealed that most sources
of common juniper analyzed (Algeria, Austria, Estonia, Italy, Lithuania,
Poland) share some achiral profile similarities, with prominent compounds and
values largely being α-pinene (10.3-90%), myrcene (1.8-52.4%), and limonene
(0.2-15.8%) [16-21]. However, many common juniper
samples also contained additional or different prominent volatile compounds
such as sabinene (12.4-42.5%) [16, 19, 20], β-phellandrene
(19.1%) [19], γ-terpinene (11.8%) [19], terpinene-4-ol (14.1%) [16], (E)-caryophyllene (10.3-11.4%) [18,20], and/or caryophyllene oxide (17.9%) [18]. The variability in these profiles was
credited to sample origin, chemotype, morphotype, and/or plant part from which
the essential oil was extracted [16-21].
Table 1. Aromatic profile
of Juniperus communis var depressa essential oil (n = 3).
|
KI |
Compound |
A |
B |
C |
|
849 |
Ethyl isovalerate |
t |
t |
t |
|
869 |
Isopentyl acetate |
t |
t |
t |
|
921 |
Tricyclene |
0.1 |
0.1 |
0.1 |
|
924 |
α-Thujene |
t |
t |
t |
|
932 |
α-Pinene |
66.8 |
63.9 |
61.1 |
|
945 |
α-Fenchene |
0.4 |
0.4 |
0.4 |
|
946 |
Camphene |
0.6 |
0.6 |
0.6 |
|
953 |
Thuja-2,4(10)-diene |
0.1 |
0.1 |
0.1 |
|
969 |
Sabinene |
0.3 |
0.3 |
0.3 |
|
974 |
β-Pinene |
6.3 |
6.1 |
6.2 |
|
988 |
Myrcene |
6.1 |
7.4 |
7.1 |
|
997 |
Ethyl hexanoate |
t |
t |
t |
|
1001 |
δ-2-Carene |
0.3 |
0.2 |
0.2 |
|
1002 |
α-Phellandrene |
0.1 |
0.1 |
0.1 |
|
1005 |
o-Cresol methyl ether |
0.1 |
0.1 |
0.1 |
|
1008 |
δ-3-Carene |
6.0 |
6.3 |
8.0 |
|
1014 |
α-Terpinene |
0.1 |
0.1 |
0.1 |
|
1020 |
p-Cymene |
0.1 |
0.1 |
0.1 |
|
1024 |
Limonene |
2.9 |
3.6 |
3.4 |
|
1025 |
β-Phellandrene |
1.2 |
1.1 |
1.2 |
|
1032 |
(Z)-β-Ocimene |
t |
t |
t |
|
1044 |
(E)-β-Ocimene |
t |
t |
t |
|
1054 |
γ-Terpinene |
0.1 |
0.1 |
0.1 |
|
1085 |
p-Mentha-2,4(8)-diene |
0.1 |
0.1 |
0.1 |
|
1086 |
Terpinolene |
0.8 |
0.9 |
1.0 |
|
1095 |
Linalool |
0.2 |
0.3 |
0.2 |
|
1102 |
Isopentyl isovalerate |
t |
0.1 |
0.1 |
|
1112 |
3-Methyl-3-butenyl-3-methyl-butanoate |
0.1 |
0.1 |
0.1 |
|
1122 |
α-Campholenal |
0.1 |
0.1 |
0.2 |
|
1135 |
trans-Pinocarveol |
0.1 |
0.1 |
0.1 |
|
1137 |
cis-Verbenol |
t |
t |
t |
|
1140 |
trans-Verbenol |
0.1 |
0.1 |
0.1 |
|
1148 |
Citronellal |
0.1 |
0.1 |
0.1 |
|
1165 |
Borneol |
t |
t |
t |
|
1166 |
p-Mentha-1,5-dien-8-ol |
t |
t |
0.1 |
|
1172 |
cis-Pinocamphone |
t |
t |
t |
|
1174 |
Terpinen-4-ol |
0.1 |
0.1 |
0.1 |
|
1179 |
p-Cymen-8-ol |
t |
t |
t |
|
1186 |
α-Terpineol |
0.1 |
0.1 |
0.1 |
|
1194 |
Myrtenol |
t |
t |
t |
|
1195 |
Myrtenal |
t |
t |
t |
|
1223 |
Citronellol |
0.2 |
0.2 |
0.3 |
|
1232 |
Thymol methyl ether |
t |
t |
t |
|
1249 |
Geraniol |
t |
t |
t |
|
1257 |
Methyl citronellate |
0.2 |
0.2 |
0.2 |
|
1284 |
Bornyl acetate |
0.4 |
0.5 |
0.5 |
|
1324 |
Myrtenyl acetate |
0.7 |
0.7 |
0.9 |
|
1346 |
α-Terpinyl acetate |
0.6 |
0.8 |
0.9 |
|
1350 |
Citronellyl acetate |
0.1 |
0.1 |
0.1 |
|
1356 |
Eugenol |
0.1 |
0.1 |
0.1 |
|
1359 |
Neryl acetate |
0.1 |
0.1 |
0.1 |
|
1379 |
Geranyl acetate |
0.2 |
0.2 |
0.2 |
|
1385 |
trans-Myrtanol acetate |
0.1 |
0.1 |
0.1 |
|
1389 |
β-Elemene |
0.3 |
0.3 |
0.3 |
|
1417 |
(E)-Caryophyllene |
0.1 |
0.1 |
t |
|
1434 |
γ-Elemene |
0.1 |
0.1 |
0.1 |
|
1452 |
α-Humulene |
0.1 |
0.1 |
t |
|
1478 |
γ-Muurolene |
t |
t |
0.1 |
|
1480 |
Germacrene D |
0.2 |
0.3 |
0.3 |
|
1489 |
β-Selinene |
t |
t |
t |
|
1500 |
α-Muurolene |
0.4 |
t |
t |
|
1505 |
β-Bisabolene |
0.1 |
0.1 |
0.2 |
|
1513 |
γ-Cadinene |
0.1 |
0.1 |
0.2 |
|
1522 |
δ-Cadinene |
0.3 |
0.3 |
0.4 |
|
1537 |
α-Cadinene |
t |
t |
t |
|
1559 |
Germacrene B |
t |
t |
t |
|
1561 |
(E)-Nerolidol |
0.1 |
0.1 |
0.1 |
|
1574 |
Germacrene D-4-ol |
0.1 |
0.1 |
0.1 |
|
1577 |
Spathulenol |
t |
t |
0.1 |
|
1608 |
Humulene epoxide II |
t |
t |
t |
|
1638 |
epi-α-Cadinol |
t |
t |
t |
|
1640 |
epi-α-Muurolol |
t |
t |
t |
|
1644 |
α-Muurolol |
t |
t |
t |
|
1652 |
α-Cadinol |
0.1 |
0.1 |
0.1 |
|
1685 |
α-Bisabolol |
0.2 |
0.2 |
0.3 |
Each sample
is referred to by a letter, A-C. The Kovat’s Index (KI), volatile compound
name, and compound average area % for each sample are provided. Each essential
oil sample was analyzed in triplicate to ensure reproducibility (Standard
Deviation < 1 for all values). Values less than 0.1% are denoted as trace
(t). The KI values were previously calculated and obtained using a linear
calculation on DB-5 column [28].
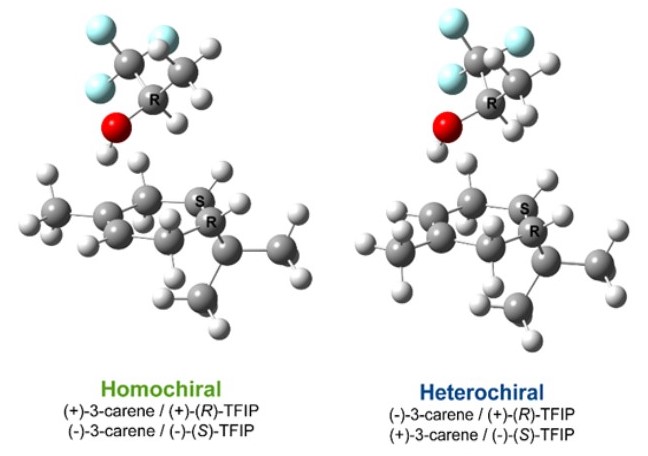
Enantiopure
reference standards were commercially available for 3 of 4 prominent (defined
as averages > 2%) chiral pairs found in these essential oils but was not
available for δ-3-carene. Chiral tagging molecular rotational resonance (MRR)
was therefore used to perform the chiral analysis of δ-3-carene in common
juniper essential oil. Fig. 1 illustrates the analysis of δ-3-carene by
chiral tagging MRR. The geometries of the non-covalent complexes of δ-3-carene
and TFIP used in the analysis are shown. These complexes are named either as
homochiral (where the optical rotations of the analyte and tag are the same,
e.g. (+)-δ-3-carene / (+)-(R)-TFIP) or heterochiral (where the optical
rotations are different[1]).
When a racemic chiral tag ((RS)-TFIP) is used, the signals of the two
complexes are approximately the same; but in the measurement with enantiopure (R)-TFIP,
only the homochiral complex is observed, allowing us to conclude that
(+)-δ-3-carene is the major enantiomer in common juniper oil. Due to the low
fractional composition of δ-3-carene in the common juniper oil and the
resulting reduced signal intensity in the chiral tag complexes, we were not
able to detect the weaker enantiomer of δ-3-carene and are only able to
determine a lower limit of ee% >85%.
Figure 1. Chiral analysis of δ-3-carene (3-carene) by chiral tagging MRR. The complex geometries of
the assigned spectra that are used for the analysis are shown in the left
panel. The middle panel shows the measurement of a standard of δ-3-carene with racemic TFIP, which is used to
normalize the instrument response. The right panel shows the measurement of the
common juniper EO sample with (R)-TFIP (ee=94.6%).
To
illustrate the similarity between values from conventional techniques (GC/MS
and GC/FID) and the novel application of chiral tagging MRR for determining
chiral profiles, ee% was determined for α-pinene using both techniques (Table
2). The results show the same trend in enantiomeric excess between the three
fractions. Values (ee%) compared between each technique agree within 6% or
less. We noted a systematic offset between the GC/FID and MRR results of
approximately 5% but were not able to determine the source of this difference.
Previous studies have validated the quantitative accuracy of MRR in comparison
to chiral GC [26,
30]. Determining the systematic
offset between values is beyond the scope of the current study and will be
explored in future studies.
Table 2. Enantiomeric excess (ee%) of α-pinene in
Juniperus communis var. depressa essential oil determined
by GC/FID and
molecular rotational resonance (MRR).
|
Method |
Enantiomer |
A |
B |
C |
|
GC/FID |
(-)-α-Pinene |
67.7 |
62.1 |
71.1 |
|
(+)-α-Pinene |
- |
- |
- |
|
|
MRR |
(-)-α-Pinene |
62.8 |
56.7 |
67.9 |
|
(+)-α-Pinene |
- |
- |
- |
|
|
Each sample (n = 3) is referred to by a letter, A-C. Each
essential oil sample was analyzed in triplicate to ensure repeatability
(standard deviation for analysis by GC/FID ≤ 0.3 for repeat injections;
standard deviation for analysis by MRR < 3% for repeat injections). Values
(ee%) between techniques agree within 6% or less. |
||||
Using ee% data (MRR) and elution order of compounds (GC/MS) from the current study, ee% was determined for δ-3-carene by GC/FID. In addition, and using enantiopure reference standards, chiral pairs were analyzed in the current study for α-pinene, β-pinene, and limonene. In these samples (-)-α-pinene, (-)-β-pinene, (+)-δ-3-carene, and (R)-(+)-limonene were the prominent enantiomers (Table 3). For β-pinene, comparable results were found in samples of European and North African origin, where (-)-β-pinene was the prominent enantiomer [16, 17, 19]. However, enantiomeric prominence appears to switch between (-)/(+)-α-pinene and (S)-(-)/(R)-(+)-limonene [16-19, 21, 22] depending on sample origin, chemotype, morphotype, and/or plant part from which the essential oil was extracted. To the authors’ best knowledge, this is the first time that chiral analysis of δ-3-carene has been performed in common juniper samples of any region.
Table 3. Enantiomeric excess of chiral compounds
that had an average area % (achiral profile)
> 2%
for Juniperus communis var. depressa.
|
Enantiomer |
A |
B |
C |
|
(-)-α-Pinene |
67.7 |
62.1 |
71.1 |
|
(+)-α-Pinene |
- |
- |
- |
|
(+)-β-Pinene |
- |
- |
- |
|
(-)-β-Pinene |
95.4 |
94.8 |
94.8 |
|
(+)-δ-3-Carene |
>99.0 |
>99.0 |
>99.0 |
|
(-)-δ-3-Carene |
- |
- |
- |
|
(S)-(-)-Limonene |
- |
- |
- |
|
(R)-(+)-Limonene |
39.5 |
48.4 |
42.3 |
|
Chiral ratios (calculated as ee%) determined by GC/FID. Each sample (n
= 3) is referred to by a letter, A-C. Each essential oil sample was
analyzed in triplicate to ensure repeatability (standard deviation for
analysis by GC/FID ≤ 0.3 for all compounds). For δ-3-carene, the
(-)-δ-3-carene enantiomer was not detected in any sample. The value
(>99.0) was determined based on the GC/FID limit of detection. |
|||
In
addition to investigating the chiral profile of common juniper samples, the
current study verifies chiral chromatography from previous studies on other
plant species. While (+)-δ-3-carene enantiopure reference standards are commercially
available, researchers have historically relied on natural standards of black
pepper (Piper nigrum)
essential oil to reference the (-)-δ-3-carene enantiomer [31-34]. The use of MRR in this
study verifies that authentic and natural sources of black pepper essential oil
can be used as a reference for (-)-δ-3-carene.
Essential
oil yield is detailed in Table 4. The average essential oil yield for J.
communis var. depressa is
0.14% (w/w). Yields from the current study are lower than those of European and
North African origin (0.2-1.6%) [16,
20, 22, 23].
Table 4. Yield data, including weight of plant material distilled (g), essential oil yield (g),
and calculated essential oil yield (%)
for Juniperus communis var. depressa samples (n=3).
|
Plant Name |
Plant Sample |
Plant Material Weight (g) |
Essential Oil Yield (g) |
Essential Oil Yield (%) |
|
J. communis var. depressa |
A |
1058.83 |
1.53 |
0.14 |
|
B |
1044.73 |
1.46 |
0.14 |
|
|
C |
1042.88 |
1.27 |
0.12 |
|
|
Avg. |
1048.81 |
1.42 |
0.14 |
|
|
Avg. RSD (n = 3) |
|
8.85 |
||
|
Each sample is referred to by a letter, A-C. The
average calculated yield for samples is 0.14%. The relative standard
deviation (RSD) is provided for essential oil yield. |
||||
To the authors’ knowledge, this is the
first time that the achiral and chiral profiles of Juniperus communis var.
depressa of Utahn origin has been fully detailed.
4. Conclusions
The achiral profile established herein
is similar to that of J. communis var. depressa from New Mexico, and
both the achiral and chiral profiles from the current study show similarities to
profiles of other varieties of J. communis from European and North
African origin. However, previous studies have shown that there is substantial
variation in both achiral and chiral profiles of J. communis essential
oil from commercially important sources (Europe and North Africa). The profiles
established herein provide fundamental data for understanding the potential use
of J. communis of North American origin in the flavor and fragrance
industry. Given that juniper cones (berries) are highly sought after in the
flavor and fragrance industry, essential oil extracted from only cones
(berries) of J. communis var. depressa should be investigated in
future studies.
The current study also demonstrates
the utility and practicality of using molecular rotational resonance (MRR) for
determining chiral profiles in essential oils. Since a complete understanding
of the achiral and chiral profiles are inherent to the utility of essential
oils, MRR is a novel technique that could have important applications in the flavor
and fragrance industry.
Footnotes
[1]In other chiral
tagging MRR studies it is more common to use the Cahn-Ingold-Prelog
nomenclature to name complexes as homochiral or heterochiral; however, as
δ-3-carene has two chiral centers with opposite nomenclature, we use the
optical rotations instead.
Supplementary Data
DOI link: https://doi.org/10.58985/jeopc.2023.v01i03.31
Authors’ contributions
Conceptualization,
A.P., T.M.W., and R.E.C.; methodology, A.P., T.M.W., R.E.S., and A.D.;
software, A.P., T.M.W., R.E.S., and A.D.; validation, J.L.N and R.E.C..; formal
analysis, A.P., T.M.W., R.E.S., and A.D.; investigation, A.P. and T.M.W.; data
curation, T.M.W. and R.E.S.; writing—original draft preparation, A.P., T.M.W.,
R.E.S, and J.L.N; writing—review and editing, A.P., A.D., R.E.S., J.L.N., and
R.E.C.; funding acquisition, R.E.C. All authors have read and agreed to the
published version of the manuscript.
Acknowledgements
The authors wish to thank the following
individuals and organizations for their assistance with the project: Chris
Bowerbank (YLEO), Chris Packer (YLEO), Emma A. Ziebarth (YLEO), Erin Riggs
(UVU), Mackenzie G. Dey (USU), Sheldon Giles (YLEO), Skyler Olson (YLEO), for
their assistance, BrightSpec for their collaboration, the Skyrider Wilderness
Ranch for permitting research on private land, and the D. Gary Young Research
Institute for providing support for this project.
Funding
This research was
funded by Young Living Essential Oils.
Availability of data and materials
All data will be
made available on request according to the journal policy.
Conflicts of interest
Authors R.E.S. and
J.L.N. have equity in BrightSpec, Inc.
References
1.
Flora of North America Editorial Committee (Eds.). Flora of North
America North of Mexico, Vol. 2. Oxford University Press: New York, NY, USA,
1993.
2.
Guenther, E. The essential oils, Vol. VI. Robert E. Kreiger
Publishing Co. Inc.: Huntington, NY, USA, 1952.
3.
Adams, R.P. Junipers of the World: The genus Juniperus. 4th
ed., Trafford Publ., Bloomington, IL., 2014.
4.
Adams, R.P.; Nguyen, S. Post-Pleistocene Geographic Variation in Juniperus
communis in North America. Phytologia. 2007, 89(1), 43-57.
5.
Adams, R.P.; Pandey, R.N.; Leverenz, J.W.; Dignard, N.; Hoegh, K.;
Thorfinnsson, T. Pan-Arctic variation in Juniperus communis: historical
biogeography based on DNA fingerprinting. Biochem. Syst. Ecol., 2003,
31, 181-192.
6.
Adams, R.P. Taxonomy of Juniperus communis in North
America: Insight from Variation in nrNDA SNPs. Phytologia. 2008, 90(2),
181-197.
7.
Adams, R.P.; Murata, J.; Takahashi, H.; Schwarzbach, A.E. Taxonomy
and Evolution of Juniperus communis: Insight from DNA Sequencing and
SNPs. Phytologia. 2011, 93(2), 185-197.
8.
Adams, R.P.; Pandey, R.N. Analysis of Juniperus communis and
its varieties based on DNA fingerprinting. Biochem. Syst. Ecol. 2003,
31, 1271-1278.
9.
Cronquist, K.; Holmgren, A.H.; Holmgren, N.H; Reveal, J.L.;
Holmgren, P.I. Intermountain Flora, Vascular Plants of the Intermountain West,
U.S.A. Vol. I. Hafner Publishing Company, Inc.: New York, NY, USA, 1972.
10.
Mozingo, H.N. Shrubs of the Great Basin: A Natural History, University
of Nevada Press, Nevada, USA, 1987.
11.
Van Buren, R.; Cooper, J.G.; Shultz, L.M.; Harper, K.T. Woody
plants of Utah: a field guide with identification keys to native and
naturalized trees, shrubs, cacti, and vines. Utah State University Press:
Logan, UT, USA, 2011.
12.
Welsh, S.L.; Atwood, N.D.; Goodrich, S.; Higgins, L.C. A Utah Flora,
Fifth Edition, revised. Brigham Young University: Provo, UT, USA, 2015.
13.
Sargent, C.S. Manual of the trees of North America, Vol. I.
Dover Publications, Inc.: New York, NY, USA, 1965.
14.
Adams, R.P.; Beauchamp, P.S.; Dev, V.; Bathala, R.M. The Leaf Essential
Oils of Juniperus communis L. Varieties in North America and the NMR and
MS Data for Isoabienol. J. Essent. Oil Res., 2010, 22(1), 23-28.
15.
Butkienė, R.; Nivinskienė, O.; Mockutė, D. Differences in the essential
oils of the leaves (needles), unripe and ripe berries of Juniperus communis
L. growing wild in Vilnius district (Lithuania). J. Essent. Oil Res. 2006,
18(5), 489-494.
16.
Dahmane, D.; Dob, T.; Chelghoum, C. Essential oil composition and
enantiomeric distribution of some monoterpenoid components of Juniperus
communis L. from Algeria. J. Essent. Oil Res., 2016, 28(4),
348-356.
17.
Foudil-Cherif, Y.; Yassaa, N. Enantiomeric and non-enantiomeric
monoterpenes of Juniperus communis L. and Juniperus oxycedrus
needles and berries determined by HS-SPME and enantioselective GC/MS. Food
Chem. 2012, 135(3), 1796-1800.
18.
Ložienė, K.; Labokas, J.; Venskutonis, P.R.; Maždžierienė, R.
Chromatographic evaluation of the composition of essential oil and α-pinene
enantiomers in Juniperus communis L. berries during ripening. J.
Essent. Oil Res., 2010, 22(5), 453-458.
19.
Ochocka, J.R.; Asztemborska, M.; Zook, D.R.; Sybilska, D.; Perez,
G.; Ossicini, L. Enantiomers of monoterpenic hydrocarbons in essential oils
from Juniperus communis. Phytochem. 1997, 44(5), 869-873.
20.
Orav, A.; Kailas, T.; Müürisepp, M. Chemical investigation of the
essential oil from berries and needles of common juniper (Juniperus communis
L.) growing wild in Estonia. Nat. Prod. Res., 2010, 24(19),
1789-1799.
21.
Sybilska, D.; Asztemborska, M.; Kowalczyk, J.; Ochocka, R.J.;
Ossicini, L.; Perez, G. Enantiomeric composition of terpenic hydrocarbons in
essential oils from Juniperus communis L. J. Chromatogr.
A, 1994, 659(2), 389-394.
23.
Ložienė, K.; Labokas, J. Effects of abiotic environmental
conditions on amount and enantiomeric composition of α-pinene in Juniperus
communis L. Biochem. Syst. Ecol., 2012, 44, 36-43.
24.
Joyce, L.A.; Schultz, D.M.; Sherer, E.C.;
Neill, J.L.; Sonstrom, R.E.; Pate, B.H. Direct regioisomer analysis of crude
reaction mixtures via molecular rotational resonance (MRR) spectroscopy. Chem.
Sci. 2020, 11, 6332-6338.
25.
Neill, J.L.; Yang, Y.; Muckle, M.T.;
Reynolds, R.L.; Evangelisti, L.; Sonstrom, R.E.; Pate, B.H.; Gupton B.F. Online
stereochemical process monitoring by molecular rotational resonance
spectroscopy. Org. Process Res. Dev. 2019, 23, 1046-1051.
26.
Sonstrom, R.E.; Neill, J.L.; Mikhonin,
A.V.; Doetzer, R.; Pate, B.H. Chiral analysis of pantolactone with molecular
rotational resonance spectroscopy. Chirality. 2022, 34, 114-125.
27.
Sonstrom, R.E.; Cannon, D.M.; Neill, J.L.
Chiral analysis of linalool, an important natural fragrance and flavor
compound, by molecular rotational resonance spectroscopy. Symmetry. 2022, 14,
917.
28.
Adams, R.P. Identification of Essential Oil Components by Gas
Chromatography/Mass Spectrometry, 4th Edn.; Allured Publ.: Carol
Stream, IL, USA, 2007.
29.
Mayer, K.; West, C.; Marshall, F.E.; Sedo, G.; Grubbs, G.S.;
Evangelisti, L.; Pate, B.H. Accuracy of quantum chemistry structures of chiral
tag complexes and the assignment of absolute configuration. Phys. Chem. Chem.
Phys. 2022, 24 (45), 27705-27721. https://doi.org/10.1039/D2CP04060C.
30.
Wilson, T.M.; Davis, A.; Sonstrom, R.E.; Neill, J.L.; Ziebarth,
E.A.; Poulson, A.; Carlson, R.E. Essential oil composition and enantioselective
profile of Agastache urticifolia (Lamiaceae) and Monardella
odoratissima (Lamiaceae) from Utah. Molecules 2023, 28,
2249. https://doi.org/10.3390/molecules28052249.
31.
Fäldt, J.; Martin, D.; Miller, B.; Rawat, S.; Bohlmann, J.
Traumatic resin defense in Norway spruce (Picea abies): Methyl jasmonate-induced
terpene synthase gene expression, and cDNA cloning and functional
characterization of (+)-3-carene synthase. Plant Mol. Biol. 2003, 51,
119-133.
32.
Sjödin, K.; Persson, M.; Borg-Karlson, A.K.; Norin, T.
Enantiomeric compositions of monoterpene hydrocarbons in different tissues of
four individuals of Pinus sylvestris. Phytochem. 1996, 41(2),
439-445.
33.
König, W.A.; Krüger, A.; Icheln, D.; Runge, T. Enantiomeric
composition of the chiral constituents in essential oils. Part 1: Monoterpene
hydrocarbons. J. High Resolut. Chromatogr., 1992, 15(3), 184-189.
34. Ganjitabar, H.; Hadidi, R.; Garcia, G.A.; Nahon, L.; Powis, I. Vibrationally-resolved photoelectron spectroscopy and photoelectron circular dichroism of bicyclic monoterpene enantiomers. J. Mol. Spectroscop. 2018, 353, 11-19.
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
Abstract
Juniperus
communis var depressa (common juniper) is an essential oil-bearing
evergreen shrub native to North America, including the state of Utah. Cones
(berries), leaves, and limb material were collected, and steam distilled
together. The resulting essential oil samples (n = 3) were analyzed, and
both the achiral and chiral profiles established by GC/FID, GC/MS,
enantioselective GC/FID, and molecular rotational resonance (MRR). Prominent
compounds of the achiral profile include (averages) α-pinene (63.9%), β-pinene
(6.2%), myrcene (6.9%), δ-3-carene (6.8%), and limonene (3.3%). Four prominent
chiral pairs (α-pinene, β-pinene, δ-3-carene, limonene) were analyzed to
determine the enantioselective profile. Where enantiopure standards were not
commercially available, (-)-δ-3-carene, MRR was used as a quick and reliable
analytical technique for chiral analysis. This study verifies the achiral
profile for North American common juniper and, for the first time to the
authors’ knowledge, establishes the achiral and chiral profiles for J.
communis var. depressa, specifically from Utah. This study
confirms the utility and practicality of using MRR for determining chiral
profiles in essential oils. Additionally, results provide a foundation for
future research in the flavor and fragrance industries for common juniper of
North American origin.
Abstract Keywords
Cupressaceae, enantioselective
profile, essential oil composition, gas chromatography, Juniperus communis var.
depressa, molecular rotational resonance (MRR)
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
Editor-in-Chief
This work is licensed under the
Creative Commons Attribution 4.0
License.(CC BY-NC 4.0).


