Research Article
Kathy Swor
Kathy Swor
Independent
Researcher, 1432 W. Heartland Dr., Kuna, ID 83634, USA
Ambika Poudel
Ambika Poudel
Aromatic
Plant Research Center, 230 N 1200 E, Suite 100, Lehi, UT 84043, USA
Prabodh Satyal
Prabodh Satyal
Aromatic
Plant Research Center, 230 N 1200 E, Suite 100, Lehi, UT 84043, USA
William N. Setzer
William N. Setzer
Corresponding
Author
Aromatic
Plant Research Center, 230 N 1200 E, Suite 100, Lehi, UT 84043, USA
And
Department of Chemistry, University of Alabama in Huntsville, Huntsville, AL 35899, USA
E-mail: setzerw@uah.edu, wsetzer@chemistry.uah.edu; Tel.: +1-256-468-2862
Abstract
Juniperus
osteosperma (Utah juniper) is an abundant tree
within its range in the Great Basin. It is an important source of food for
birds and mule deer and has served as traditional medicine for Native
Americans. The purpose of this work is to examine essential oils of J. osteosperma
growing in the northern limits of its range and to evaluate the enantiomeric
distribution of chiral monoterpenoids. Monoterpene hydrocarbons (23.7-31.3%)
and oxygenated monoterpenoids (58.5-70.1%) made up the bulk of the essential
oil compositions. The main constituents in J. osteosperma essential oils
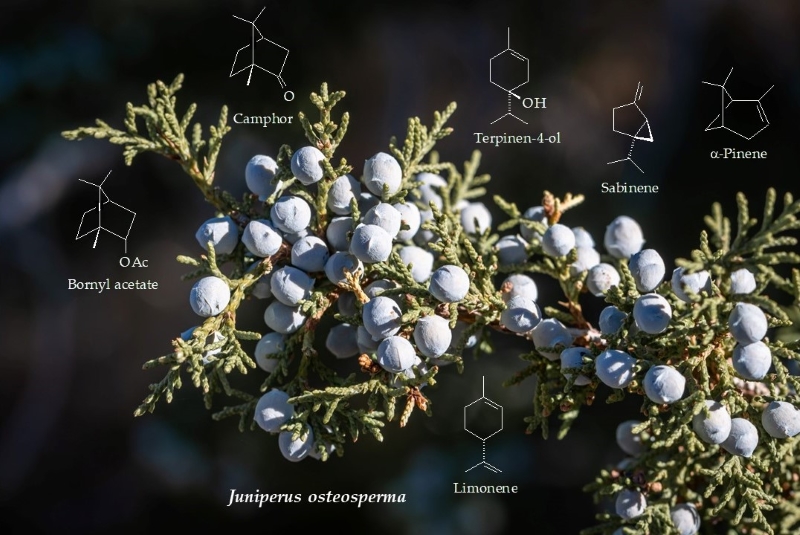
were bornyl acetate (9.3-27.0%), camphor (3.9-31.1%), terpinen-4-ol
(7.9-15.3%), sabinene (4.3-12.3%), α-pinene (3.9-9.6%), limonene (2.6-5.2%),
borneol (1.6-4.2%), trans-verbenol (up to 10.2%), p-cymene
(1.2-4.9%), α-elemol
(1.2-6.3%), and γ-terpinene
(1.4-2.0%). Chiral GC-MS revealed (+)-α-pinene (³
99.5%), (+)-camphene (³
93.0%), (+)-sabinene (100%), (+)-β-pinene (100%), (+)-limonene (³ 97.7%), (+)-cis-sabinene
hydrate (³ 93.7%), (+)-trans-sabinene
hydrate (³ 95.8%),
(+)-camphor (³ 98.3%),
(+)-α-terpineol (100%), and (+)- verbenol (100%) to be the predominant
enantiomers. The essential oil compositions of J. osteosperma from
southern Idaho are similar to those from California, Nevada, and Utah, with bornyl
acetate, sabinene, terpinen-4-ol and camphor dominating the essential oils. The
reported biological activities of the major components are consistent with the
Native American traditional uses of the plant.
Abstract Keywords
Utah
juniper, Cupressaceae, chiral, enantiomer, multivariate analysis, gas
chromatography
1. Introduction
Juniperus
osteosperma (Torr.) Little, Cupressaceae (Utah
juniper) is a conspicuous and abundant tree in the Great Basin. The tree grows
to 4 m tall with scalelike leaves, female cones that turn blue when mature
(6-12 mm), and bark that shreds (Fig. 1) [1]. The tree occurs occasionally in
southern Idaho and southern Montana and ranges south through Nevada, Utah,
western Colorado, and into Arizona and southeastern California (Fig. 2) [2]. Utah juniper is an important source of
cover and food for wildlife, including bird species such as western screech owl
(Megascops kennicottii Elliot), gray flycatcher (Empidonax wrightii
S.F. Baird), Woodhouse’s scrub jay (Aphelocoma woodhouseii S.F. Baird), juniper
titmouse (Baeolophus ridgwayi Richmond), chipping sparrow (Spizella
passerina Bechstein), dark-eyed junco (Junco hyemalis Linnaeus), and
warbling vireo (Vireo gilvus Vieillot) [2, 3]. Rodents such as desert woodrat (Neotoma
lepida Thomas) and pinyon pine mouse (Peromyscus truei Shufeldt)
rely on J. osteosperma for food [4–6]. The foliage is browsed by mule deer (Odocoileus
hemionus Rafinesque) and elk (Cervus canadensis Erxleben) during
deep snow when other food sources are scarce [7]. Several Native American tribes (e.g.,
Paiute, Shoshoni) have used Utah juniper to treat colds and coughs, rheumatism,
and skin infections [8]. Samples from southern Idaho are near
the northern limit of the range, so the purpose of this investigation is to
compare the compositions from Idaho with essential oil samples from other
geographical regions as well as to evaluate the distribution of enantiomeric
monoterpenoids.
Figure
1.
Juniperus osteosperma from southern Idaho. A: Foliage (leaves,
female and male cones). B: Bark.
Figure 2. Range map of Juniperus
osteosperma (U.S. Geological Survey, Public domain, via Wikimedia Commons,
https://commons.wikimedia.org/wiki/File:Juniperus_osteosperma_range_map.jpg
Accessed on 11 November 2023)
2.
Materials and methods
2.1.
Plant material
Foliage
was obtained from six individual trees growing near Juniper, Idaho, in April
2023. For comparison, one sample from southern Utah (near Toquerville, Utah)
was also collected (see Table 1). The trees were identified in the field by
W.N. Setzer using a field guide [9]
and verified by comparison with herbarium samples from the Southern Utah
University Herbarium [10]. A voucher specimen (WNS-Jo-7049) has
been deposited in the University of Alabama in Huntsville herbarium. The fresh
foliage was frozen (–20
°C) until
hydrodistillation.
Table 1. Collection and essential oil extraction details of Juniperus osteosperma.
|
Sample number |
Collection date |
Collection location |
Mass foliage (g) |
Mass essential oil (g) |
Yield (%) |
|
#1* |
25 April 2023 |
42°4′29″ N,
112°54′44″W, 1453 m asl |
87.62 |
0.9445 |
1.078% |
|
#2 |
25 April 2023 |
42°4′30″ N,
112°54′44″W, 1453 m asl |
89.36 |
0.7803 |
0.873% |
|
#3* |
25 April 2023 |
42°4′32″ N,
112°54′44″W, 1454 m asl |
125.51 |
2.6575 |
2.117% |
|
#4 |
25 April 2023 |
42°4′32″ N,
112°54′44″W, 1454 m asl |
91.99 |
1.1703 |
1.272% |
|
#5* |
30 April 2023 |
42°4′21″ N,
112°54′14″W, 1449 m asl |
172.98 |
4.6847 |
2.708% |
|
#6 |
30 April 2023 |
42°4′21″ N,
112°54′14″W, 1449 m asl |
162.47 |
4.4478 |
2.738% |
|
#7*(sU) |
26 April 2023 |
37°16′56″ N,
113°18′33″W, 1168 m asl |
173.64 |
4.0849 |
2.353% |
|
Sample numbers with
an asterisk (*) had abundant female cones (berries). Sample #7*(sU) was
collected in southern Utah |
|||||
2.2.
Essential oil
For
each sample, the foliage (leaves, no “berries”) was hydrodistilled for four
hours using a Likens-Nickerson apparatus with continuous extraction of the
distillate with dichloromethane to give pale yellow essential oils (Table 1).
2.3.
Gas chromatographic analyses
The
foliar essential oils of J. osteosperma were analyzed by gas
chromatography as previously described [11]. GC-MS: Shimadzu GCMS-QP2010 Ultra
instrument (Shimadzu Scientific Instruments, Columbia, MD, USA), electron
impact (EI) mode (electron energy = 70 eV), scan range = 40–400 atomic mass
units, scan rate = 3.0 scans/s, and GC-MS solution software, ZB-5ms column
(Phenomenex, Torrance, CA, USA, 60 m length, 0.25 mm inner diameter, 0.25 μm
film thickness), He carrier gas (column head pressure = 208.2 kPa, flow rate =
2.0 mL/min, injector temperature = 260 °C, ion source temperature = 260 °C; GC
oven temperature program (50 °C initial temperature, increased at a rate of 2
°C/min to 260 °C, then held at 260 °C for 5 min. For each essential oil sample,
0.1 μL (5% w/v solution in dichloromethane) was injected, splitting mode =
24.5:1. Retention index (RI) values were calculated according to the linear
equation of van den Dool and Kratz [12]. The J. osteosperma components
were identified by comparing their RI values (within 10 RI units) and their MS
fragmentation patterns (> 80% similarity) with those reported in the Adams [13],
FFNSC3 [14], NIST20 [15], and Satyal [16]
databases. GC-FID: Shimadzu GC 2010 instrument with FID detector (Shimadzu
Scientific Instruments, Columbia, MD, USA), ZB-5 GC column (Phenomenex,
Torrance, CA, USA, 60 m ´
0.25 mm ´ 0.25 μm film
thickness), same operating conditions as above for GC-MS. The component
percentages were calculated from raw peak integration without standardization.
Chiral GC-MS: Shimadzu GCMS-QP2010S instrument (Shimadzu Scientific
Instruments, Columbia, MD, USA), Restek B-Dex 325 column (Restek Corp.,
Bellefonte, PA, USA, 30 m ´
0.25 mm diameter ´
0.25 μm film thickness), injector and detector temperatures = 240 °C. He
carrier gas (column head pressure = 53.6 kPa, flow rate = 1.00 mL/min); GC oven
temperature program (50 °C initial temperature held for 5 min, increased to 100
°C at a rate of 1.0 °C/min, then increased to 220 °C at a rate of 2 °C/min).
For each sample, 0.3 μL (5% w/v solution in dichloromethane) was injected,
splitting mode = 24.0:1. The enantiomers were determined by comparison of RI
values with authentic samples (Sigma-Aldrich, Milwaukee, WI, USA), which are
compiled in our own in-house database; enantiomer ratios were calculated from
raw peak areas.
2.4.
Multivariate analyses
Multivariate analyses were carried out using XLSTAT v. 2018.1.1.62926 (Addinsoft, Paris, France). Hierarchical cluster analysis (HCA) was carried out using the concentrations of the 12 most abundant components (bornyl acetate, camphor, terpinen-4-ol, sabinene, α-pinene, limonene, borneol, trans-verbenol, p-cymene, α-elemol, γ-terpinene, and β-phellandrene) from this study as well as previously reported compositions from the literature [17–21]. Dissimilarity was used to determine clusters considering Euclidean distance and Ward’s method was used to define agglomeration. Principal component analysis (PCA, type Covariance) was used to verify the similarity of essential oil samples based on the HCA analysis.
3.
Results and discussion
3.1.
Chemical composition
Hydrodistillation
of the foliage of J. osteosperma collected from southern Idaho gave pale
yellow essential oils in yields ranging from 0.873% to 2.738%. Gas
chromatographic analysis led to the identification of 106 chemical components
comprising 98.5-99.4% of the essential oil compositions (Table 2). The major
components in J. osteosperma essential oils were bornyl acetate
(9.3-27.0%), camphor (3.9-31.1%), terpinen-4-ol (7.9-15.3%), sabinene
(4.3-12.3%), α-pinene
(3.9-9.6%), limonene (2.6-5.2%), borneol (1.6-4.2%), trans-verbenol (up
to 10.2%), p-cymene (1.2-4.9%), α-elemol (1.2-6.3%), and γ-terpinene (1.4-2.0%).
Adams
and co-investigators have previously examined J. osteosperma leaf essential
oils from Nevada, northern Utah, southern California, and Arizona [17–20]; Wilson and co-workers have also
examined the essential oils of J. osteosperma from Utah, including
trunk, limb, leaf [21], and “berries” [22]. Based on the main components in the
essential oils from this study and those from the previous reports, a
hierarchical cluster analysis (HCA) and principal component analysis (PCA) were
carried out to place the chemical compositions into perspective. The HCA reveals
four well-defined chemical groupings (Fig. 3): A camphor/bornyl acetate group,
a camphor/terpinen-4-ol/trans-verbenol group, a bornyl acetate/sabinene
group, and a single camphor-rich sample. The PCA (Fig. 4) agrees with the HCA
and shows the three closely related groupings correlating with camphor, bornyl
acetate, and terpinen-4-ol, and the lone camphor-rich sample. Based on the
multivariate analyses, there does not seem to be any correlation with
geographical location or the presence/absence of juniper “berries”. That is,
samples from Idaho, Utah, Nevada, and California are found in the
camphor/bornyl acetate group; samples from Idaho, and Utah are found in the
camphor/terpinen-4-ol/trans-verbenol group; and samples from Idaho,
Utah, Arizona, and Nevado occupy the bornyl acetate/sabinene group. Likewise,
samples with and without “berries” were distributed in all three groups.
Table 2. Chemical composition (percent of total) of the foliar essential oils of Juniperus osteosperma.
RIcalc | RIdb | Compound | Sample number | ||||||
#1* | #2 | #3* | #4 | #5* | #6 | #7*(sU) | |||
922 | 923 | Tricyclene | 0.5 | 0.4 | 0.6 | 0.7 | 0.7 | 0.9 | 0.6 |
924 | 927 | 4-Methyl-3-heptanone | 0.1 | 0.2 | tr | tr | tr | tr | 0.1 |
925 | 925 | α-Thujene | 0.6 | 0.5 | 0.5 | 0.6 | 0.5 | 0.6 | 0.3 |
933 | 933 | α-Pinene | 4.8 | 3.9 | 5.6 | 6.6 | 5.7 | 4.7 | 9.6 |
949 | 950 | Camphene | 0.7 | 0.5 | 0.6 | 0.8 | 0.8 | 1.0 | 0.8 |
953 | 953 | Thuja-2,4(10)-diene | tr | 0.1 | 0.2 | 0.1 | 0.1 | 0.1 | tr |
972 | 972 | Sabinene | 12.3 | 7.1 | 7.0 | 10.4 | 6.2 | 4.3 | 7.5 |
977 | 978 | β-Pinene | 0.1 | 0.1 | 0.1 | 0.2 | 0.1 | 0.1 | 0.2 |
988 | 989 | Myrcene | 1.3 | 0.8 | 0.9 | 1.2 | 1.0 | 0.8 | 1.9 |
1007 | 1007 | α-Phellandrene | 0.1 | tr | 0.1 | tr | 0.1 | 0.1 | 0.1 |
1009 | 1009 | δ-3-Carene | - | - | - | - | - | - | 0.2 |
1017 | 1017 | α-Terpinene | 1.0 | 0.6 | 1.0 | 0.9 | 1.1 | 1.1 | 0.8 |
1025 | 1025 | p-Cymene | 1.8 | 4.9 | 3.6 | 3.3 | 2.0 | 2.9 | 1.2 |
1027 | 1026 | 2-Acetyl-3-methylfuran | tr | tr | tr | tr | tr | tr | - |
1029 | 1030 | Limonene | 4.2 | 2.6 | 3.3 | 3.4 | 3.8 | 5.2 | 5.2 |
1031 | 1031 | β-Phellandrene | 0.3 | 0.2 | 0.3 | 0.2 | 0.2 | 0.2 | 0.6 |
1036 | 1035 | Lavender lactone | - | 0.1 | tr | tr | tr | tr | - |
1045 | 1045 | (E)-β-Ocimene | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 |
1058 | 1057 | γ-Terpinene | 2.0 | 1.4 | 1.8 | 1.7 | 1.9 | 1.8 | 1.5 |
1070 | 1069 | cis-Sabinene hydrate | 1.3 | 1.1 | 0.7 | 1.1 | 0.8 | 0.5 | 0.7 |
1085 | 1086 | Terpinolene | 0.7 | 0.5 | 0.7 | 0.7 | 0.6 | 0.6 | 0.7 |
1086 | 1086 | trans-Linalool oxide (furanoid) | tr | 0.1 | 0.1 | 0.1 | 0.1 | tr | tr |
1090 | 1091 | p-Cymenene | 0.1 | 0.2 | 0.1 | 0.1 | 0.1 | 0.1 | tr |
1098 | 1098 | Perillene | 0.1 | tr | tr | tr | 0.1 | tr | tr |
1099 | 1101 | Linalool | 0.1 | 0.3 | 0.2 | 0.3 | 0.2 | 0.2 | 0.1 |
1101 | 1101 | trans-Sabinene hydrate | 1.3 | 1.0 | 0.6 | 1.0 | 0.6 | 0.4 | 0.5 |
1106 | 1104 | 2-Methylbutyl isovalerate | - | - | - | - | - | - | tr |
1115 | 1114 | 3-Methyl-3-butenyl isovalerate | - | - | - | - | - | - | tr |
1118 | 1117 | β-Thujone | 0.1 | 0.2 | 0.2 | 0.1 | 0.1 | 0.1 | tr |
1119 | 1118 | Dehydrosabina ketone | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | tr | - |
1122 | 1122 | trans-p-Mentha-2,8-dien-1-ol | 0.2 | 0.1 | 0.1 | 0.1 | 0.1 | 0.2 | 0.5 |
1125 | 1124 | cis-p-Menth-2-en-1-ol | 0.8 | 0.8 | 0.7 | 0.7 | 0.6 | 0.5 | - |
1127 | 1126 | α-Campholenal | 0.2 | 0.4 | 0.4 | 0.4 | 0.3 | 0.3 | 0.1 |
1137 | 1138 | trans-Limonene oxide | 0.6 | 0.3 | 0.5 | 0.2 | 0.3 | 0.3 | tr |
1140 | 1140 | trans-Sabinol | 0.4 | 0.6 | 0.8 | 0.6 | 0.4 | 0.5 | 0.1 |
1141 | 1141 | trans-Pinocarveol | 0.1 | 0.3 | 0.1 | 0.1 | 0.1 | - | - |
1142 | 1141 | cis-Verbenol | - | - | 0.1 | 0.3 | 0.1 | 0.2 | tr |
1143 | 1142 | trans-p-Menth-2-en-1-ol | 0.6 | 0.8 | 0.6 | 0.5 | 0.3 | 0.3 | 0.4 |
1146 | 1145 | trans-Verbenol | 0.4 | 10.2 | 9.3 | 3.3 | - | - | - |
1148 | 1149 | Camphor | 3.9 | 7.3 | 14.4 | 5.3 | 30.6 | 31.1 | 18.4 |
1156 | 1156 | Camphene hydrate | 1.4 | 0.7 | 0.8 | 1.1 | 1.2 | 1.0 | 1.3 |
1158 | 1157 | Sabina ketone | 1.1 | 1.0 | 1.4 | 0.9 | 0.7 | 0.6 | 0.1 |
1163 | 1164 | Pinocarvone | tr | 0.1 | 0.1 | 0.1 | tr | tr | tr |
1164 | 1165 | iso-Borneol | 0.1 | tr | tr | 0.1 | 0.1 | 0.1 | 0.1 |
1170 | 1168 | α-Phellandrene epoxide | 0.5 | 0.5 | 0.3 | 0.2 | 0.2 | 0.1 | tr |
1173 | 1173 | Borneol | 2.9 | 3.8 | 4.2 | 3.2 | 4.0 | 4.2 | 1.6 |
1182 | 1180 | Terpinen-4-ol | 12.1 | 15.3 | 12.4 | 11.9 | 10.2 | 10.2 | 7.9 |
1184 | 1184 | Thuj-3-en-10-al | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | tr |
1187 | 1186 | p-Cymen-8-ol | 0.6 | 1.0 | 1.0 | 0.6 | 0.5 | 0.7 | 0.2 |
1192 | 1194 | p-Mentha-1,5-dien-7-ol | 0.6 | 0.4 | 0.4 | 0.3 | 0.3 | 0.3 | tr |
1195 | 1195 | α-Terpineol | 0.9 | 0.9 | 0.8 | 0.8 | 0.7 | 0.7 | 0.6 |
1197 | 1195 | Myrtenol | tr | tr | 0.3 | 0.2 | 0.1 | 0.1 | tr |
1197 | 1196 | cis-Piperitol | 0.2 | 0.5 | 0.1 | 0.1 | 0.1 | tr | 0.2 |
1200 | 1201 | cis-Piperitenol | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.2 | tr |
1207 | 1208 | Verbenone | 0.3 | 2.0 | 2.0 | 1.4 | 0.6 | 0.9 | 0.2 |
1209 | 1208 | trans-Piperitol | 0.3 | 0.3 | 0.2 | 0.2 | 0.2 | 0.2 | 0.2 |
1219 | 1218 | trans-Carveol | 1.0 | 1.2 | 1.5 | 0.8 | 0.7 | 1.0 | 0.2 |
1227 | 1228 | Citronellol | - | - | - | 0.2 | - | - | 0.9 |
1232 | 1232 | cis-Carveol | 0.2 | 0.1 | 0.1 | 0.1 | 0.1 | 0.2 | tr |
1242 | 1242 | Cuminaldehyde | 0.2 | 0.4 | 0.4 | 0.3 | 0.2 | 0.2 | tr |
1244 | 1246 | Carvone | 0.6 | 0.6 | 0.7 | 0.5 | 0.5 | 0.7 | 0.1 |
1254 | 1254 | Piperitone | 0.2 | 0.2 | 0.1 | 0.1 | - | - | tr |
1257 | 1257 | Methyl citronellate | - | - | - | 0.1 | tr | tr | 0.1 |
1265 | 1265 | 3,5-Dimethoxytoluene | 0.1 | - | - | 0.1 | 0.1 | 0.2 | - |
1278 | 1277 | Phellandral | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | tr |
1284 | 1285 | Bornyl acetate | 27.0 | 11.7 | 9.3 | 23.5 | 12.0 | 10.0 | 23.1 |
1287 | 1287 | iso-Bornyl acetate | 0.7 | 0.4 | 0.4 | 0.6 | 0.4 | 0.4 | 0.5 |
1288 | 1287 | α-Terpinen-7-al | 0.1 | 0.2 | 0.2 | 0.1 | 0.1 | 0.1 | tr |
1291 | 1291 | p-Cymen-7-ol | 1.2 | 2.3 | 1.5 | 1.4 | 0.7 | 0.9 | 0.2 |
1297 | 1300 | Carvacrol | tr | 0.2 | 0.1 | 0.1 | tr | 0.1 | tr |
1299 | 1299 | Perilla alcohol | 0.1 | tr | tr | 0.1 | 0.1 | 0.1 | - |
1304 | 1309 | 4-Vinylguaiacol | 0.7 | 0.2 | 0.3 | 0.3 | 0.2 | 0.2 | 1.0 |
1315 | --- | Unidentified a | 0.1 | 0.2 | 0.3 | 0.2 | 0.8 | 1.8 | 0.9 |
1320 | 1320 | Methyl geranate | - | 0.2 | - | 0.1 | 0.3 | 0.1 | tr |
1328 | 1327 | p-Mentha-1,4-dien-7-ol | 0.9 | 0.9 | 1.2 | 0.8 | 0.7 | 0.9 | 0.1 |
1345 | 1346 | α-Terpinyl acetate | - | tr | 0.1 | - | 0.1 | 0.1 | 0.1 |
1356 | --- | p-Mentha-1,3-dien-7-ol (= Anthemol) | 0.2 | 0.2 | 0.3 | 0.3 | 0.2 | 0.2 | - |
1374 | 1374 | Bornyl propionate | tr | 0.1 | 0.1 | 0.1 | 0.1 | tr | tr |
1418 | 1417 | (E)-β-Caryophyllene | 0.1 | tr | tr | tr | tr | tr | 0.1 |
1447 | 1450 | trans-Muurola-3,5-diene | - | - | - | - | tr | - | - |
1454 | 1455 | α-Humulene | - | - | 0.1 | 0.1 | - | - | 0.1 |
1467 | --- | Methyl (2E,4E)-3,7-dimethyl-2,4,6-octatrienoate | - | - | - | - | 0.1 | tr | tr |
1470 | 1472 | trans-Cadina-1(6),4-diene | - | - | - | - | tr | - | - |
1490 | 1490 | γ-Amorphene | - | - | - | - | 0.1 | - | - |
1496 | 1497 | epi-Cubebol | - | - | - | - | 0.1 | - | - |
1498 | 1500 | α-Muurolene | - | - | - | - | - | tr | - |
1511 | 1512 | γ-Cadinene | tr | tr | tr | - | - | tr | - |
1514 | 1515 | Cubebol | - | 0.1 | tr | - | 0.1 | - | - |
1516 | 1518 | δ-Cadinene | 0.1 | 0.1 | 0.1 | - | 0.2 | 0.1 | tr |
1549 | 1549 | α-Elemol | 1.2 | 3.2 | 1.6 | 1.9 | 2.6 | 2.4 | 6.3 |
1577 | 1574 | Germacra-1(10),5-dien-4β-ol | 0.1 | tr | tr | - | - | 0.1 | tr |
1582 | 1587 | Caryophyllene oxide | 0.2 | 0.3 | 0.2 | 0.2 | 0.1 | 0.1 | 0.1 |
1603 | 1605 | β-Oplopenone | - | - | - | - | - | tr | - |
1610 | 1611 | Humulene epoxide II | 0.1 | 0.1 | 0.2 | 0.2 | tr | tr | tr |
1622 | 1624 | 10-epi-γ-Eudesmol | - | - | - | - | - | - | 0.1 |
1628 | 1628 | 1-epi-Cubenol | - | 0.1 | 0.1 | - | 0.2 | tr | tr |
1632 | 1632 | γ-Eudesmol | 0.1 | 0.3 | 0.1 | 0.2 | 0.1 | 0.2 | 0.5 |
1642 | 1640 | τ-Cadinol | tr | 0.1 | tr | - | - | 0.1 | tr |
1643 | 1643 | Cubenol | - | - | - | - | tr | - | - |
1644 | 1643 | τ-Muurolol | 0.1 | 0.1 | tr | - | tr | 0.1 | tr |
1655 | 1655 | α-Eudesmol | 0.5 | 0.8 | 0.3 | 0.4 | 0.3 | 0.6 | 1.0 |
1662 | 1660 | ar-Turmerone | 0.2 | 0.3 | 0.1 | 0.2 | tr | 0.1 | tr |
1668 | 1668 | α-Turmerone | 0.2 | 0.3 | 0.1 | 0.2 | 0.1 | 0.1 | tr |
1700 | 1699 | β-Turmerone (= Curlone B) | 0.1 | 0.2 | 0.1 | 0.1 | tr | tr | tr |
1732 | 1735 | Oplopanone | 0.2 | 0.2 | 0.1 | tr | - | 0.3 | tr |
1740 | 1740 | 8α,11-Elemodiol | - | - | - | 0.2 | - | - | - |
1991 | 1994 | Manoyl oxide | 0.1 | - | tr | tr | - | tr | tr |
Compound Classes |
|
|
|
|
|
|
| ||
Monoterpene hydrocarbons | 30.5 | 23.7 | 26.7 | 31.0 | 24.9 | 24.7 | 31.3 | ||
Oxygenated monoterpenoids | 63.9 | 69.1 | 69.2 | 64.4 | 70.1 | 68.9 | 58.5 | ||
Sesquiterpene hydrocarbons | 0.1 | 0.1 | 0.1 | 0.1 | 0.2 | 0.1 | 0.2 | ||
Oxygenated sesquiterpenoids | 2.9 | 6.0 | 2.8 | 3.6 | 3.6 | 4.1 | 7.9 | ||
Diterpenoids | 0.1 | 0.0 | tr | tr | 0.0 | tr | tr | ||
Benzenoid aromatics | 0.8 | 0.2 | 0.3 | 0.4 | 0.2 | 0.4 | 1.0 | ||
Others | 0.1 | 0.2 | tr | tr | tr | tr | 0.1 | ||
Total identified | 98.5 | 99.4 | 99.0 | 99.4 | 99.1 | 98.0 | 98.9 | ||
RIcalc = Retention index determined using a homologous series of n-alkanes on a ZB-5ms column. RIdb = Reference retention index from the databases. Sample numbers with an asterisk (*) had abundant female cones (berries). Sample #7(sU) was collected in southern Utah. tr = trace (< 0.05%). - = not detected. a MS(EI): 164(20%), 140(100%), 91(44%), 77(10%), 65(7%), 51(7%), 43(8%) (the MS looks like either thymyl methyl ether or carvacryl methyl ether, but the RI is too high).
Figure 3. Hierarchical cluster analysis (HCA) of Juniperus osteosperma foliar essential oils. Samples #1-#7 are from this work, samples with an asterisk had abundant female cones (berries); samples NV (Nevada), nUT (northern Utah), sCA (southern California), AZ(12) (Arizona, 2012) are from Adams, 2012 [17]; samples wNV (western Nevada) are from Adams, 2013 [18]; sample AZ(14) (Arizona, 2014) is from Adams et al. 2014 [19]; samples Utb (Utah, browsed) and Utnb (Utah, not browsed) are from Adams et al., 2016 [20]; sample Utah is from Wilson et al. 2019 [21].
Figure 4. Principal component analysis (PCA) of Juniperus osteosperma foliar essential oils. Samples #1-#7 are from this work, samples with an asterisk had abundant female cones (berries); samples NV (Nevada), nUT (northern Utah), sCA (southern California), AZ(12) (Arizona, 2012) are from Adams, 2012 [17]; samples wNV (western Nevada) are from Adams, 2013 [18]; sample AZ(14) (Arizona, 2014) is from Adams et al. 2014 [19]; samples Utb (Utah, browsed) and Utnb (Utah, not browsed) are from Adams et al., 2016 [20]; sample Utah is from Wilson et al. 2019 [21].
Several of the major components have demonstrated relevant biological activities. Bornyl acetate [23–25], sabinene [26], camphor [27], borneol [28], terpinen-4-ol [29–31], (+)-α-pinene [32,33], (+)-limonene [34,35], and p-cymene [36] have shown anti-inflammatory activities. Camphor [27], (+)-limonene [37], and p-cymene [36, 38] have analgesic properties. Camphor has demonstrated antitussive effects in rodent models [39, 40] and has been used as a home treatment for colds and as a topical analgesic [41]. (+)-α-Pinene, sabinene, (+)-limonene, (–)-borneol, and bornyl acetate have shown antimicrobial activities against several pulmonary and dermal pathogens [26,42]. The anti-inflammatory, analgesic, antitussive, and antimicrobial activities of the major components of J. osteosperma essential oil are consistent with the Native American traditional uses of the plant to treat coughs and colds, rheumatism, and skin infections.
3.2. Enantiomeric distribution
The J. osteosperma essential oils in this study were subjected to chiral GC-MS in order to evaluate the enantiomeric ratios of chiral monoterpenoid components (Table 3). The dominant enantiomers were (+)-α-pinene (³ 99.5%), (+)-camphene (³ 93.0%), (+)-sabinene (100%), (+)-β-pinene (100%), (+)-limonene (³ 97.7%), (+)-cis-sabinene hydrate (³ 93.7%), (+)-trans-sabinene hydrate (³ 95.8%), (+)-camphor (³ 98.3%), (+)-α-terpineol (100%), and (+)- verbenol (100%). The enantiomeric distribution in terpinen-4-ol was less extreme with (+)-terpinen-4-ol (64.0-70.3%) predominating over (–)-terpinen-4-ol. Unfortunately, it was not possible to distinguish the enantiomers for α-thujene (although only one peak was observed in each essential oil sample, the RI values are too similar for the two reference enantiomers), β-phellandrene (although only one peak was observed in the essential oils, the observed RI value was in between the RI values for the reference enantiomers), borneol (although only one peak was observed in the essential oils, the observed RI value was in between the RI values for the reference enantiomers), or bornyl acetate (the concentrations of bornyl acetate were very large so separation of enantiomers was not likely possible).
Although previous investigations of J. osteosperma essential oils did not include enantioselective GC-MS, there have been several reports on enantiomeric distributions in other Juniperus species (Table 4) [43–46]. There does not seem to be enantiomeric consistency in α-pinene, camphene, sabinene, terpinen-4-ol or α-terpineol. Interestingly, the major enantiomer was (+)-limonene in all Juniperus essential oils. In fact, in members of the Cupressaceae, (+)-limonene seems to predominate over (–)-limonene, including essential oils of Chamaecyparis, Cupressus, and Thuja, as well as Juniperus [47–53], while (–)-limonene predominates in members of the Pinaceae (Abies, Picea, Pinus) [48, 51–54].
Table 3. Enantiomeric distribution of terpenoid constituents in Juniperus osteosperma essential oils.
Compound | RIdb | RIcalc | Sample number | ||||||
#1 | #2 | #3 | #4 | #5 | #6 | #7 | |||
(–)-α-Pinene | 976 | 976 | 0.5 | 0.2 | 0.2 | 0.2 | 0.3 | 0.3 | 0.4 |
(+)-α-Pinene | 982 | 978 | 99.5 | 99.8 | 99.8 | 99.8 | 99.7 | 99.7 | 99.6 |
(–)-Camphene | 998 | 1001 | 7.1 | 6.5 | 6.9 | 6.6 | 6.8 | 6.3 | 7.0 |
(+)-Camphene | 1005 | 1005 | 93.0 | 93.5 | 93.1 | 93.3 | 93.2 | 93.7 | 93.0 |
(+)-Sabinene | 1021 | 1019 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 |
(–)-Sabinene | 1030 | - | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
(+)-β-Pinene | 1027 | 1027 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 |
(–)-β-Pinene | 1031 | - | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
(–)-Limonene | 1073 | 1076 | 1.5 | 2.3 | 2.1 | 1.6 | 2.1 | 1.4 | 2.2 |
(+)-Limonene | 1081 | 1080 | 98.5 | 97.7 | 97.9 | 98.4 | 97.9 | 98.6 | 97.8 |
(+)-cis-Sabinene hydrate | 1199 | 1201 | 95.4 | 94.8 | 93.7 | 96.1 | 96.1 | 94.8 | 94.4 |
(–)-cis-Sabinene hydrate | 1202 | 1204 | 4.6 | 5.2 | 6.3 | 3.9 | 3.9 | 5.2 | 5.6 |
(+)-trans-Sabinene hydrate | 1231 | 1231 | 97.6 | 97.4 | 97.8 | 98.2 | 97.8 | 97.9 | 95.8 |
(–)-trans-Sabinene hydrate | 1235 | 1236 | 2.4 | 2.6 | 2.2 | 1.8 | 2.2 | 2.1 | 4.2 |
(–)-Camphor | 1253 | 1254 | 1.7 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
(+)-Camphor | 1259 | 1258 | 98.3 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 |
(+)-Terpinen-4-ol | 1297 | 1296 | 70.3 | 64.0 | 69.3 | 66.0 | 68.1 | 66.5 | 67.5 |
(–)-Terpinen-4-ol | 1300 | 1300 | 29.7 | 36.0 | 30.7 | 34.0 | 31.9 | 33.5 | 32.5 |
(–)-α-Terpineol | 1347 | - | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
(+)-α-Terpineol | 1356 | 1359 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 |
(–)-Verbenone | 1368 | - | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 | 0.0 |
(+)-Verbenone | 1380 | 1378 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 | 100.0 |
RIdb = Retention index from our in-house database. RIcalc = Calculated retention index based on a homologous series of n-alkanes on
a Restek B-Dex 325 capillary column. - = compound not detected.
Table 4. Enantiomeric distribution of chiral monoterpenoids in Juniperus species.
Compound | J. osteosperma (this work) a | J. brevifolia [43] a | J. communis [44] | J. horizontalis [46] | J. oxycedrus [45] | J. phoenicea [45] | J. scopulorum [46] |
(–)-α-Pinene | 0.3 | 96.4 | 43.0 | 18.6 | 11.4 | 10.9 | 8.3 |
(+)-α-Pinene | 99.7 | 3.6 | 57.0 | 81.4 | 88.6 | 89.1 | 91.7 |
(–)-Camphene | 6.7 | 61.7 | 37.3 | 20.9 | 19.9 | 48.0 | |
(+)-Camphene | 93.3 | 38.3 | 62.7 | 79.1 | 80.1 | 52.0 | |
(+)-Sabinene | 100.0 | 100.0 | 40.1 b | 39.6 | 100.0 | ||
(–)-Sabinene | 0.0 | 0.0 | 50.9 b | 60.4 | 0.0 | ||
(+)-β-Pinene | 100.0 | 45.7 | 100.0 | 56.4 | 51.7 | 100.0 | |
(–)-β-Pinene | 0.0 | 54.3 | 0.0 | 43.6 | 48.3 | 0.0 | |
(–)-Limonene | 2.0 | 0.2 | 22.1 | 17.2 | 4.9 | 18.4 | 9.9 |
(+)-Limonene | 98.0 | 99.8 | 77.9 | 82.8 | 95.1 | 81.6 | 90.1 |
(+)-cis-Sabinene hydrate | 95.0 | 97.7 | 95.9 | ||||
(–)-cis-Sabinene hydrate | 5.0 | 2.3 | 4.1 | ||||
(+)-trans-Sabinene hydrate | 97.5 | 97.8 | |||||
(–)-trans-Sabinene hydrate | 2.5 | 2.2 | |||||
(–)-Camphor | 0.0 | ||||||
(+)-Camphor | 100.0 | ||||||
(+)-Terpinen-4-ol | 66.9 | 32.8 | 66.6 | 22.5 | 31.5 | 53.0 | |
(–)-Terpinen-4-ol | 33.1 | 67.2 | 33.4 | 77.5 | 68.5 | 47.0 | |
(–)-α-Terpineol | 0.0 | 49.4 | 46.0 | ||||
(+)-α-Terpineol | 100.0 | 50.6 | 54.0 |
a Averages. b The sum of enantiomers does not add up to 100%.
4. Conclusions
This work presents the foliar essential oil compositions of six individual J. osteosperma from southern Idaho. In comparison with essential oils of J. osteosperma from California, Nevada, and Utah, the dissimilarity is slight, with bornyl acetate, sabinene, terpinen-4-ol and camphor dominating the essential oils. The reported biological activities of the major essential oil components are consistent with the Native American traditional uses of the plant. Additionally, the enantiomeric distribution of chiral monoterpenoids has been assessed in J. osteosperma. The (+)-enantiomers dominated the distributions for α-pinene, β-pinene, camphene, limonene, cis- and trans-sabinene hydrate, camphor, α-terpineol, and verbenone, and add to our understanding of the volatile components of Juniperus species. Although this work expands our understanding of J. osteosperma essential oils to include southern Idaho, additional information on essential oils from Wyoming and Arizona would fill in some gaps. Additional work is needed on other members of the Cupressaceae to further delineate compositional trends in the family.
Authors’ contributions
Conceptualization, W.N.S.; Methodology, P.S. and W.N.S.; Software, P.S.; Validation, W.N.S., Formal Analysis, A.P., P.S., and W.N.S.; Investigation, K.S., A.P., P.S., and W.N.S.; Resources, P.S. and W.N.S.; Data Curation, W.N.S.; Writing – Original Draft Preparation, W.N.S.; Writing – Review & Editing, K.S., A.P., P.S., and W.N.S.; Project Administration, W.N.S.
Acknowledgements
This work was carried out as part of the activities of the Aromatic Plant Research Center (APRC, https://aromaticplant.org/).
Funding
This research received no specific grant from any funding agency.
Availability of data and materials
All data will be made available on request according to the journal policy.
Conflicts of interest
The authors declare no conflict of interest.
References
1. National
Park Service, N.P.S. Cupressaceae Juniperus osteosperma. Available online:
https://www.nps.gov/arch/learn/nature/cupressaceae_juniperus_osteosperma.htm
(accessed on Nov 18, 2023).
2. Zlatnik, E. Juniperus osteosperma. In
Fire Effects Information System [Online]; U.S. Department of Agriculture,
Forest Service, Rocky Mountain Research Station, Fire Sciences Laboratory,
1999. Available online:
https://www.fs.usda.gov/database/feis/plants/tree/junost/all.html (accessed on
Nov 18, 2023).
3. Zillig, M.W.; Fogarty, F.A.; Fleishman, E.
Bird associations with floristics and physiognomy differ across five
biogeographic subregions of the Great Basin, USA. Ornithol. Appl. 2023, 125,
duac040. https://doi.org/10.1093/ornithapp/duac040.
4. Riskas, H.L. Climate Effects on Canopy
Arthropod Communities of Pinyon Pine and Juniper Trees, M.S. thesis, Northern
Arizona University, 2021.
5. Stapleton, T.E.; Weinstein, S.B.; Greenhalgh,
R.; Dearing, M.D. Successes and limitations of quantitative diet metabarcoding
in a small, herbivorous mammal. Mol. Ecol. Resour. 2022, 22, 2573–2586. https://doi.org/10.1111/1755-0998.13643.
6. Schramm, K.; Skopec, M.; Dearing, D.
Metabolomic evidence of independent biotransformation pathways for terpenes in
two specialist mammalian herbivores (genus Neotoma). Integr. Zool. 2024,
19, 143–155. https://doi.org/10.1111/1749-4877.12734.
7. Tanner, D.A. Novel Fire and Herbivory Regime
Impacts on Forest Regeneration and Plant Community Assembly, Ph.D.
dissertation, Brigham Young University, 2023.
8. Moerman, D.E. Native American Ethnobotany;
Timber Press, Inc.: Portland, OR, 1998; ISBN 978-0-88192-453-4.
9. Van Buren, R.; Cooper, J.G.; Shultz, L.M.;
Harper, K.T. Woody Plants of Utah; Utah State University Press: Logan, Utah,
USA, 2011; ISBN 978-0-87421-824-4.
10. Southern Utah University. Available online:
https://www.intermountainbiota.org/portal/collections/list.php (accessed on May
4, 2023).
11. Satyal, P.; Dosoky, N.S.; Poudel, A.; Swor, K.;
Setzer, W.N. Chemical composition of the aerial parts essential oil of Chrysothamnus
viscidiflorus from southwestern Idaho. J. Essent. Oil Plant Compos. 2023, 1,
115–121. https://doi.org/10.58985/jeopc.2023.v01i02.16.
12. van den Dool, H.; Kratz, P.D. A generalization
of the retention index system including linear temperature programmed
gas-liquid partition chromatography. J. Chromatogr. A 1963, 11, 463–471. https://doi.org/10.1016/S0021-9673(01)80947-X.
13. Adams, R.P. Identification of Essential Oil
Components by Gas Chromatography/Mass Spectrometry, 4th ed.; Allured
Publishing: Carol Stream, IL, USA, 2007; ISBN 978-1-932633-21-4.
14. Mondello, L. FFNSC 3; Shimadzu Scientific
Instruments: Columbia, Maryland, USA, 2016.
15. NIST20; National Institute of Standards and
Technology: Gaithersburg, Maryland, USA, 2020.
16. Satyal, P. Development of GC-MS Database of
Essential Oil Components by the Analysis of Natural Essential Oils and
Synthetic Compounds and Discovery of Biologically Active Novel Chemotypes in
Essential Oils, Ph.D. dissertation, University of Alabama in Huntsville,
Huntsville, AL, USA, 2015.
17. Adams, R.P. Geographic variation in the leaf
essential oils of Juniperus osteosperma (Cupressaceae) II. Phytologia
2012, 94, 118–132.
18. Adams, R.P. Hybridization between Juniperus
grandis, J. occidentalis and J. osteosperma in northwest
Nevada I: Terpenes, Leviathan Mine, Nevada. Phytologia. 2013, 95, 58–69.
19. Adams, R.P.; Skopec, M.M.; Kohl, K.D.; Dearing,
M.D. Comparison of volatile leaf terpenoids from Juniperus monosperma
and J. osteosperma leaves: Intact, ground and exposed to ambient
temperature. Phytologia 2014, 96, 207–217.
20. Adams, R.P.; Skopec, M.M.; Muir, J.P.
Comparison of leaf terpenoids and tannins in Juniperus osteosperma from
woodrat (Neotoma stephensi) browsed and not-browsed trees. Phytologia
2016, 98, 17–25.
21. Wilson, T.M.; Poulson, A.; Packer, C.;
Marshall, J.; Carlson, R.; Buch, R.M. Essential oils of whole tree, trunk,
limbs and leaves of Juniperus osteosperma from Utah. Phytologia 2019,
101, 188–193.
22. Wilson, T.M.; Ziebarth, E.A.; Poulson, A.;
Packer, C.; Carlson, R.E. An investigation of cones (berries) of Juniperus
spp. of Utahn origin as a flavoring source for gin. J. Agric. Food Sci.
Biotechnol. 2023, 1, 211–218. https://doi.org/10.58985/jafsb.2023.v01i03.25.
23. Yang, H.; Zhao, R.; Chen, H.; Jia, P.; Bao, L.;
Tang, H. Bornyl acetate has an anti-inflammatory effect in human chondrocytes
via induction of IL-11. IUBMB Life 2014, 66, 854–859. https://doi.org/10.1002/iub.1338.
24. Chen, N.; Sun, G.; Yuan, X.; Hou, J.; Wu, Q.;
Soromou, L.W.; Feng, H. Inhibition of lung inflammatory responses by bornyl
acetate is correlated with regulation of myeloperoxidase activity. J. Surg.
Res. 2014, 186, 436–445. https://doi.org/10.1016/j.jss.2013.09.003.
25. Zhao, Z.; Sun, Y.; Ruan, X. Bornyl acetate: A
promising agent in phytomedicine for inflammation and immune modulation.
Phytomed. 2023, 114, 154781. https://doi.org/10.1016/j.phymed.2023.154781.
26. Valente, J.; Zuzarte, M.; Gonçalves, M.J.;
Lopes, M.C.; Cavaleiro, C.; Salgueiro, L.; Cruz, M.T. Antifungal, antioxidant
and anti-inflammatory activities of Oenanthe crocata L. essential oil.
Food Chem. Toxicol. 2013, 62, 349–354. https://doi.org/10.1016/j.fct.2013.08.083.
27. dos Santos, E.; Leitão, M.M.; Aguero Ito, C.N.;
Silva-Filho, S.E.; Arena, A.C.; Silva-Comar, F.M. de S.; Nakamura Cuman, R.K.;
Oliveira, R.J.; Nazari Formagio, A.S.; Leite Kassuya, C.A. Analgesic and
anti-inflammatory articular effects of essential oil and camphor isolated from Ocimum
kilimandscharicum Gürke leaves. J. Ethnopharmacol. 2021, 269, 113697. https://doi.org/10.1016/j.jep.2020.113697.
28. Ehrnhöfer-Ressler, M.M.; Fricke, K.; Pignitter,
M.; Walker, J.M.; Walker, J.; Rychlik, M.; Somoza, V. Identification of
1,8-cineole, borneol, camphor, and thujone as anti-inflammatory compounds in a Salvia
officinalis L. infusion using human gingival fibroblasts. J. Agric. Food
Chem. 2013, 61, 3451–3459. https://doi.org/10.1021/jf305472t.
29. Hart, P.H.; Brand, C.; Carson, C.F.; Riley, T.
V.; Prager, R.H.; Finlay-Jones, J.J. Terpinen-4-ol, the main component of the
essential oil of Melaleuca alternifolia (tea tree oil), suppresses
inflammatory mediator production by activated human monocytes. Inflamm. Res.
2000, 49, 619–626. https://doi.org/10.1007/s000110050639.
30. Ning, J.; Xu, L.; Zhao, Q.; Zhang, Y.; Shen, C.
The protective effects of terpinen-4-ol on LPS-induced acute lung injury via
activating PPAR-γ. Inflammation 2018, 41, 2012–2017. https://doi.org/10.1007/s10753-018-0844-1.
31. Aslam, S.; Younis, W.; Malik, M.N.H.; Jahan,
S.; Alamgeer; Uttra, A.M.; Munir, M.U.; Roman, M. Pharmacological evaluation of
anti-arthritic potential of terpinen-4-ol using in vitro and in vivo assays.
Inflammopharmacol. 2022, 30, 945–959. https://doi.org/10.1007/s10787-022-00960-w.
32. Rufino, A.T.; Ribeiro, M.; Judas, F.;
Salgueiro, L.; Lopes, M.C.; Cavaleiro, C.; Mendes, A.F. Anti-inflammatory and
chondroprotective activity of (+)-α-pinene: Structural and enantiomeric
selectivity. J. Nat. Prod. 2014, 77, 264–269. https://doi.org/10.1021/np400828x.
33. Allenspach, M.; Steuer, C. α-Pinene: A
never-ending story. Phytochem. 2021, 190, 112857. https://doi.org/10.1016/j.phytochem.2021.112857.
34. D’Alessio, P.A.; Ostan, R.; Bisson, J.F.;
Schulzke, J.D.; Ursini, M. V.; Béné, M.C. Oral administration of d-limonene
controls inflammation in rat colitis and displays anti-inflammatory properties
as diet supplementation in humans. Life Sci. 2013, 92, 1151–1156. https://doi.org/10.1016/j.lfs.2013.04.013.
35. Yu, L.; Yan, J.; Sun, Z. d-Limonene exhibits
anti-inflammatory and antioxidant properties in an ulcerative colitis rat model
via regulation of iNOS, COX-2, PGE2 and ERK signaling pathways. Mol. Med. Rep.
2017, 15, 2339–2346. https://doi.org/10.3892/mmr.2017.6241.
36. Bonjardim, L.R.; Cunha, E.S.; Guimarães, A.G.;
Santana, M.F.; Oliveira, M.G.B.; Serafini, M.R.; Araújo, A.A.S.; Antoniolli,
Â.R.; Cavalcanti, S.C.H.; Santos, M.R.V.; et al. Evaluation of the anti-inflammatory
and antinociceptive properties of p-cymene in mice. Zeitschrift fur
Naturforsch.- Sect. C J. Biosci. 2012, 67, 15–21. https://doi.org/10.1515/znc-2012-1-203.
37. do Amaral, J.F.; Silva, M.I.G.; Neto, M.R. de
A.; Neto, P.F.T.; Moura, B.A.; de Melo, C.T.V.; de Araújo, F.L.O.; de Sousa,
D.P.; de Vasconcelos, P.F.; de Vasconcelos, S.M.M.; et al. Antinociceptive
effect of the monoterpene R-(+)-limonene in mice. Biol. Pharm. Bull.
2007, 30, 1217–1220. https://doi.org/10.1248/bpb.30.1217.
38. Balahbib, A.; El Omari, N.; Hachlafi, N. EL;
Lakhdar, F.; El Menyiy, N.; Salhi, N.; Mrabti, H.N.; Bakrim, S.; Zengin, G.;
Bouyahya, A. Health beneficial and pharmacological properties of p-cymene.
Food Chem. Toxicol. 2021, 153, 112259. https://doi.org/10.1016/j.fct.2021.112259.
39. Laude, E.A.; Morice, A.H.; Grattan, T.J. The
antitussive effects of menthol, camphor and cineole in conscious Guinea-pigs.
Pulm. Pharmacol. 1994, 7, 179–184. https://doi.org/10.1006/pulp.1994.1021.
40. Gavliakova, S.; Dolak, T.; Licha, H.; Krizova,
S.; Plevkova, J. Cineole, thymol and camphor nasal challenges and their effect
on nasal symptoms and cough in an animal model. Acta Medica Martiniana 2013, 13,
5–13. https://doi.org/10.2478/acm-2013-0012.
41. Zuccarini, P.; Soldani, G. Camphor: Benefits
and risks of a widely used natural product. Acta Biol. Szeged. 2009, 53, 77–82.
https://doi.org/10.4314/jasem.v13i2.55317.
42. Poudel, A.; Dosoky, N.S.; Satyal, P.; Swor, K.;
Setzer, W.N. Essential oil composition of Grindelia squarrosa from
southern Idaho. Molecules 2023, 28, 3854. https://doi.org/10.3390/molecules28093854.
43. da Silva, J.A.; Pedro, L.G.; Santos, P.A.G.;
Figueiredo, A.C.; Barroso, J.G.; Tenreiro, R.P.; Ribeiro, C.A.; Deans, S.G.;
Looman, A.; Scheffer, J.J.C. Essential oils from seven populations of Juniperus
brevifolia (Seub.) Antoine, an endemic species of the Azores. Flavour
Fragr. J. 2000, 15, 31–39. https://doi.org/10.1002/(SICI)1099-1026(200001/02)15:1<31::AID-FFJ862>3.0.CO;2-C.
44. Dahmane, D.; Dob, T.; Chelghoum, C. Essential
oil composition and enantiomeric distribution of some monoterpenoid components
of Juniperus communis L. from Algeria. J. Essent. Oil Res. 2016, 28,
348–356. https://doi.org/10.1080/10412905.2015.1133458.
45. Dahmane, D.; Dahmane, F.A.; Dob, T.; Chelghoum,
C. Qualitative, quantitative analysis and chiral characterization of the
essential oils of Juniperus phoenicea L. and Juniperus oxycedrus
L. Nat. Prod. Sci. 2020, 26, 97–107. https://doi.org/10.20307/nps.2020.26.1.97.
46. Swor, K.; Poudel, A.; Satyal, P.; Setzer, W.N.
Enantiomeric distribution of terpenoids in Juniperus essential oils:
Composition of Juniperus horizontalis and Juniperus scopulorum
leaf essential oils from southwestern Idaho. Nat. Prod. Commun. 2022, 17, 1934578X221091006.
https://doi.org/10.1177/1934578X221091006.
47. Ochocka, J.R.; Asztemborska, M.; Zook, D.R.;
Sybilska, D.; Perez, G.; Ossicini, L. Enantiomers of monoterpenic hydrocarbons
in essential oils from Juniperus communis. Phytochem. 1997, 44, 869–873.
https://doi.org/10.1016/s0031-9422(96)00587-0.
48. Wibe, A.; Borg-Karlson, A.-K.; Persson, M.;
Norin, T.; Mustaparta, H. Enantiomeric composition of monoterpene hydrocarbons
in some conifers and receptor neuron discrimination of α-pinene and limonene
enantiomers in the pine weevil, Hylobius abietis. J. Chem. Ecol. 1998,
24, 273–287. https://doi.org/10.1023/A:1022580308414.
49. Kimball, B.A.; Russell, J.H.; Griffin, D.L.;
Johnston, J.J. Response factor considerations for the quantitative analysis of
western redcedar (Thuja plicata) foliar monoterpenes. J. Chromatogr.
Sci. 2005, 43, 253–258. https://doi.org/10.1093/chromsci/43.5.253.
50. Leontiev, V.; Kavalenka, N.; Supichenka, H.;
Shutava, H. [Enantiomers of monoterpene compounds in the Thuja essential
oils] in Russian. Dokl. Natl. Acad.
Sci. Belarus 2014, 58, 74–76.
51. Pavela, R.; Maggi, F.; Mazzara, E.; Torresi,
J.; Cianfaglione, K.; Benelli, G.; Canale, A. Prolonged sublethal effects of
essential oils from non-wood parts of nine conifers on key insect pests and
vectors. Ind. Crops Prod. 2021, 168, 113590. https://doi.org/10.1016/j.indcrop.2021.113590.
52. Wang, Y.; Luo, W.; Rosenstiel, T.N.; Pankow,
J.F. Measurement of enantiomer percentages for five monoterpenes from six
conifer species by cartridge-tube-based passive sampling adsorption-thermal
desorption (ps-ATD). Atmos. Meas. Tech. 2022, 15, 4651–4661. https://doi.org/10.5194/amt-15-4651-2022.
53. Swor, K.; Satyal, P.; Poudel, A.; Setzer, W.N.
Gymnosperms of Idaho: Chemical compositions and enantiomeric distributions of
essential oils of Abies lasiocarpa, Picea engelmannii, Pinus
contorta, Pseudotsuga menziesii, and Thuja plicata. Molecules.
2023, 28, 2477. https://doi.org/10.3390/molecules28062477.
54. Swor, K.; Poudel, A.; Rokaya, A.; Satyal, P.; Setzer, W.N. Foliar essential oil of white fir (Abies concolor (Gordon) Lindl. ex Hildebr.): Chemical composition and enantiomeric distribution. Nat. Prod. Commun. 2022, 17, 1934578x221086901. https://doi.org/10.1177/1934578X221086901.
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
Abstract
Juniperus
osteosperma (Utah juniper) is an abundant tree
within its range in the Great Basin. It is an important source of food for
birds and mule deer and has served as traditional medicine for Native
Americans. The purpose of this work is to examine essential oils of J. osteosperma
growing in the northern limits of its range and to evaluate the enantiomeric
distribution of chiral monoterpenoids. Monoterpene hydrocarbons (23.7-31.3%)
and oxygenated monoterpenoids (58.5-70.1%) made up the bulk of the essential
oil compositions. The main constituents in J. osteosperma essential oils
were bornyl acetate (9.3-27.0%), camphor (3.9-31.1%), terpinen-4-ol
(7.9-15.3%), sabinene (4.3-12.3%), α-pinene (3.9-9.6%), limonene (2.6-5.2%),
borneol (1.6-4.2%), trans-verbenol (up to 10.2%), p-cymene
(1.2-4.9%), α-elemol
(1.2-6.3%), and γ-terpinene
(1.4-2.0%). Chiral GC-MS revealed (+)-α-pinene (³
99.5%), (+)-camphene (³
93.0%), (+)-sabinene (100%), (+)-β-pinene (100%), (+)-limonene (³ 97.7%), (+)-cis-sabinene
hydrate (³ 93.7%), (+)-trans-sabinene
hydrate (³ 95.8%),
(+)-camphor (³ 98.3%),
(+)-α-terpineol (100%), and (+)- verbenol (100%) to be the predominant
enantiomers. The essential oil compositions of J. osteosperma from
southern Idaho are similar to those from California, Nevada, and Utah, with bornyl
acetate, sabinene, terpinen-4-ol and camphor dominating the essential oils. The
reported biological activities of the major components are consistent with the
Native American traditional uses of the plant.
Abstract Keywords
Utah
juniper, Cupressaceae, chiral, enantiomer, multivariate analysis, gas
chromatography
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
Editor-in-Chief
This work is licensed under the
Creative Commons Attribution 4.0
License.(CC BY-NC 4.0).


