Research Article
Lantomalala Elsa Razafindrabenja
Lantomalala Elsa Razafindrabenja
Laboratoire de Chimie et de Biotechnologie des Produits Naturels (ChemBioPro), Université de La Réunion, 15 Avenue René Cassin, CS 92 003, 97 744 Saint-Denis Cedex 9, La Réunion, France.
E-mail: lantomalala.razafindrabenja@univ-reunion.fr
Jocelyne Rasolondramanitra
Jocelyne Rasolondramanitra
Laboratoire de
Chimie, École Supérieure Polytechnique d’Antananarivo, Université
d’Antananarivo, B.P. 1500, 101 Antananarivo, Madagascar.
E-mail:
j.rasolondramanitra@blueline.mg
André Razafimandefitra
André Razafimandefitra
Laboratoire de Chimie, École Supérieure Polytechnique d’Antananarivo, Université d’Antananarivo, B.P. 1500, 101 Antananarivo, Madagascar.
E-mail: mandefitra@yahoo.fr
Isabelle Grondin
Isabelle Grondin
Laboratoire de Chimie et de Biotechnologie des Produits Naturels (ChemBioPro), Université de La Réunion, 15 Avenue René Cassin, CS 92 003, 97 744 Saint-Denis Cedex 9, La Réunion, France.
E-mail: isabelle.grondin@univ-reunion.fr
Anne Gauvin-Bialecki
Anne Gauvin-Bialecki
Corresponding
Author
Laboratoire de Chimie et de Biotechnologie des Produits Naturels (ChemBioPro), Université de La Réunion, 15 Avenue René Cassin, CS 92 003, 97 744 Saint-Denis Cedex 9, La Réunion, France.
E-mail: anne.bialecki@univ-reunion.fr, Tel.: +262(0) 262 93 81 97
Abstract
The present study was carried out to compare
the chemical composition of the leaf essential
oils (EOs) from six Psiadia species
endemic to Madagascar, namely P.
altissima, P. stenophylla, P. hispida,
P. leucophylla, P. lucida and P. salviifolia.
Three of these species (P. altissima,
P. lucida and P. salviifolia) are
traditionally used for treating diarrhea, stomach ache and skin diseases. They are
also known for their antiseptic,
expectorant, hemostatic, carminative and anti-inflammatory properties. The EOs of the six species were obtained by hydrodistillation and
analysed by capillary GC-FID and GC-MS. Their oil yields ranged from 0.04% to 0.80%. 149 components were identified,
accounting for 84.0 to 97.9% of the
total composition. The EOs from the four species P. altissima, P. stenophylla,
P. hispida and P. salviifolia showed
similar chemical composition which was dominated by monoterpene (63.1-78.4%)
and sesquiterpene (5.7-25.7%) hydrocarbons. The major compounds identified in these oils were β-pinene (17.2-46.5%), limonene (10.4-28.5%) and (Z)-β-ocimene
(5.4-7.3%). The two other
species exhibited qualitative and quantitative differences in the chemical
composition of their EOs. The essential
oil (EO) from P. lucida
was found to be rich in sesquiterpene hydrocarbons (46.6%) and oxygenated sesquiterpenes (32.0%). α-cadinol (11.1%), α-muurolene (10.6%) and δ-cadinene (7.5%) were the main components. The P. leucophylla EO was characterized by a prominent content of sesquiterpene hydrocarbons (66.4%). The main compounds
included β-caryophyllene (10.6%), δ-cadinene (8.9%) and (E)-muurola-4(14),5-diene (7.2%).
This was the first report on the chemical composition of the EOs from P. stenophylla, P. hispida and P. leucophylla.
Abstract Keywords
Psiadia, essential oils, chemical composition, Madagascar, GC-MS, GC-FID.
1. Introduction
The genus Psiadia belonging to the Asteraceae (Compositae) family includes about 60 species. One species is present in Indonesia and Sri Lanka, several species in Arabia and Africa (tropical Africa and South Africa) [1], 26 species in the Mascarene Islands (Reunion Islands, Mauritius and Rodrigues) [2] and 28 species in Madagascar [3]. Some Psiadia species have been traditionally used for a long time to treat a variety of ailments such as abdominal pains [4], cold, fever [5, 6], bronchitis and asthma [7]. They are also used in casts for broken bones by the Bedouins [8] or to treat minor wounds and burns [9]. P. punctulata roots have been used by the Maasai to fight malaria [10].
P. altissima, P. salviifolia and P. lucida are three well-known species in the Malagasy pharmacopeia. Their leaves are notably used to treat tooth [3,11,12], and stomach aches [13] and skin diseases like scabies, eczema and wounds [11,12,14–16]. They are also recognised for their hemostatic, anti-diarrheal, carminative, anti-hypertensive and disinfectant properties [17–21].
As reported in a recent paper [22], several Psiadia species produce EOs [9,17,21,23–26] and contain various specialized metabolites including flavonoids [8, 27–29], terpenoids [23,30–32], phenylpropanoids [28,33–35] and coumarins [27, 36]. These metabolites displayed a variety of biological effects notably antimicrobial and antifungal [5, 6, 17, 24, 25, 37, 38], antiviral [39–43], antiplasmodial [30, 44–46], anti-inflammatory [36, 39, 47], cytotoxic [48, 49] and wound healing activity [38].
Considering the medicinal potential of the
genus Psiadia, extensive
investigation on six species endemic to Madagascar Island was undertaken. The
chemical composition of the EOs from P.
altissima, P. stenophylla, P. hispida, P. leucophylla, P. lucida
and P. salviifolia was studied. EO
components from three of them, namely P.
stenophylla, P. hispida and P. leucophylla were characterized for
the first time.
2. Materials and methods
2.1
Plant materials
Leaves of the six Psiadia species (Fig. 1) were collected in Madagascar, during their flowering periods (October-January). The species were botanically identified by the botanists of the National Herbarium of the Tsimbazaza Botanical and Zoological Garden in Antananarivo (Madagascar). Each collected specimen was deposited both in the national Herbarium of the Tsimbazaza Botanical and Zoological Garden and in the Herbarium of the University of Reunion. The date of harvest, collection site, herbarium voucher number, and geographic coordinates are listed in Table 1.
Figure 1. Pictures of the six Psiadia species investigated: a-
P. altissima (PAL), b- P.
stenophylla (PST), c- P. hispida (PHI), d- P. leucophylla (PLE), e- P.
lucida (PLU) and f- P. salviifolia (PSA).
Table 1. Date of harvest, collection site, herbarium voucher
number, and geographic coordinates for the Psiadia
specimens.
|
Species |
Date of harvest |
Collection site |
M/car voucher number |
Reunion vou-cher number |
Coordinates Latitude/Longitude |
Altitude (m) |
|
Psiadia altissima |
02 Nov.
2018 |
Moramanga |
RIR 3196 |
REU024089 |
18°58'16.7"S/48°19'16.7"E |
919 |
|
Psiadia stenophylla |
31 Oct. 2018 |
Behenjy |
RIR 3189 |
REU024090 |
19°15'22.7"S/47°28'53.0"E |
1 395 |
|
Psiadia hispida |
14 Jan.
2019 |
Angavokely |
RZK 8265 |
REU024086 |
18°55’38.1"S/47°44’13.2"E |
1 758 |
|
Psiadia leucophylla |
02 Nov.
2018 |
Angavobe |
RIR 3193 |
REU024087 |
18°55'34.9"S/47°45'07.8"E |
1 743 |
|
Psiadia lucida |
14 Jan.
2019 |
Andasibe |
RIR 3195 |
REU024082 |
18°55'18.8"S/48°25'30.9"E |
967 |
|
Psiadia salviifolia |
30 Oct. 2018 |
Antsirabe |
RIR 3185 |
REU024085 |
19°52'32.7"S/47°05'18.7"E |
1 648 |
m: meters;
M/car: Madagascar
2.2 Essential
oil extraction
After 24h of collection, the EOs were
separately extracted from the leaves of each species by hydrodistillation for three
hours using a Clevenger-type apparatus. The recovered oils were dried over
anhydrous sodium sulphate Na2SO4 and kept at 4 °C until
analysis. The extraction yields were calculated based on the weight of fresh
plant material and reported in Fig. 2.
2.3 Gas
chromatography analysis
The total chemical constituents were
identified using the GC Agilent Technologies 6890 Network, equipped with mass
spectrometer (MS) Agilent Technologies 5973 Network, with a SPB-5 MS
fused-silica capillary column (60 m × 0.32 mm i.d., 0.25 μm film thickness).
Helium was used as the carrier gas at a flow rate of 0.7 mL/min. The pure EO
was injected with a volume of 0.01 µL in a split ratio of 1:50 to enhance
sensitivity and minimize peak broadening. The injection was performed manually.
Oven temperature was programmed from 40 °C to 250 °C at 4 °C/min, and held at
250 °C for 50 min. Injector and source temperatures were set at 250 °C and 280
°C, respectively. The GC-MS instruments were operated at 70 eV in the
Electronic Ionisation (EI) mode and mass spectra (MS) were scanned in the range
20-400 Daltons.
Quantitative data regarding the volatile constituents were performed by using GC Varian CP-3800, coupled to a flame ionization detector (FID). The analysis is operating at the same conditions as GC-MS, except for the helium flow rate which was set at 1.0 mL/min. The injected volume of pure essential oil was 0.01 µL in splitless mode to ensure the complete transfer of the sample. The FID detector temperature was set at 270 °C.
2.4 Identification
and quantification of compounds
The retention index (RI) for all volatile components of the EOs was calculated using a homologous series of C8-C28 n-alkanes as standards. The identification of volatile components was carried out by comparing their retention index and their mass spectral fragmentation pattern with the literature [50,51] and data stored in MS libraries (Wiley 07 and NIST 02).
The relative amount of each identified compound, expressed as a percentage (%), was calculated by comparing the area of the corresponding peak in the chromatogram with the total area of detected peaks, without using FID correction factors. This approach allowed for the quantification of each compound in the EOs, based on their peak areas in the chromatogram.
3. Results and discussion
3.1 Essential oil yields
The EOs from the leaves of the six Psiadia species were obtained as a pale yellow to dark yellow liquid. Oil yields, calculated from the weight of fresh plant material, ranged from 0.04% to 0.80% as shown in Fig. 2, which is typical in Psiadia species [17, 21, 23, 24, 52, 53].
Figure 2. Essential
oil yields obtained from the leaves of Psiadia
species: P. altissima (PAL), P. stenophylla (PST), P. hispida (PHI), P. leucophylla (PLE), P. lucida (PLU) and P. salviifolia (PSA).
In an earlier study, oil yield ranging from 0.13% to 0.70% was reported for P. altissima [17, 26] and 0.18% for P. salviifolia [21]. Our results (0.25% for P. altissima and 0.19% for P. salviifolia) were consistent with these values which are typical to the two Psiadia species. EOs from P. lucida and P. leucophylla were obtained in 0.04% yield. This was a poor value compared to the literature value of 0.91% for P. lucida [25] which means that the oil yield of P. lucida may be growing site dependent. P. stenophylla and P. hispida exhibited the highest values of oil yield, 0.30% and 0.80% respectively. Interestingly, the oil yield of most Psiadia species from different origins did not exceed 1% [17, 23, 24, 52, 53].
3.2 Chemical composition
The chemical composition of six Psiadia species was investigated using both GC-MS and GC-FID techniques. Identification was performed by correlating their retention index with their mass spectra. Table 2 lists the components identified in their EOs, their RI, and their concentration in the different samples.
Table 2. Essential oils chemical composition of the six Psiadia species: P. altissima (PAL), P. stenophylla (PST), P. hispida (PHI), P.
leucophylla (PLE), P. lucida (PLU) and P. salviifolia (PSA).
|
No. |
Compound |
RI (exp) |
RI (lit) |
Composition (%) |
|||||
|
PAL |
PST |
PHI |
PLE |
PLU |
PSA |
||||
|
Alcohols |
|
|
|
|
|
|
|
|
|
|
1 |
(3E)-hexenol |
857 |
854 |
- |
- |
- |
0.4 |
0.2 |
- |
|
Aldehydes |
|
|
|
|
|
|
|
|
|
|
2 |
(2E)-hexenal |
853 |
855 |
tr |
- |
- |
- |
- |
- |
|
3 |
n-octanal |
1004 |
999 |
- |
- |
- |
0.2 |
- |
- |
|
Monoterpene hydrocarbons |
|
|
|
|
|
|
|
|
|
|
4 |
α-thujene |
931 |
930 |
0.1 |
- |
0.6 |
- |
- |
0.1 |
|
5 |
α-pinene |
940 |
939 |
4.5 |
4.7 |
4.2 |
- |
- |
2.4 |
|
6 |
camphene |
956 |
954 |
2.9 |
4.8 |
- |
- |
- |
- |
|
7 |
sabinene* |
980 |
975 |
0.4 |
- |
46.9 |
- |
0.5 |
1.4 |
|
8 |
β-pinene* |
987 |
979 |
44.9 |
34.9 |
tr |
- |
17.2 |
|
|
9 |
myrcene* |
993 |
991 |
- |
0.1 |
- |
2.1 |
||
|
10 |
α-phellandrene |
1011 |
1003 |
tr |
tr |
- |
- |
- |
2.1 |
|
11 |
p-mentha-1(7),8-diene |
1017 |
1004 |
tr |
- |
- |
- |
- |
0.5 |
|
12 |
α-terpinene* |
1022 |
1017 |
tr |
tr |
1.2 |
- |
- |
3.2 |
|
13 |
p-cymene* |
1030 |
1025 |
0.4 |
1.2 |
- |
- |
tr |
|
|
14 |
limonene* |
1035 |
1029 |
10.4 |
15.8 |
16 |
- |
0.4 |
28.5 |
|
15 |
β-phellandrene* |
1037 |
1030 |
- |
- |
- |
- |
3 |
|
|
16 |
(Z)-β-ocimene |
1039 |
1037 |
7.3 |
5.4 |
5.7 |
0.6 |
0.5 |
2.7 |
|
17 |
(E)-β-ocimene |
1050 |
1050 |
2.3 |
0.6 |
3 |
- |
- |
- |
|
18 |
γ-terpinene |
1064 |
1060 |
tr |
- |
- |
- |
- |
2.1 |
|
19 |
terpinolene |
1095 |
1089 |
0.3 |
0.5 |
0.5 |
- |
- |
2.9 |
|
20 |
p-1,3,8-menthatriene |
1126 |
1103 |
- |
- |
0.3 |
- |
tr |
- |
|
21 |
allo-ocimene |
1132 |
1132 |
0.1 |
- |
- |
- |
- |
- |
|
Oxygenated monoterpenes |
|
||||||||
|
22 |
(Z)-sabinene hydrate |
1075 |
1070 |
- |
- |
- |
- |
- |
0.4 |
|
23 |
linalol |
1103 |
1095 |
1.8 |
0.7 |
- |
1.3 |
0.1 |
tr |
|
24 |
(E)-sabinene hydrate |
1107 |
1098 |
- |
- |
0.1 |
- |
- |
tr |
|
25 |
α-fenchol |
1123 |
1117 |
tr |
0.1 |
- |
- |
- |
- |
|
26 |
(Z)-p-menth-2-en-1-ol |
1130 |
1122 |
tr |
- |
tr |
- |
- |
tr |
|
27 |
(E)-p-menth-2-en-1-ol |
1148 |
1141 |
- |
- |
tr |
- |
- |
tr |
|
28 |
borneol |
1177 |
1169 |
0.1 |
0.2 |
- |
- |
- |
- |
|
29 |
terpinen-4-ol |
1186 |
1177 |
0.7 |
0.4 |
2.7 |
- |
0.1 |
1 |
|
30 |
α-terpineol |
1199 |
1189 |
1.3 |
1.1 |
0.6 |
- |
tr |
0.2 |
|
31 |
fragranol |
1219 |
1216 |
- |
- |
- |
0.1 |
- |
- |
|
32 |
(E)-carveol |
1227 |
1217 |
- |
- |
- |
- |
- |
tr |
|
33 |
nerol |
1234 |
1230 |
0.1 |
- |
- |
tr |
- |
- |
|
34 |
thymol methyl ether |
1239 |
1235 |
- |
- |
- |
- |
tr |
- |
|
35 |
geraniol |
1258 |
1253 |
0.2 |
- |
- |
0.1 |
0.2 |
- |
|
36 |
bornyl acetate |
1293 |
1289 |
tr |
1 |
- |
- |
- |
- |
|
37 |
carquejol acetate |
1306 |
1299 |
0.1 |
- |
- |
tr |
- |
- |
|
38 |
(Z)-dimethoxy-citral |
1326 |
1318 |
tr |
- |
- |
- |
- |
- |
|
39 |
(E)-carvyl acetate |
1341 |
1342 |
- |
- |
- |
- |
- |
0.1 |
|
40 |
nerol acetate |
1365 |
1362 |
- |
- |
- |
0.3 |
- |
- |
|
41 |
verbanol acetate |
1372 |
1344 |
- |
- |
tr |
- |
- |
- |
|
42 |
(E)-myrtenyl acetate |
1388 |
1387 |
- |
- |
0.1 |
- |
- |
- |
|
43 |
(Z)-jasmone |
1417 |
1393 |
- |
- |
- |
- |
- |
0.1 |
|
44 |
neryl acetone |
1457 |
1436 |
- |
- |
- |
tr |
- |
- |
|
Sesquiterpene hydrocarbons |
|
||||||||
|
45 |
silphiperfol-5-ene |
1338 |
1329 |
- |
- |
- |
- |
- |
0.1 |
|
46 |
δ-elemene |
1347 |
1338 |
0.7 |
- |
0.2 |
0.1 |
0.1 |
0.6 |
|
47 |
7-epi-silphiperfol-5-ene* |
1357 |
1348 |
- |
- |
- |
- |
- |
0.6 |
|
48 |
α-cubebene* |
1359 |
1351 |
tr |
0.1 |
- |
0.8 |
0.2 |
|
|
49 |
silphiperfola-4,7(14)-diene |
1370 |
1361 |
- |
- |
- |
- |
- |
0.1 |
|
50 |
α-ylangene |
1384 |
1375 |
0.1 |
0.1 |
- |
- |
tr |
- |
|
51 |
α-copaene |
1388 |
1377 |
0.4 |
0.5 |
- |
5 |
0.4 |
0.3 |
|
52 |
β-maaliene |
1397 |
1382 |
- |
- |
- |
- |
- |
0.8 |
|
53 |
7-epi-sesquithujene |
1397 |
1391 |
- |
0.1 |
- |
- |
- |
- |
|
54 |
β-bourbonene* |
1398 |
1388 |
- |
- |
- |
- |
4.1 |
- |
|
55 |
β-cubebene* |
1402 |
1388 |
- |
- |
- |
- |
- |
|
|
56 |
β-elemene* |
1402 |
1391 |
0.6 |
1.0 |
0.7 |
- |
1.2 |
|
|
57 |
cyperene |
1418 |
1399 |
- |
tr |
- |
- |
- |
- |
|
58 |
α-gurjunene* |
1424 |
1410 |
tr |
0.1 |
- |
0.2 |
0.1 |
0.1 |
|
59 |
β-cedrene* |
1427 |
1421 |
- |
- |
- |
- |
- |
|
|
60 |
longifolene |
1433 |
1408 |
tr |
- |
- |
- |
- |
- |
|
61 |
β-ylangene* |
1434 |
1421 |
- |
- |
- |
tr |
2.5 |
- |
|
62 |
β-caryophyllene* |
1436 |
1419 |
1.7 |
2.3 |
1.0 |
10.6 |
2.9 |
|
|
63 |
β-copaene |
1444 |
1432 |
- |
0.3 |
- |
4.6 |
1.1 |
- |
|
64 |
γ-elemene |
1444 |
1437 |
- |
- |
- |
- |
- |
- |
|
65 |
β-gurjunene |
1444 |
1434 |
0.7 |
- |
- |
- |
tr |
1 |
|
66 |
α-guaiene |
1450 |
1440 |
- |
- |
0.1 |
- |
- |
- |
|
67 |
aromadendrene |
1460 |
1441 |
- |
- |
0.1 |
- |
tr |
- |
|
68 |
(Z)-muurola-3,5-diene* |
1462 |
1450 |
0.9 |
- |
- |
3.2 |
tr |
0.6 |
|
69 |
(E)-muurola-3,5-diene* |
1465 |
1454 |
0.3 |
- |
- |
- |
- |
|
|
70 |
α-humulene |
1471 |
1455 |
1.3 |
3.7 |
1.7 |
6.9 |
2.9 |
1.4 |
|
71 |
(Z)-muurola-4(14),5-diene |
1478 |
1467 |
0.2 |
0.1 |
tr |
1.5 |
0.9 |
0.3 |
|
72 |
(E)-cadina-1(6),4-diene |
14 87 |
1477 |
tr |
- |
tr |
- |
- |
- |
|
73 |
γ-curcumene |
1489 |
1483 |
0.7 |
5.8 |
- |
- |
- |
2.2 |
|
74 |
γ-muurolene |
1491 |
1480 |
0.3 |
- |
0.3 |
4.6 |
2.4 |
0.3 |
|
75 |
α-amorphene |
1494 |
1485 |
- |
- |
- |
- |
- |
- |
|
76 |
germacrene D |
1498 |
1485 |
0.9 |
1.0 |
- |
- |
- |
- |
|
77 |
β-selinene |
1504 |
1490 |
0.7 |
- |
- |
- |
- |
1.4 |
|
78 |
(E)-muurola-4(14),5-diene |
1509 |
1494 |
0.2 |
0.6 |
0.1 |
7.2 |
0.9 |
2.3 |
|
79 |
α-muurolene |
1513 |
1500 |
- |
- |
tr |
3.6 |
10.6 |
- |
|
80 |
(E)-β-guaiene |
1513 |
1503 |
- |
2.3 |
- |
- |
6.9 |
- |
|
81 |
(Z)-β-guaiene |
1514 |
1494 |
3.5 |
0.5 |
- |
- |
- |
1 |
|
82 |
γ-amorphene |
1514 |
1496 |
- |
- |
- |
- |
- |
1.4 |
|
83 |
β-humachalene |
1520 |
1505 |
- |
0.6 |
- |
- |
- |
- |
|
84 |
α-bulnesene |
1520 |
1510 |
- |
- |
0.3 |
- |
- |
- |
|
85 |
δ-amorphene |
1521 |
1512 |
0.2 |
- |
- |
2.4 |
1 |
0.9 |
|
86 |
γ-cadinene |
1528 |
1514 |
0.7 |
0.6 |
0.2 |
2.6 |
3.1 |
1.1 |
|
87 |
δ-cadinene |
1537 |
1523 |
1.7 |
0.3 |
1 |
8.9 |
7.5 |
3.3 |
|
88 |
zonarene |
1541 |
1530 |
tr |
0.1 |
- |
- |
- |
0.7 |
|
89 |
(E)-cadina-1(2),4-diene |
1547 |
1535 |
0.1 |
tr |
0.1 |
0.2 |
0.4 |
- |
|
90 |
α-cadinene |
1552 |
1539 |
0.2 |
tr |
- |
1 |
0.2 |
- |
|
91 |
α-calacorene |
1552 |
1546 |
- |
0.4 |
- |
2.6 |
0.4 |
1.2 |
|
92 |
germacrene B |
1577 |
1561 |
0.2 |
- |
- |
- |
0.8 |
0.1 |
|
93 |
β-calacorene |
1580 |
1566 |
- |
- |
- |
0.1 |
- |
- |
|
94 |
cadalene |
1693 |
1677 |
- |
- |
- |
0.2 |
- |
- |
|
Oxygenated sesquiterpenes |
|
||||||||
|
95 |
epi-cubebol |
1509 |
1494 |
- |
- |
- |
- |
3.6 |
1 |
|
96 |
cubebol |
1531 |
1515 |
tr |
- |
0.2 |
2.6 |
tr |
0.5 |
|
97 |
10-epi-cubebol |
1554 |
1535 |
- |
- |
- |
- |
- |
2.8 |
|
98 |
elemol |
1563 |
1550 |
0.4 |
0.6 |
- |
- |
3.5 |
0.3 |
|
99 |
(E)-nerolidol |
1569 |
1563 |
0.3 |
0.2 |
0.1 |
tr |
- |
- |
|
100 |
(E)-cadinene ether |
1587 |
1559 |
- |
- |
- |
- |
- |
0.1 |
|
101 |
ledol |
1588 |
1569 |
- |
0.2 |
- |
- |
1.1 |
- |
|
102 |
(Z)-muurol-5-en-4-α-ol* |
1591 |
1561 |
- |
- |
- |
- |
- |
0.2 |
|
103 |
germacrene D-4-ol* |
1592 |
1576 |
- |
- |
tr |
tr |
tr |
|
|
104 |
spathulenol |
1597 |
1578 |
- |
- |
- |
0.2 |
0.3 |
- |
|
105 |
guaiol |
1610 |
1601 |
tr |
0.1 |
- |
- |
- |
- |
|
106 |
gleenol |
1600 |
1587 |
- |
- |
tr |
- |
- |
- |
|
107 |
caryophyllene oxide |
1603 |
1583 |
- |
- |
- |
0.1 |
0.2 |
0.2 |
|
108 |
(Z)-β-elemone |
1618 |
1590 |
- |
- |
- |
- |
- |
- |
|
109 |
viridiflorol |
1625 |
1593 |
- |
0.2 |
- |
0.4 |
1.6 |
0.3 |
|
110 |
humulene epoxyde II |
1630 |
1608 |
- |
- |
- |
- |
0.5 |
- |
|
111 |
β-copaen-4-α-ol |
1631 |
1591 |
- |
- |
- |
- |
- |
0.1 |
|
112 |
1,10-di-epi-cubenol |
1633 |
1619 |
tr |
0.1 |
- |
0.2 |
tr |
- |
|
113 |
10-epi-γ-eudesmol |
1642 |
1624 |
tr |
0.1 |
- |
- |
tr |
- |
|
114 |
1-epi-cubenol |
1647 |
1629 |
0.1 |
0.2 |
tr |
tr |
0.5 |
0.1 |
|
115 |
γ-eudesmol |
1651 |
1632 |
0.2 |
0.2 |
- |
- |
1.0 |
- |
|
116 |
τ-cadinol* |
1658 |
1640 |
0.2 |
0.3 |
0.2 |
1.3 |
7.3 |
0.7 |
|
117 |
τ-muurolol* |
1661 |
1642 |
0.1 |
|||||
|
118 |
α-muurolol* |
1664 |
1646 |
- |
0.2 |
||||
|
119 |
β-eudesmol* |
1672 |
1651 |
0.8 |
0.6 |
- |
- |
11.1 |
tr |
|
120 |
α-cadinol* |
1674 |
1654 |
0.2 |
0.4 |
1.2 |
0.4 |
||
|
121 |
α-eudesmol |
1677 |
1654 |
0.1 |
0.2 |
- |
- |
- |
- |
|
122 |
epi-β-bisabolol* |
1681 |
1672 |
- |
0.3 |
- |
- |
- |
- |
|
123 |
selin-11-en-4-α-ol* |
1683 |
1660 |
0.1 |
- |
0.1 |
- |
0.1 |
|
|
124 |
(E)-14-hydroxy-9-epi-caryophyllene |
1690 |
1670 |
- |
- |
- |
- |
- |
0.1 |
|
125 |
epi-α-bisabolol |
1695 |
1685 |
- |
tr |
- |
- |
- |
- |
|
126 |
α-bisabolol |
1697 |
1686 |
- |
tr |
- |
- |
- |
- |
|
127 |
khusinol |
1706 |
1680 |
- |
- |
tr |
- |
- |
- |
|
128 |
eudesma-4(15),7-dien-1-β-ol |
1706 |
1688 |
- |
- |
- |
0.3 |
0.1 |
- |
|
129 |
(E)-α-bergamotol |
1711 |
1691 |
- |
- |
- |
- |
- |
0.1 |
|
130 |
oplopanone* |
1760 |
1740 |
- |
- |
- |
- |
0.4 |
- |
|
131 |
cyclocolorenone* |
1763 |
1761 |
- |
- |
- |
- |
- |
|
|
132 |
14-oxy-α-muurolene |
1787 |
1769 |
- |
- |
- |
- |
0.3 |
- |
|
133 |
14-hydroxy-α-muurolene |
1793 |
1780 |
- |
- |
- |
tr |
0.2 |
- |
|
134 |
bicyclovetivenol |
1815 |
1793 |
- |
- |
- |
- |
0.4 |
- |
|
135 |
14-hydroxy-δ-cadinene |
1820 |
1804 |
- |
- |
- |
- |
tr |
- |
|
136 |
(Z)-lanceol acetate |
1964 |
1856 |
- |
- |
- |
- |
- |
tr |
|
Sulfurized sesquiterpenes |
|
|
|
|
|
|
|
||
|
137 |
mint sulfide |
1764 |
1741 |
- |
- |
- |
0.1 |
- |
- |
|
|
Diterpene hydrocarbons |
|
|
|
|
|
|
|
|
|
138 |
rimuene |
1925 |
1896 |
- |
- |
- |
- |
- |
tr |
|
139 |
pimaradiene |
1987 |
1950 |
tr |
- |
- |
0.9 |
- |
- |
|
140 |
cembrene |
2004 |
1939 |
- |
- |
- |
- |
- |
0.1 |
|
141 |
sclarene |
2071 |
1975 |
- |
- |
- |
0.9 |
- |
- |
|
142 |
kaurene |
2078 |
2043 |
- |
- |
- |
- |
- |
0.1 |
|
Oxygenated diterpenes |
|
|
|
|
|
|
|
||
|
143 |
13-epi-oxyde de manol |
2046 |
2017 |
- |
- |
- |
- |
- |
0.4 |
|
144 |
manol |
2082 |
2057 |
tr |
- |
0.4 |
6.4 |
- |
tr |
|
145 |
phytol |
2116 |
1943 |
tr |
tr |
0.1 |
- |
tr |
- |
|
146 |
nezukol |
2230 |
2133 |
- |
- |
- |
tr |
- |
- |
|
Aromatic compounds |
|
|
|
|
|
|
|
||
|
147 |
perillene |
1118 |
1101 |
- |
0.3 |
- |
- |
tr |
- |
|
148 |
methyl eugenol |
1408 |
1404 |
- |
tr |
- |
- |
- |
- |
|
149 |
benzyl benzoate |
1783 |
1760 |
- |
- |
- |
tr |
0.1 |
- |
|
Total alcohols |
- |
- |
- |
0.4 |
0.2 |
- |
|||
|
Total aldehydes |
tr |
- |
- |
0.2 |
- |
- |
|||
|
Total monoterpene
hydrocarbons |
73.4 |
67.9 |
78.4 |
0.6 |
6.4 |
63.1 |
|||
|
Total oxygenated
monoterpenes |
4.2 |
3.4 |
3.5 |
1.8 |
0.3 |
1.8 |
|||
|
Total sesquiterpene
hydrocarbons |
16.2 |
20.7 |
5.7 |
66.4 |
46.6 |
25.7 |
|||
|
Total oxygenated
sesquiterpenes |
2.5 |
3.4 |
0.9 |
6.2 |
32 |
6.7 |
|||
|
Total sulfurized
sesquiterpenes |
- |
- |
- |
0.1 |
- |
- |
|||
|
Total diterpene hydrocarbons |
tr |
- |
- |
1.8 |
- |
0.1 |
|||
|
Total oxygenated diterpenes |
tr |
tr |
0.5 |
6.4 |
tr |
0.4 |
|||
|
Aromatic compounds |
- |
0.3 |
tr |
tr |
0.1 |
tr |
|||
|
Total identified |
96.3 |
95.8 |
89.0 |
84.0 |
85.6 |
97.9 |
|||
RI (exp): experimental
retention index; RI (lit): retention index from the literature [50,51]; tr = trace, less than
0.1%; -: absent. * Co-eluting compounds;
Composition (%) framed by dotted
line= % of
co-eluting compounds.
A total of 149 compounds
were identified for the six Psiadia species
EOs, representing from 84.0 to 97.9% of the total composition. These compounds
are divided into 10 chemical families, including monoterpene hydrocarbons,
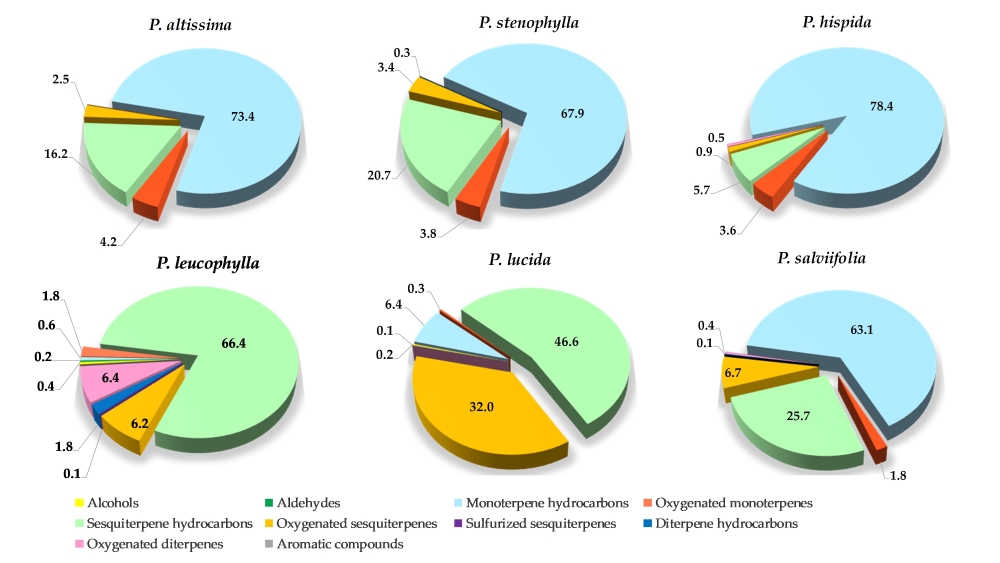
oxygenated sesquiterpenes and aromatic compounds. Fig. 3 illustrates how the
constituents in the EO of each species are distributed among the different
chemical families.
Figure 3. Distribution of EO
constituents in Psiadia species
categorized by chemical families.
The chemical composition analysis
of the EOs from the six Psiadia
species has revealed a diverse array of chemical families. This diversity highlighted
the complex nature of these EOs and underscored the potential for unique
aromatic and therapeutic properties and applications. In the following discussion,
the individual characteristics of each Psiadia
species, their chemical profiles and their potential target application areas will
be explored.
3.1.1
Psiadia altissima
In the chemical
compositions of P. altissima EO,
monoterpene hydrocarbons were identified as the main constituents, comprising 73.4%
of the composition, followed by sesquiterpene hydrocarbons (16.2%). Although
there were 26 sesquiterpene hydrocarbons in the sample, they were only present
at a concentration of 4.2%. In this EO, the major compounds were β-pinene (44.9%), limonene (10.4%), (Z)-β-ocimene
(7.3%) and α-pinene (4.5%). Previous
studies [17,26] have already investigated the
chemical composition of P. altissima
EO. They have found that the leaves of this species were rich in both monoterpene
and sesquiterpene hydrocarbons, with β-pinene
(39.7-49.7%), limonene (3.8-9.3%), (E,Z)-β-ocimene
(5.9-6.9%), and α-pinene (3.5-3.9%)
as major compounds. Our study on the chemical composition of P. altissima EO aligned with these
established findings. Additionally, sesquiterpene hydrocarbons were detected, including
β-caryophyllene (1.7%), α-humulene (1.3%), (Z)-β-guaiene (3.5%) and δ-cadinene (1.7%), which was consistent
with the earlier studies [17,26]. In contrast to the
findings of these previous investigations, ledene (3.9%), cyperene (3.4%),
viridiflorene (3.0%) and aromadendrene (1.2%) were not observed in the present
study.
3.1.2
Psiadia stenophylla and Psiadia
hispida
P. stenophylla and P. hispida,
both investigated for the first time, displayed a chemical composition
relatively close to that of P. altissima.
Their EOs are characterized by a significant concentration of monoterpene
hydrocarbons (67.9% and 78.4% respectively) as well as sesquiterpene
hydrocarbons (20.7% and 5.7% respectively). Similar to P. altissima EO, the main compounds in these two EOs were β-pinene (34.9-46.9%), limonene (15.8-16.0%),
(Z)-β-ocimene (5.4-5.7%) and α-pinene
(4,2-4.7%). Furthermore, their EOs included identical sesquiterpene
hydrocarbons, comprising β-caryophyllene,
α-humulene, and δ-cadinene. However, in P.
stenophylla, γ-curcumene (5.8%), α-humulene (3.7%), and (E)-β-guaiene
(2.3%) were found in higher quantities compared to P. altissima, where they were either absent or occurred in lower
amounts.
3.1.3
Psiadia leucophylla
In this initial
investigation of P. leucophylla,
84.0% of the EO chemical compositions were identified, with several minor
compounds that remained unidentified. P.
leucophylla exhibited significant dissimilarities in its chemical composition
compared to the other species investigated. A diverse array of chemical
families was observed in the EO, which was predominantly composed of
sesquiterpene hydrocarbons (66.4%). Additionally, it contained 6.4% of
oxygenated diterpenes and 6.2% of oxygenated sesquiterpenes. In contrast to the
other species, P. leucophylla had notably
very low levels of hydrocarbon monoterpenes (0.6%). The EO also contained other
compound families but in minimal quantities, such as alcohols (0.4%), aldehydes
(0.2%), sulphurated sesquiterpenes (0.1%), and trace amounts of aromatic
compounds. The major compounds in this EO included β-caryophyllene (10.6%), δ-cadinene
(8.9%), (E)-muurola-4(14),5-diene
(7.2%), α-humulene (6.9%), and manool
(6.4%). Additionally, the EO also showed a significant amount of α-copaene (5.0%), β-copaene (4.6%), ɤ-muurolene (4.6%), and α-muurolene (3.6%).
3.2.4 Psiadia lucida
The EO of P. lucida has revealed high levels of
sesquiterpene hydrocarbons (46.6%) and oxygenated sesquiterpenes (32.0%).
Several compounds were identified in relatively significant amounts, including α-cadinol (11.1%), α-muurolene (10.6%), δ-cadinene
(7.5%), τ-muurolol (7.3%) and (Z)-β-guaiene
(6.9%). Notably, the results from this study showed a qualitative difference
compared to the previous research conducted on P. lucida [25]. In this earlier study,
terpinolene (38.0%), α-humulene
(21.2%), and limonene (10.2%) were identified as the main compounds [25], which differed from the
current results. These findings suggest that the chemical profile of P. lucida EO may have varied depending
on the harvest date and/or collection location.
3.2.5
Psiadia salviifolia
P. salviifolia chemical compositions were characterized by a high proportion
of monoterpene hydrocarbons (63.1%) and sesquiterpene hydrocarbons (25.7%). The
most abundant compounds were β-pinene
(17.2%) and limonene (28.5%). This composition was relatively close to that of P. altissima, P. stenophylla and P. hispida.
These results were consistent with previous research by Andrianarison [13], which reported monoterpene
hydrocarbons (76.7%) as the predominant chemical family in the EO of P. salviifolia. The major compounds
identified in that study included β-pinene
(22.1%), limonene (19.9%), β-phellandrene
(10.4%), and sabinene (9.7%). However, the present study did not corroborate
the results of the initial investigation of this species by Dennis [21]. According to Dennis, EO
was primarily composed of a mixture of linalool and β-bourbonene (10.7%), along with significant amounts of oxygenated
sesquiterpenes (25.8%) and diterpenes (11.8%). In our study, the major
compounds identified were β-pinene (17.2%)
and limonene (28.5%), which were also reported by Dennis but at relatively
lower levels (4.6 % and 2.1%, respectively).
In previous studies,
several major compounds that are present in Psiadia
species EOs have been highlighted for their biological activities. Specifically,
studies have highlighted the strong effects of compounds such as (+)-α-pinene and (-)-α-pinene [54–57], (-)-β-pinene
[58] and (+)-β-pinene [59], (+)-limonene [60, 61] and (-)-limonene [61], β-caryophyllene [62] and δ-cadinene [63, 64] against pathogenic yeasts
and bacteria. Because of the hemostatic and disinfectant properties of these
compounds, our findings therefore validated the traditional uses of P. altissima, P. lucida, and P. salviifolia
species in the treatment of various wounds such as scabies, eczematous,
ulcerated, and syphilitic. Our findings also validated their potential as a
remedy against a variety of microbial infections, including diarrhea [18]. It is important to determine the
enantiomeric composition of the studied essential oil as it significantly
influenced its biological activity and potential therapeutic effects. Additionally,
previous studies on P. altissima, P. lucida and P. salviifolia EOs have demonstrated their antimicrobial
activities against Gram-positive strains, including Staphylococcus aureus, Sarcina
lutea, Enterococcus faecalis and Bacillus subtilis, as well as
Gram-negative strains such as Escherichia
coli, Salmonella enteridis, Salmonella
typhii, Shigella boydii and Branhamella catarrhalis [13, 17, 25,
38]. Furthermore, P. altissima and P. salviifolia EOs have exhibited antifungal properties against Candida albicans [13,38].
Moreover, the EO from P. altissima leaves has been
specifically studied for its in vivo wound-healing
activity [38]. In this study, significant
wound-healing properties were revealed, with 98.37% healing after 14 days of
treatment, which was close to the activity of a standard reference ointment.
This aligned with the traditional use of this species for treating wounds [11,14]. This activity may be
attributed to the presence of specific compounds detected in the EO, notably α-pinene enantiomers and β-caryophyllene, both of which have been
previously shown to have wound-healing properties [65, 66].
According to the
literature, the main compounds highlighted in the studied EOs, notably α-pinene
[67,68], limonene [69,70], α-humulene [71], and β-caryophyllene [62, 72]) have
also displayed significant anti-inflammatory and antioxidant potential. These results
supported the traditional uses of P.
altissima, P. lucida and P.
salviifolia species, especially in remedying ailments such as bronchitis,
cough and asthma, potentially due to their ability to reduce respiratory system
inflammation. Furthermore, Rakotomalala reported that the EOs from the aerial
parts of P. altissima had strong
broncho-relaxant properties [38].
Most of the predominant
compounds found in our EOs have demonstrated cytotoxic and antitumor properties,
including α-pinene [73], β-pinene [74], limonene [75], (Z)-β-ocimene [76], β-caryophyllene [62], α-cadinol [77], and manool [78]. These findings suggest
that the six Psiadia EOs may have
broader applications in the treatment of various ailments and potentially even
in addressing issues related to abnormal tissue growth. This highlights the
significance of further research and suggests the potential for harnessing the
rich medicinal properties of Psiadia species
to develop novel therapeutic agents for modern healthcare, particularly by
further exploring their cytotoxic properties.
Beyond these categories, the six Psiadia EOs contained several major compounds with a diverse range of biological activity. Antileishmanial activity has been demonstrated for (Z)-β-ocimene [76]. β-pinene [79] and α-humulene [80] both had a protective role against gastric injury. This may support the lengthy decoction of P. altissima leaves and stems that were prescribed in traditional pharmacopoeia to cure ulcerated wounds [18]. Antimalarial effects of α-pinene and β-caryophyllene have been reported [81]. Furthermore, α-pinene has shown therapeutic effect in ovalbumin (OVA)-sensitized allergic rhinitis [82]. These compounds could play roles in traditional applications, possibly in treating colds and fever.
4. Conclusions
In the current investigation, the chemical composition of EOs extracted from fresh leaves of six Psiadia species endemic to Madagascar was evaluated. This was the first report on the volatile phytochemicals found in P. stenophylla, P. hispida, and P. leucophylla. This research revealed that the EOs from four species, namely P. altissima, P. stenophylla, P. hispida, and P. salviifolia, had a similar chemical composition. Their EOs were dominated by monoterpenes and sesquiterpenes hydrocarbons. In contrast, the remaining two species, P. lucida and P. leucophylla, displayed distinct chemical profiles. The EO of P. lucida was rich in sesquiterpene hydrocarbons and oxygenated sesquiterpenes. P. leucophylla was characterized by sesquiterpene hydrocarbons and oxygenated diterpenes. These results not only contributed to the chemical knowledge of some Psiadia species but also lent support to their traditional uses and biological potential. Moreover, this finding offered a promising outlook for potential applications and therapeutic properties associated with the major compounds in these species, emphasizing the importance of further exploration into the multifaceted benefits that these EOs may offer.
Authors’ contributions
Conceptualization, L.E.R., A.G.B., I.G. and
J.R.; Methodology, L.E.R., A.G.-B., I.G. and J.R.; Resources, L.E.R., A.R.
(plant collections); Chemical investigation (extraction and analysis), L.E.R.;
Writing-original draft preparation, L.E.R.; Writing-review and editing, L.E.R.,
A.G.-B., I.G., A.R. and J.R.; Supervision, A.G.B., I.G. and J.R.; Project
administration and funding acquisition, A.G.B.
The authors express their gratitude to José
Rakotoandrianomanana from the chemistry laboratory, École Supérieure
Polytechnique d’Antananarivo, for his invaluable assistance during the plant
collections. We acknowledge the contributions of Richardson Razakamalala and
Richard Randrianarivo, botanists from Missouri Botanical Garden (MBG)
(Madagascar) for their expertise in plant identification. We also extend our
sincere appreciation to Patricia Clerc from the ChemBioPro laboratory for his
technical expertise in handling specific instruments and his invaluable
assistance during the operation of the equipment.
Funding
This work was funded by the European
Regional Development Funds GURDTI 2018-1828-0002370 (FEDER PHAR, EU-Région
Réunion-French State national counterpart). Razafindrabenja Lantomalala Elsa is
a recipient of a fellowship from the Région Réunion (DIRED/20171403).
Availability of data and
materials
All data
will be made available on request according to the journal policy.
Conflicts of interest
The authors declare no conflict of
interest.
References
1. Bosser
J.; Guého J.; Jeffrey C. Flore des Mascareignes: La Réunion, Maurice,
Rodrigues. 109. Composées; Reduit: Port Louis Maurice;
The Sugar Industry Research Institute:
Mauritius, L’Institut Français
de Recherche Scientifique pour le Developpement en Cooperation (ORSTOM): Paris;
The Royal Botanic
Gardens: Kew London,
1993; ISBN 2-7099-1407-7.
2. Cordemoy, E.J. Flore de l’île de la Réunion:
(phanérogames, cryptogames, vasculaires, muscinées) avec l’indication des propriétés
économiques et industrielles des plantes;
Librairie des Sciences Naturelles Paul Klincksieck: Paris France; J. Cramer, H. K. Swann Editions: New York USA, 1895; ISBN: 3-7682 -0758-7.
3.
Humbert, H. H. Flore de Madagascar et des Comores:
plantes vasculaires; Muséum National d’Histoire Naturelle: Paris, 1960.
4.
Kokwaro, J.O. Medicinal plants of east Africa; East african literature bureau:
Kampala, 1976; ISBN: 978-9966846846.
5.
Aumeeruddy-Elalfi, Z.; Gurib-Fakim,
A.; Mahomoodally, F. Antimicrobial, antibiotic potentiating activity and
phytochemical profile of essential oils from exotic and endemic medicinal
plants of Mauritius. Ind. Crops Prod.
2015, 71, 197–204.
6.
Wang, Y.; Hamburger, M.; Gueho, J.;
Hostettmann, K. Antimicrobial flavonoids from Psiadia trinervia and
their methylated and acetylated derivatives. Phytochem. 1989, 28, 2323–2327.
7.
Al-Yahya, M.A.; Hifnawy, M.S.; Mossa,
J.S.; El-Feraly, F.S.; McPhail, D.R.; McPhail, A.T. X-ray structure of
psiadiarabin, a flavone from Psiadia
arabica. Phytochem. 1987, 26, 2648-2649.
8.
Abou-Zaid, M.M.; El-Karemy, Z.;
EI-Negoumy, S.I.; Altosaar, I.; Saleh, N.A.M. The flavonoids of Psiadia punctulata. Bull. Chem. Soc. Ethiop. 1991, 5, 37–40.
9.
Gurib-Fakim, A.; Bourrel, C.; Kodja,
H.; Govinden, J. Chemical composition of the essential oils of Psiadia lithospermifolia (Lam.) Cordem.
and P. Viscosa (Lam.) A. J. Scott of
the Asteraceae family. J. Essent. Oil
Res. 1995, 7, 533–535.
10.
Koch, A.; Tamez, P.; Pezzuto, J.;
Soejarto, D. Evaluation of plants used for antimalarial treatment by the Maasai
of Kenya. J. Ethnopharmacol. 2005, 101, 95–99.
11.
Danthu, P.; Rakotobe, M.; Mauclère, P.;
Andrianoelisoa, H.; Behra, O.; Rahajanirina, V.; Mathevon, B.; Ralembofetra,
E.; Collas de Chatelperron, P. Essential oil production increases value of Psiadia altissima fallows in
Madagascar’s eastern forests. Agrofor.
Syst. 2008, 72, 127–135.
12.
Samyn, J.-M. Plantes utiles de hautes
terres de Madagascar, 1st Ed.; Librairie d'Antananarivo: Madagascar, 1999.
13.
Andrianarison, E.R.; Rakotosaona, R.;
Andrianaivoravelona, O.J.; Andrianarison, R.J. Composition chimique et activité
antimicrobienne d’huiles essentielles de Psiadia
salviifolia Baker (Asteraceae) ou « kijitina » provenant de la région Amoron’i Mania à Madagascar.
Mada-hary. 2015, 4, 1-9.
14.
Nicolas, J.-P. Plantes médicinales du
Nord de Madagascar: Ethnobotanique antakarana et informations scientifiques.
Jardins du monde, Brasparts: Antsiranana Madagascar, 2012; ISBN:
978-2-9543726-0-0.
15.
Baron, R.; Dorr, L.J. Compendium des
plantes malgaches. Taxon. 1906, 36, 39–46.
16.
Pernet, R. Pharmacopées de Madagascar.
Institut de Recherche Scientifique: Tananarive-Tsimbazaza, Madagascar, 1957.
17.
Ramanoelina, P.A.R.; Rasoarahona,
J.R.E.; Masotti, V.; Viano, J.; Gaydou, E.M.; Bianchini, J.P. Chemical
composition of the leaf oil of Psiadia
altissima (Compositeae). J.
Essent. Oil Res. 1994, 6, 565–570.
18.
Razafindrambao, R. Contribution à
l’inventaire des plantes médicinales des hauts plateaux de Madagascar. Debray:
Maurice ; ORSTOM: Paris France, 1971.
19.
Rakotondrafara, A.; Rakotondrajaona, R.;
Rakotoarisoa, M.; Ratsimbason, M.; Rasamison, V.; Rakotonandrasana, S.R.
Ethnobotany of medicinal plants used by the Zafimaniry clan in Madagascar. J.
Phytopharm. 2018, 7, 483–494.
20.
Debray, M. Médecine et pharmacopée
traditionnelles à Madagascar. Etudes Médicales 1975, 1, 69–83.
21.
Dennis, R. Essential oil of Psiadia salviifolia. Phytochem. 1973, 12, 2705–2708.
22.
Mahadeo, K.; Grondin, I.; Kodja, H.;
Govinden-Soulange, J.; Jhaumeer-Laulloo, S.B.; Frédérich, M.; Gauvin-Bialecki,
A. The genus Psiadia: Review of
traditional uses, phytochemistry and pharmacology. J. Nat. Prod. 2018, 82, 1361–1366.
23.
Gauvin-Bialecki, A.; Susperregui, J.;
Barthes, P.; Louis, R.; Deleris, G.; Smadja, J. An acetylated monoterpene and a
sesquiterpene alcohol from Psiadia
anchusifolia. Phytochem. 2004, 7,
897-901.
24.
Govinden-Soulange, J.; Magan, N.;
Gurib-Fakim, A.; Gauvin, A.; Smadja, J.; Kodja, H. Chemical composition and in vitro antimicrobial activities of the
essential oils from endemic Psiadia
species growing in Mauritius. Biol. Pharm. Bull. 2004, 27,
1814–1818.
25.
Andriamanantoanina, H.; Mananjarasoa,
E.; Ramaroson, L.; Casabianca, H.; Grenier-Loustalot, M.F. Composition and
antimicrobial activity of the leaf of Psiadia Lucida (Cass.) Drake
(Asteraceae). J. Essent. Oil Res. 2004,
16, 623–625.
26.
Rakotomalala, N.H.; Razafimandefitra,
A.; Rabehaja, D.; Rasolondramanitra, J. Étude de la composition chimique des
huiles essentielles de Psiadia altissima
(Asteraceae), plante médicinale endémique de Madagascar. Afr. Sci. Rev. Int.
Sci. Technol. 2016, 12, 1–11.
27.
Jakobsen, T.H.; Marcussen, H.V.;
Adsersen, A.; Strasberg, D.; Smitt, U.W.; Jaroszewski, J.W. 3-Methoxyflavones
and a novelcoumarin from Psiadia dentata.
Biochem. Syst. Ecol. 2001, 29, 963–965.
28.
Marie, D.; Gurib-Fakim, A.; Gray, A.;
Waterman, P. Constituents of Psiadia
terebinthina A.J. Scott, an endemic Asteraceae from Mauritius. Nat. Prod. Res. 2006, 20, 1169–1175.
29.
Mossa, J.S.; El-domiaty, M.; Al-meshal,
I.; El-Feraly, F.; Hufford, C.D.; McPhail, D.; McPhail, A. A flavone and
diterpene from Psiadia arabica. Phytochem. 1992, 31, 2863–2868.
30.
Mahadeo, K.; Herbette, G.; Grondin, I.;
Jansen, O.; Kodja, H.; Soulange, J.; Jhaumeer-Laulloo, S.; Clerc, P.;
Gauvin-Bialecki, A.; Frederich, M. Antiplasmodial diterpenoids from Psiadia arguta. J. Nat. Prod. 2019, 82, 1361–1366.
31.
Midiwo, J.; Owuor, F.A.O.; Juma, B.F.;
Waterman, P.G. Diterpenes from the feaf exudate of Psiadia punctulata. Phytochem.
1997, 45, 117–120.
32.
Canonica, L.; Rindone, B.; Scolastico,
C.; Ferrari, G.; Casagrande, C. Structure and stereochemistry of psiadiol, a
new diterpenoid. Tetrahedron Lett. 1967,
8, 2639–2643.
33.
Keriko, J.M.; Nakajima, S.; Baba, N.;
Iwasa, J. Eicosanyl p-coumarates from
a Kenyan plant, Psiadia punctulata:
plant growth inhibitors. Biosci., Biotechnol.,
Biochem. 1997, 61, 2127–2128.
34.
Juma, B.F.; Yenesew, A.; Midiwo, J.O.;
Waterman, P.G. Flavones and phenylpropenoids in the surface exudate of Psiadia punctulata. Phytochem. 2001, 57, 571–574.
35.
Wang, Y.; Hamburger, M.; Gueho, J.;
Hostettmann, K. Cyclohexanecarboxylic-acid derivatives from Psiadia trinervia. Helv. Chim. Acta. 1992, 75, 269–275.
36.
Fortin, H.; Tomasi, S.; Jaccard, P.;
Robin, V.; Boustie, J. A prenyloxycoumarin from Psiadia dentata. Chem. Pharm. Bull. 2001, 49, 619–621.
37.
Kauroo, S.; Govinden-Soulange, J.;
Marie, D.E.P. Endemic Asteraceae from Mauritius islands as potential
phytomedicines. Int. J. Chem. Environ. Biol. Sci. 2016, 4, 23–27.
38.
Rakotomalala, H. Étude chimique et biochimique d’une plante endémique de
Madagascar : Psiadia
altissima var. altissima Benth.et
Hook. ou Dingadingana, DEA Thesis. University of Antananarivo, Madagascar,
2014.
39.
Robin, V.; Boustie, J.; Amoros, M.;
Girre, L. In vitro antiviral activity
of seven Psiadia species, Asteraceae:
Isolation of two antipoliovirus flavonoids from Psiadia dentata. J. Pharm. Pharmacol. 1998, 4, 61–64.
40.
Robin, V.; Irurzun, A.; Amoros, M.;
Boustie, J.; Carrasco, L. Antipoliovirus flavonoids from Psiadia Dentata.
Antiviral Chem. Chemother.
2001, 12, 283–291.
41.
Fortin, H.; Vigor, C.; Devehat, F.L.L.;
Robin, V.; Bosse, B.L.; Boustie, J.; Amoros, M. In vitro antiviral activity of thirty-six plants from La Reunion
island. Fitoterapia. 2002, 73, 346–350.
42.
Vanden Berghe, D.A.; Vhetinck, A.J.; Van
Hoof, L. Plant products as potential antiviral agents. Pediatr. Infect. Dis. J. 1986, 6, 226–227.
43.
Castrillo, J.; Berghe, D.V.; Carrasco,
L. 3-Methylquercetin is a potent and selective inhibitor of poliovirus RNA
synthesis. Virology. 1986, 152, 219–227.
44.
Jonville, M.C.; Kodja, H.; Humeau, L.;
Fournel, J.; De Mol, P.; Cao, M.; Angenot, L.; Frédérich, M. Screening of
medicinal plants from Reunion island for antimalarial and cytotoxic activity. J. Ethnopharmacol. 2008, 120, 382–386.
45.
Bero, J.; Frédérich, M.;
Quetin‐Leclercq, J. Antimalarial compounds isolated from plants used in
traditional medicine. J. Pharm.
Pharmacol. 2009, 61, 1401–1433.
46.
Abdel-Sattar, E.; Maes, L.; Salama, M.M.
In vitro activities of plant extracts
from Saudi Arabia against malaria, leishmaniasis, sleeping sickness and chagas
diseas. Phytother. Res. 2010, 24, 1322–1328.
47.
Mahadeo, K. Étude métabolomique et
valorisation pharmacologique et biotechnologique d’éspèces du genre Psiadia endémiques de la Réunion et de
l’ile Maurice, Ph.D. Thesis, University of
Reunion Island, 2018.
48.
Kauroo, S.; Govinden-Soulange, J.;
Ranghoo-Sanmukhiya, V.M.; Miranda, K.; Cotham, W.E.; Walla, M.D.; Nagarkatti,
M.; Nagarkatti, P. Extracts of select endemic plants from the republic of
Mauritius exhibiting anti-cancer and immunomodulatory properties. Sci. Rep. 2021, 11, 1–27.
49.
Orabi, K.; Abaza, M.; Kurien, S.
Plectranthone and psiadin as anticancer leads. Planta Med. 2015, 81, PB15.
50.
Adams, R.P. Identification of essential
oil components by gas chromatography/quadrupole mass spectroscopy, 3rd ed., Allured Publishing
Corporation: Illinois USA, 2001; ISBN: 978-0931710858.
51.
Adams, R.P. Identification of essential
oil components by gaz chromatography/mass spectrometry. 5th ed. Texensis
Publishing Gruver: Texas USA, 2017; ISBN: 978-0-9981557-2-2.
52.
Mekkawi, A.G.; Mossa, J.S.; Hifnawy,
M.S.; Karawya, M.S. Essential oil of Psiadia
arabica Jaub. et Spach. Pharmazie. 1984, 39, 419–420.
53.
Gauvin, A.; Smadja, J. Essential oil
composition of four Psiadia species
from Reunion Island: A chemotaxonomic study. Biochem. Syst. Ecol. 2005,
33, 705–714.
54.
Da Silva Rivas, A.C.; Lopes, P.M.; de
Azevedo Barros, M.M.; Costa Machado, D.C.; Alviano, C.S.; Alviano, D.S.
Biological activities of α-pinene and
β-pinene enantiomers. Molecules. 2012, 17,
6305–6316.
55.
Kovač, J.; Šimunović, K.; Wu, Z.;
Klančnik, A.; Bucar, F.; Zhang, Q.; Možina, S.S. Antibiotic resistance
modulation and modes of action of (-)-α-pinene
in Campylobacter Jejuni. PLoS One. 2015, 10, 1–14.
56.
Nóbrega, J.R.; Silva, D. de F.; Andrade
Júnior, F.P. de; Sousa, P.M.S.; Figueiredo, P.T.R. de; Cordeiro, L.V.; Lima, E.
de O. Antifungal action of α-pinene against Candida
spp. isolated from patients with otomycosis and effects of its association with
boric acid. Nat. Prod. Res. 2021, 35, 6190–6193.
57.
Hammer, K.A.; Carson, C.F.; Riley, T.V.
Antifungal activity of the components of Melaleuca
alternifolia (Tea tree) oil. J.
Appl. Microbiol. 2003, 95, 853–860.
58.
Julaeha, E.; Herlina, T.; Nurzaman, M.;
Mayanti, T.; Kurnia, D.; Sari, E.F. The antibacterial effect of β-pinene derived from Citrus aurantifolia peel against oral Streptococcus mutans ATCC 25175. Padja. J. Dent. 2021, 33, 88–93.
59.
Andrade, A.; Rosalen, P.; Freires, I.;
Scotti, L.; Scotti, M.; Aquino, S.; Castro, R. Antifungal activity, mode of
action, docking prediction and anti-biofilm effects of (+)-β-pinene enantiomers against Candida
spp. Curr. Top. Med. Chem. 2018,
18, 2481–2490.
60.
Han, Y.; Sun, Z.; Chen, W. Antimicrobial
susceptibility and antibacterial mechanism of limonene against Listeria monocytogenes. Molecules. 2019, 25, 33.
61.
Vuuren, S.F. van; Viljoen, A.M.
Antimicrobial activity of limonene enantiomers and 1,8-cineole alone and in
combination. Flavour Fragr J. 2007, 22, 540–544.
62.
Dahham, S.S.; Tabana, Y.M.; Iqbal, M.A.;
Ahamed, M.B.K.; Ezzat, M.O.; Majid, A.S.A.; Majid, A.M.S.A. The anticancer,
antioxidant and antimicrobial properties of the sesquiterpene β-caryophyllene from the essential oil
of Aquilaria crassna. Molecules. 2015, 20,
11808–11829.
63.
Qin, R.; Yang, S.; Fu, B.; Chen, Y.; Qi,
Y.; Xu, N.; Wu, Q.; Hua, Q.; Wu, Y.; Liu, Z. Antibacterial activity and
mechanism of the sesquiterpene δ-cadinene
against Listeria monocytogenes and
its application in milk. Food Microbiol. 2023,
1–24.
64.
Pérez-López, A.; Cirio, A.T.;
Rivas-Galindo, V.M.; Aranda, R.S.; de Torres, N.W. Activity against Streptococcus pneumoniae of the
essential oil and δ-cadinene isolated
from Schinus molle fruit. J. Essent. Oil Res. 2011, 23, 25–28.
65.
Salas-Oropeza, J.; Jimenez-Estrada, M.;
Perez-Torres, A.; Castell-Rodriguez, A.E.; Becerril-Millan, R.;
Rodriguez-Monroy, M.A.; Jarquin-Yañez, K.; Canales-Martinez, M.M. Wound healing
activity of α-pinene and α-phellandrene. Molecules. 2021, 26, 2488.
66.
Koyama, S.; Purk, A.; Kaur, M.; Soini,
H.A.; Novotny, M.V.; Davis, K.; Kao, C.C.; Matsunami, H.; Mescher, A.
Beta-caryophyllene enhances wound healing through multiple routes. PLoS One. 2019, 14,
e0216104.
67.
Ueno, H.; Shimada, A.; Suemitsu, S.;
Murakami, S.; Kitamura, N.; Wani, K.; Matsumoto, Y.; Okamoto, M.; Ishihara, T.
Attenuation effects of alpha-pinene inhalation on mice with dizocilpine-induced
psychiatric-like behaviour. Evid.-based Complement. Altern. Med. 2019, e2745453.
68.
Karthikeyan, R.; Kanimozhi, G.; Prasad,
N.R.; Agilan, B.; Ganesan, M.; Srithar, G. Alpha pinene modulates UVA-induced
oxidative stress, DNA damage and apoptosis in human skin epidermal
keratinocytes. Life Sci. 2018, 212, 150–158.
69.
Frum, Y.; Viljoen, A. In vitro 5-lipoxygenase activity of
three indigenous south african aromatic plants used in traditional healing and
the stereospecific activity of limonene in the 5-lipoxygenase. J. Essent. Oil Res. 2006,
18, 831–839.
70.
Keinan, E.; Alt, A.; Amir, G.; Bentur,
L.; Bibi, H.; Shoseyov, D. Natural ozone scavenger prevents asthma in
sensitized rats. Bioorg. Med.
Chem. 2005, 13, 557–562.
71.
Fernandes, E.S.; Passos, G.F.; Medeiros,
R.; da Cunha, F.M.; Ferreira, J.; Campos, M.M.; Pianowski, L.F.; Calixto, J.B.
Anti-inflammatory effects of compounds alpha-humulene and
(−)-trans-caryophyllene isolated from the essential oil of Cordia verbenacea. Eur. J.
Pharmacol. 2007, 569, 228–236.
72.
Francomano, F.; Caruso, A.; Barbarossa,
A.; Fazio, A.; La Torre, C.; Ceramella, J.; Mallamaci, R.; Saturnino, C.;
Iacopetta, D.; Sinicropi, M.S. β-caryophyllene:
a sesquiterpene with countless biological properties. Appl. Sci. 2019, 9, 5420.
73.
Chen, W.; Liu, Y.; Li, M.; Mao, J.;
Zhang, L.; Huang, R.; Jin, X.; Ye, L. Anti-tumor effect of α-pinene on human hepatoma cell lines through inducing G2/M cell
cycle arrest. J. Pharm. Sci. 2015, 127, 332–338.
74.
Zhang, Z.; Guo, S.; Liu, X.; Gao, X.
Synergistic antitumor effect of α-pinene
and β-pinene with paclitaxel against
non-small-cell lung carcinoma (NSCLC). Drug
Res. 2015, 65, 214–218.
75.
Gould, M.N.; Moore, C.J.; Zhang, R.;
Wang, B.; Kennan, W.S.; Haag, J.D. Limonene chemoprevention of mammary
carcinoma induction following direct in
situ transfer of V-Ha-Ras1. Cancer
Res. 1994, 54, 3540–3543.
76.
Sousa, J.M.S. de; Nunes, T.A. de L.;
Rodrigues, R.R.L.; Sousa, J.P.A. de; Val, M. da C.A.; Coelho, F.A. da R.; Santos,
A.L.S.D.; Maciel, N.B.; Souza, V.M.R. de; Machado, Y.A.A.; et al. Cytotoxic and
antileishmanial effects of the monoterpene β-ocimene.
Pharmaceuticals. 2023, 16, 183.
77.
Yap, Y.; Muria-Gonzalez, M.J.; Kong,
B.H.; Stubbs, K.; Tan, C.-S.; Ng, S.; Tan, N.; Solomon, P.; Fung, S.; Chooi,
Y.-H. Heterologous expression of cytotoxic sesquiterpenoids from the medicinal
mushroom Lignosus rhinocerotis in
yeast. Microb. Cell. Factories.
2017, 16, 1–13.
78.
De Oliveira, P.F.; Munari, C.C.;
Nicolella, H.D.; Veneziani, R.C.S.; Tavares, D.C. Manool, a Salvia officinalis diterpene, induces
selective cytotoxicity in cancer cells. Cytotechnol.
2016, 68, 2139–2143.
79.
Jucá, D.M.; Silva, M.T.B. da; Junior,
R.C.P.; Lima, F.J.B. de; Okoba, W.; Lahlou, S.; Oliveira, R.B. de; Santos, A.A.
dos; Magalhães, P.J.C. The essential oil of Eucalyptus
tereticornis and its constituents, α-
and β-pinene, show accelerative
properties on rat gastrointestinal transit. Planta Med. 2011,
77, 57–59.
80.
Yeo, D.; Hwang, S.-J.; Song, Y.-S.; Lee,
H.-J. Humulene inhibits acute gastric mucosal injury by enhancing mucosal
integrity. Antioxidants. 2021, 10, 761.
81.
Govindarajan, M.; Rajeswary, M.; Hoti,
S.L.; Bhattacharyya, A.; Benelli, G. Eugenol, α-pinene and β-caryophyllene from
Plectranthus barbatus essential oil
as eco-friendly larvicides against malaria, dengue and japanese encephalitis
mosquito vectors. Parasitol. Res. 2016,
115, 807–815.
82. Nam, S.Y.; Chung, C.; Seo, J.-H.; Rah, S.-Y.; Kim, H.-M.; Jeong, H.-J. The therapeutic efficacy of α-pinene in an experimental mouse model of allergic rhinitis. Int. Immunopharmacol. 2014, 23, 273–282.
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
Abstract
The present study was carried out to compare
the chemical composition of the leaf essential
oils (EOs) from six Psiadia species
endemic to Madagascar, namely P.
altissima, P. stenophylla, P. hispida,
P. leucophylla, P. lucida and P. salviifolia.
Three of these species (P. altissima,
P. lucida and P. salviifolia) are
traditionally used for treating diarrhea, stomach ache and skin diseases. They are
also known for their antiseptic,
expectorant, hemostatic, carminative and anti-inflammatory properties. The EOs of the six species were obtained by hydrodistillation and
analysed by capillary GC-FID and GC-MS. Their oil yields ranged from 0.04% to 0.80%. 149 components were identified,
accounting for 84.0 to 97.9% of the
total composition. The EOs from the four species P. altissima, P. stenophylla,
P. hispida and P. salviifolia showed
similar chemical composition which was dominated by monoterpene (63.1-78.4%)
and sesquiterpene (5.7-25.7%) hydrocarbons. The major compounds identified in these oils were β-pinene (17.2-46.5%), limonene (10.4-28.5%) and (Z)-β-ocimene
(5.4-7.3%). The two other
species exhibited qualitative and quantitative differences in the chemical
composition of their EOs. The essential
oil (EO) from P. lucida
was found to be rich in sesquiterpene hydrocarbons (46.6%) and oxygenated sesquiterpenes (32.0%). α-cadinol (11.1%), α-muurolene (10.6%) and δ-cadinene (7.5%) were the main components. The P. leucophylla EO was characterized by a prominent content of sesquiterpene hydrocarbons (66.4%). The main compounds
included β-caryophyllene (10.6%), δ-cadinene (8.9%) and (E)-muurola-4(14),5-diene (7.2%).
This was the first report on the chemical composition of the EOs from P. stenophylla, P. hispida and P. leucophylla.
Abstract Keywords
Psiadia, essential oils, chemical composition, Madagascar, GC-MS, GC-FID.
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
Editor-in-Chief
This work is licensed under the
Creative Commons Attribution 4.0
License.(CC BY-NC 4.0).


