Research Article
Terrafe Chafik
Terrafe Chafik
Laboratory of Pharmacology, Neurobiology,
Anthropology and Environment Department of Biology. Faculty of Sciences
Semlalia. University Cadi Ayyad. BP 2390-40080. Marrakech, Morocco.
E-mail: chafik.terrafe@edu.uca.ac.ma
Majda Badaoui
Majda Badaoui
Laboratory of Pharmacology, Neurobiology,
Anthropology and Environment Department of Biology. Faculty of Sciences
Semlalia. University Cadi Ayyad. BP 2390-40080. Marrakech, Morocco.
Ait Laaradia Mehdi
Ait Laaradia Mehdi
Laboratory of Pharmacology, Neurobiology,
Anthropology and Environment Department of Biology. Faculty of Sciences
Semlalia. University Cadi Ayyad. BP 2390-40080. Marrakech, Morocco.
Souad Moubtakir
Souad Moubtakir
Laboratory of Pharmacology, Neurobiology,
Anthropology and Environment Department of Biology. Faculty of Sciences
Semlalia. University Cadi Ayyad. BP 2390-40080. Marrakech, Morocco.
Rachida Aboufatima
Rachida Aboufatima
Laboratory of Genie Biologic, Sultan Moulay Slimane
University, Faculty of Sciences and Techniques, Beni Mellal, Morocco.
Chait Abderrahman
Chait Abderrahman
Corresponding Author
Laboratory of Pharmacology, Neurobiology, Anthropology and Environnement Department of Biology. Faculty of Sciences Semlalia. University Cadi Ayyad. BP 2390-40080. Marrakech, Morocco.
E-mail: chait@uca.ac.ma
Received: 2023-03-10 | Revised:2023-04-04 | Accepted: 2023-04-08 | Published: 2023-05-09
Pages: 176-184
DOI: https://doi.org/10.58985/jeopc.2023.v01i03.22
Abstract
Tribolium
castaneum (Tc) is one of the
principal pests affecting cereals, they provoke considerable quantitative and
qualitative losses of grains. The purpose of this investigation was to compare for the
first time, the antioxidant and insecticidal activities and chemical
composition of aerial parts of Pistacia
lentiscus essential oils (PLEOs). The major components are α-myrcene,
limonene and α-pinene, for leaves, stems and fruits respectively. Concerning
antioxidant activity, the results indicated a high activity of PLEOs with IC 50
of 5, 46±0, 12 mg/ml, 4, 67±0,18 mg/ml and 2, 75±0,12 mg/ml of DPPH assay
compared to the control groups for leaves, stems and fruits respectively. Also,
complementary assays: FRAP and ABTS revealed an important antioxidant capacity
confirming those funded by the DPPH assay. Contact toxicity demonstrated that PLEOs possess strong insecticidal
efficiency against reed flour beetle with LD50 values 0, 77 µL/cm2,
0, 53 µL/cm2 and 0, 49 µL/cm2 for leaves, stems and
fruits respectively. The LT 50 values ranged 42, 80 to 76 hours for
leaves, 5, 5 to 60, 23 hours for stems and 6 to 51 hours for fruits. About
locomotor activity, insects tend to spend more time in the untreated half
arena. However, their instantaneous growth rate was significantly reduced which
is caused by the presence of the essential oil in the arena.
Keywords
Pistacia lentiscus, GC-MS analysis, antioxidant activity,
fumigant and contact toxicity, behavior and locomotors responses.
1.
Introduction
The world population is
growing it will be more than 8,5 billion in 2025 [1], according to economists this is an alarming
situation in the face of the demand that will be increasing for food products.
However, stored foodstuffs are often attacked
and deteriorated by insect pests during the storage period. Tc (Coleoptera,
Tenebrionidae) is one of the species worldwide that causes significant economic
losses for stored products such as cereals [2]. Today,
several synthetic insecticides are ineffective against a wide range of insect
pests. Previous studies have shown that these synthetic products cause problems
in human health and environmental pollution [3].
To this end, the management of this pest is focused on using eco-friendly
products such as essential oils due to their biomolecules as well as biological
activities as antioxidant, antimicrobial, antifungal and insecticidal
activities [4]. Essential oils have been
proposed as alternative products to control stored pests because of their
mechanisms of action, they act on GABAergic receptors
[5], octopamine system [6], inhibition of acetyl cholinesterase [7] and destruction of neurophysiologic
functions in the insect nervous system.
Pistacia
lentiscus (Pl) is a plant
belonging to the Anacardiaceae family that grows on all types of soil and is
widely distributed in the Mediterranean circuit. Pl is currently
considered an interesting plant for its biological effects, which justifies its
extensive use in traditional pharmacopoeia for the treatment of several
diseases in Morocco including digestive [8],
cardiovascular and diabetes illnesses [9].
One of the most remarkable biological effects of Pl is anticancer effect
due to the resin [10-14].
The insecticidal activity of PLEOs against Tc was mentioned
in previous studies [15-16] which is based
solely on the fumigant toxicity. However, the potential effect involved on
locomotor strategies used by these pests to mitigate the effects of these oils.
Locomotor responses and instantaneous development rate were completely
neglected. The objective of our investigation was. Firstly, comparison of the
chemical composition of PLEOs from
aerial parts (stems, fruits and leaves) and antioxidant activity. Secondary,
evaluation of insecticidal activity (contact and fumigant toxicity), locomotor
responses and instantaneous rate of development.
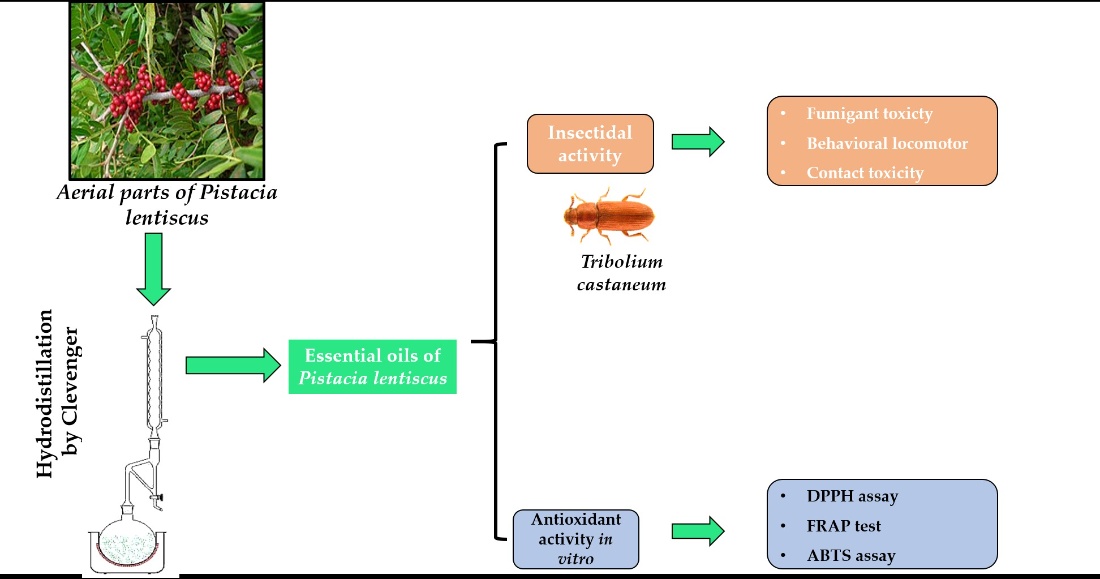
2.
Materials and methods
2.1. Plant material
Aerial parts (fruits, leaves
and stems) of Pistacia lentiscus, were collected in the region of Ourika
(31°23' latitude N / 7° 42 ' longitude W; 35 KM from Marrakech, Morocco). They
were harvested in March 2018. The samples were identified by Professor CHAIT
Abderrahman and stored as a voucher specimen (P 18) in the plant herbarium of
laboratory, department of biology, faculty of sciences Semlalia, Marrakech,
Morocco. The plant was dried in the open air at room temperature until the
weight stabilization for extraction of essential oils.
2.2 Extraction of PLEOs
The
essential oils were obtained from dry material by hydro-distillation, Clevenger
type apparatus. Also, 500 g, 300 g and 500 g of ground plant materials (leaves,
fruits and stems respectively) were added to distilled water. Then, essential
oils were collected manually, using sodium sulfate to remove water. Finally,
the PLEO samples were conserved in a refrigerator at 4°C.
2.3
GC-MS analysis
The analysis protocol of
PLEOs was mentioned in our previous study (Terrafe et al., 2022). Identification of chemical compounds was based on the Adam’s library.
2.4
Antioxidant activity
2.4.1 DPPH activity
The free radical activity of
PLEOs was determined by the stable radical (DPPH), according to similar method
described by Sahin [17]. Briefly,
1,0-10 µL/ml of PLEOs was added to 2 ml of 60 μM methanol solution of DPPH and
incubated at ambient temperature in obscurity. After thirty minutes, the
absorbance was recorded against methanol as a blank at 517 nm. Quercetin and
BHT (butylated hydroxyanisole) were used as positive control. The concentration
of the PLEOs that neutralizes 50![]() of DPPH (IC50) was estimated using
the following formula:
of DPPH (IC50) was estimated using
the following formula:
I
(%) = [(A-B)/A] ×100
Where, A is the absorption of
the control at thirty minutes. B is the absorption of the PLEOs after thirty
minutes.
2.4.2
FRAP assay
The free radical activity of PLEOs
was determined by inhibition of the formation of the Fe (II) -Ferrozine complex
after incubation of the samples with the divalent iron according to the method
described by [18]. The method was based on
the chemical conversion reaction of Fe3+ to Fe2+.
Briefly, the sample and control substance were mixed with phosphate buffer (2,5
ml, 0,2M, ph= 6,6) and potassium ferricyanide [K3Fe (CN) 6] (2.5 ml, 1 %).
Thirty minutes later, trichloroacetic acid was added (2,5 ml of 10 % (w/v)).
The obtained mixture was centrifuged 650 ˣ g for 10 min. Finally, the upper layer was
mixed with 2,5 ml distilled water and 0,5 ml of FeCl3 (ferric chloride 1%). The
absorbance was measured at 700 nm after 15 min time of reaction. Quercitine and
BHT were used as positive control. Three replications were performed to
calculate the mean value of the IC50.
2.4.3 ABTS assay
The ABTS
test was used to assess the free radical scavenging activity of the essential
oil samples. Briefly, 100 µL of PLEOs or water (control) were mixed with 1 mL
of diluted ABTS+ solution, and the absorbance was measured at 734 nm six
minutes later [19].
2.5
Insecticidal activity
2.5.1 Insect cultures
Tc
colonies were housed in the laboratory without any exposure to insecticides.
Sixty insects of both genders are reared on a mixture of wheat flour, wheat
germ and yeast extract (13:6:1 w/w/w) in borosilicate glass jars (16cm
[diameter] × 22 cm [height]). These jars are covered with mosquito netting to
allow insects to breathe. The cultures were kept in a growth room at 26 ± 1°C,
a relative humidity and a photoperiod of 16:8 hours (light: darkness). Only
adults are used for biological tests by contact and fumigation toxicity
bioassays. All tests were carried out under conditions identical to those of
the breeding.
2.5.2 Contact toxicity assay
The insecticidal activity of
PLEOs against Tc adults was measured by contact toxicity assay. Four
doses were prepared for each aerial part (leaves, stems and fruits) by diluting
each time in 1 ml of acetone the respective volumes of 40, 50, 60 and 70 μL of
essential oil. These volumes correspond to doses of 0,62, 0,78, 0,94 and 1,10
μL/cm2 for leaves. Then, four doses 30, 35, 40 and 45 μL of
essential oil. These volumes correspond to doses of 0,47, 0,55, 0,62 and
0,71μL/cm2 for stems. Other four doses were prepared 10, 15, 25 and
25 µL of PLEOs. These doses correspond to 0,15, 0,23, 0,31 and 0,39 for fruits.
Each of solutions prepared was spread uniformly over a 9 cm diameter (i.e.
63,62 cm 2 surface area) filter paper washer (Whatman No. 1) placed
in a glass Petri dish of the same diameter. After fifteen minutes, (solvent was
evaporated) ten unsexed adults freshly collected from their breeding
environment and 7 to 14 days old was added to each Petri dish and these boxes
were immediately closed. Three replicates were performed for each dose.
Mortality was recorded after 2, 4, 6, 8, 24, 48 and 72 hours. The signs of
mortality are: absence of antennal movements and leg. Bio-tests were designed
to determine the lethal dose LD50, LD90 values doses and
the lethal time LT50, LT90 values of exposed insects [19].
2.5.3 Fumigant toxicity assay
Fumigant toxicity was
evaluated using filter paper (Whatman No.1) that was impregnated with different
PLEOs doses. The filter paper was attached to the inside of the small bottles
of a volume of 60 ml, each of them containing 10 individual insects aged
between 1 to 7 days. Repetitions of 3 times were carried out for each dose. The
mortality was noted after 2 h, 4 h, 6 h, 8 h, 12 h, 24 h,48 h and 72 h of
exposure, we consider a dead individual when no movement of the antennal and
leg was observed [19].
2.5.4 Population growth bioassay
The PLEOs were used to evaluate their effect on the
biological development rate of the insect population. The experiment consisted of
exposing the insects to the different doses of the essential oils (60, 120, 250
and 500 μL/ kg) in glass jars (0,8 L) each containing 250 g of the barley grains.
Twenty insects were released in each jar to colonize the grain mass for sixty
days. Control was treated only by acetone under standard conditions mentioned
previously. After this period, the number of living insects and mass of grains were
measured. Four replications were performed for each dose. The instantaneous
growth rate has been calculated according to the following formula: ri = [ln (Nf / Ni)] / ΔT,
where Nf and Ni are the final and initial numbers of living (adult) insects,
respectively, and ΔT is the duration of experience in days [20].
2.5.5 Behavioral and locomotor responses
The behavioral bioassays of
PLEOs against Tc in circular arenas that were either half or fully
treated were evaluated by similar method described by Braga, Haddi, Correa,
Pereira, Guedes [21-25]. Briefly, the filter
was impregnated with 3 mL of PLEOs (1,05 µL of PLEOs/cm2) at the
concentration corresponding to the estimated LC90. The control was
treated with acetone only. The filter papers were placed in Petri dishes (135ˣ20 mm),
after drying for 20 min. The Teflon® was used to coat the inner walls of each
Petri dish to prevent movement of each insect from escaping. Two parameters
were recorded for the fully treated arenas for 10 min such as, number of stops
and walked distance (cm) in the arena. In order to calculate the proportion of
time spent in each compartment of the arena (completely or half-Treated),
bioassays were carried out with individual insects adults, twenty insects were
used and for each replicate, the filter paper was replaced and the side which
the insects were released was randomly selected.
2.6
Statistical analysis
The
PROC PROBIT procedure was used to estimate dose-mortality curves in probit
analysis. The finding of the survival bioassays was subjected to a survival
analysis with Sigma Plot software, which used the Kaplan-Meier estimators
(Log-rank method). A paired student’s t-test (p<0.05) was used to compare
pairwise differences in walking behavior in the half treated arenas.
3.
Results and discussion
3.1. Chemical composition
The clear and yellowish essential oils obtained by hydrodistillation of
aerial parts of PL yielded 0, 61 %, 0, 45 % and 0, 78 % respectively for leaves, stems and fruits. PLEOs
Analysis by GC-MS reveal 22 compounds for all parts representing
a total 98,41 %, 98,35 % and 98,05 %
for leaves, fruits and stems respectively. The results are shown
in Table 1. Regarding, yield is significantly lower than those obtained by
Congiu [28] in Italy and Zrira et al [29] in Morocco. The yield of essential
oil seems to depend on the nature of the parts of plants used, method of
extraction, period of harvest, altitude and climatic conditions.
Table 1. Chemical composition of
PLEOs from aerial parts
|
Peak n° |
RT (min) |
Compounds |
PLSEO (%) |
PLLEO (%) |
PLFEO (%) |
|
1 |
1,86 |
Tricyclene |
1,45 |
1,14 |
1,12 |
|
2 |
2,32 |
α-thujene |
3,11 |
2,06 |
2,42 |
|
3 |
2,43 |
α-pinene |
5,16 |
20,75 |
24,12 |
|
4 |
2,56 |
Camphene |
3,57 |
2,54 |
5,31 |
|
5 |
2,78 |
α-phellandrene |
4,21 |
3,23 |
0,66 |
|
6 |
4,17 |
α-myrcene |
10,26 |
36,18 |
13,89 |
|
7 |
6,83 |
o cymene |
2,73 |
2,46 |
4,89 |
|
8 |
8,12 |
Terpinene |
5,13 |
3,45 |
0,28 |
|
9 |
8,92 |
Limonene |
31,54 |
3,15 |
0,96 |
|
10 |
9,34 |
Verbenol |
6,23 |
3,07 |
6,25 |
|
11 |
10,84 |
Borneol |
0,45 |
2,15 |
2,92 |
|
12 |
11,15 |
Terpinen-4-ol |
1,19 |
4,11 |
2,14 |
|
13 |
13,3 |
α-terpineol |
1,63 |
0,78 |
3,41 |
|
14 |
14,46 |
Terpineol |
5,36 |
6,49 |
16,94 |
|
15 |
16,52 |
Bornyl acetate |
4,65 |
1,87 |
1,54 |
|
16 |
17,44 |
Caryophyllene |
3,28 |
0,17 |
1,64 |
|
17 |
18,85 |
ç muurolene |
0,65 |
1,23 |
4,11 |
|
18 |
19,05 |
Germacrene D |
2,81 |
0,45 |
0,15 |
|
19 |
21,68 |
α-muurolene |
0,89 |
0,34 |
1,36 |
|
20 |
22,84 |
α-cadinol |
1,56 |
0,47 |
0,24 |
|
21 |
24,37 |
p-camphrene |
0,14 |
1,57 |
1,19 |
|
22 |
26,69 |
Cadinene |
2,35 |
0,75 |
2,51 |
|
Total (%) |
98,35 |
98,41 |
98,05 |
||
|
Oil yeld (%, w/w) |
0,62 |
0,45 |
0,78 |
||
|
Grouped compounds (%) |
|||||
|
Monoterpene hydrocarbons |
68,25 |
74,96 |
53,65 |
||
|
Oxygenated monoterpenes |
19,16 |
19,98 |
36,14 |
||
|
Sesquiterpene hydrocarbos |
9,38 |
2,94 |
8,02 |
||
|
|
Oxygenated sesquiterpenes |
|
1,56 |
0,53 |
0,24 |
|
50 m × 0.25 mm × 1.0 mm; Nd= non determined (p<0.05%).
LEO= leaves essential oil, EOF= fruits essential oil, EOS= stems essential
oil.
|
|||||
Our
results are consistent with previous studies in terms of quality but not quantitatively.
Concerning fruits oil, the main components are α-myrcene (13,89 %), α-pinene
(24,12 %), 4-terpineol (4,37 %), verbenol (6,25 %), o cymene (2,28%). On the
other hand, the major components present in the stems oil are: limonene (31,54
%), α-myrcene (12,07 %). Our results differ slightly with those reported by
Amhamdi et al [26] on
the same species from the east of Morocco. However, obtained results are in
agreement with those reported by Zrira et al [29] from three regions of Morocco, in this study the major oil
components are terpinene-4-ol (43,80%), α-pinene (38,50%), β-myrcene
(11.50%), and limonene (9,8%).
Nevertheless, they are in agreement with those reported by Bouyahya et al [30] because the study was conducted on the same
aerial parts. These findings are in accordance
with those reported by Amhamdi et al.,
Castola et al [26-27]. The comparison of essential oil composition from leaves, stems, fruits of Pl was
not previously reported and therefore our study can be considered as the first
report on the PLEOs.
3.2.
Antioxidant activity
To evaluate the antioxidant properties of PLEOs, three assays were used:
FRAP, ABTS and DPPH assays. As listed in Table 2, in comparison to stems and then
leaves. In fact, PLFEO IC50 values, recorded by DPPH, FRAP and ABTS
tests were 2,75, 6,75 and 5,75 mg/mL respectively. These results suggest a high
antioxidant activity of the PLEOs since the IC50 values obtained are
comparable to those of the standard antioxidants quercitine and BHT. Meanwhile,
PLSEO presented IC50 equivalent to 4,67, 6,24 and 9,16 mg/mL by
DPPH, FRAP and ABTS assays. Whereas, PLLEO exhibited a significant antioxidant
activity with IC50 equal to 5,46, 7,59 and 8,16 mg/mL by DPPH, FRAP
and ABTS respectively.
Table 2. The antioxidant capacity of PLEOs expressed in value of IC50
(mg/mL).
|
|
Essential oils IC50 mg/mL |
Standard antioxydants IC50µg/mL |
|||
|
|
Leaves |
Stems |
Fruits |
Quercetine |
BHT |
|
DPPH |
5,46±0,12 |
4,67±0,18* |
2,75±0,12 |
2,10±0,06 |
4,25±0,09 |
|
FRAP |
7,59±0,07 |
6,24±0.05 |
6,75±0,12 |
3,65±0,01 |
3,65±0,01 |
|
ABTS |
8.16±1.13 |
9,16±0,03 |
5,75±0,12 |
2.57±0.25 |
3.34±0.05 |
*Values represent means
standard deviations for three replicates
Recently, the investigation of a novel antioxidant biomolecules is a
crucial research topic due to their implication in treatment of several
diseases. Few studies were conducted on the antioxidant property of Pistacia
lentiscus essential oils. Our outcomes are in accordance with those described
by many studies [30-32]. Another study [33] reported a low
antioxidant activity of Pl essential oil of leaves, also [34] mentioned that
essential oil of Pl var Chia did not possess antiradical activity
evaluated by the DPPH test. On the other hand, Pistacia atlantica
essential oil possesses a low
capacity of neutralizing free radicals and interesting ferric reducing power [35]. The essential oils contain several classes
of terpenes such as; oxygenated monoterpenes, diterpenes, triterpenes and
sesquiterpenes. Monoterpenes provide redox properties to essential oils and
consequently antioxidant capacities. This activity could be due according to
Aissi et al [31]
to the presence of several bioactive compounds as p cymene and
muurolene for DPPH assay, muurolol and cadinol for FRAP assay
3.3 Insecticidal
activity
3.3.1 Fumigant and contact toxicity bioassays
The insecticidal effect of PLEOs was evaluated by the fumigant
and contact toxicity assays against adults of Triobolium castaneum. As
mentioned in Tables 3 and 4, the LD50 values were 0,77 µL/cm2,
0,53 µL/cm2 and 0,49 µL/cm2 respectively for leaves,
stems and fruits. The highest insecticidal activity against this stored pest
was recorded by PLFEO, PLSEO and PLLEO. Concerning the fumigant toxicity, LD50
values were 176 µL/L air, 107,83 µL/L and 89,16 µL/L respectively for leaves,
stems and fruits. On the other hand, LT50 reduced as oil
concentrations increased which is reported in Table 4 and 5. The lowest LT50
39,20 h, 42,80 h perfectly correspond to the highest concentrations 1.10 μL/ cm2
and 333.33 μL/ L in contact and fumigant toxicity tests respectively.
Table 3. LD50 and LD90 values for contact toxicity and fumigant
toxicity of PLEOs against adults of Tc
|
Essential
oils |
LD50 (95%CI) |
LD90
(95%CI) |
Slope±S.E |
X2 |
Df |
|
|
Contact Toxicity µL/cm2 |
Leaves |
0,77(0,66-0,87) |
1,05(0,91-1,58) |
2,43±0,21 |
1,88 |
2 |
|
Stems |
0,53(0,45-0,58) |
0,67(0,61-0,85) |
3,26±0,01 |
1,32 |
2 |
|
|
Fruits |
0,49(0,18-0,61) |
0,84(0,69-1,37) |
1,25±0,10 |
0,77 |
2 |
|
|
Fumigant Toxicity µL/cm2 |
Leaves |
176(160,34-191,03) |
466,24(346,19-588,2) |
3,14±0,35 |
2,59 |
2 |
|
Stems |
107,83(74,5-136,66) |
179,5(147,66-267,5) |
2,16±0,27 |
1,1 |
2 |
|
|
Fruits |
89,16(48,83-118,16) |
167,16(133,33-284,33) |
2.23±0,10 |
0.10 |
2 |
|
|
X²: Chi square test; CI: Confidence intervals at 95%; Df:
Degree of freedom |
||||||
Table 4.
LT50
and LT90 values of PLEOs
against Tc in contact toxicity.
|
|
Oil concentrations |
LT50 (CI 95%) |
LT90 (CI 95%) |
Slope±S.E |
X2 |
Df |
|
|
Contact toxicity µL/cm2 |
|||||
|
Leaves |
0,62 |
76.00
(37,58-114,41) |
136,80
(114,41-160,45) |
3,12±0,28 |
4,26 |
3 |
|
0,72 |
69,20
(32,37-106,02) |
124,50
(106,02- 141,52) |
2,34±1,32 |
2,8 |
3 |
|
|
0,94 |
48,08
(18,34-77,81) |
86,54 (77,81-96,35) |
2,57±1,40 |
3,29 |
3 |
|
|
1,11 |
42,80
(37,58-72,54) |
77,04
(72,54- 85,34) |
1,89±1,78 |
2,75 |
3 |
|
|
Stems |
0,47 |
60,23(36,48-83,52) |
108,00
(42,5-132) |
3,12±0,28 |
3,15 |
3 |
|
0,55 |
18,15(13,76-22,23) |
32,40
(12,43-39,15) |
2,34±1,32 |
2,16 |
3 |
|
|
0,62 |
13,42(11,21-15,64) |
24,15(8,11-32,11) |
2,57±1,40 |
4,13 |
3 |
|
|
0,71 |
5,50(1,10-3,32) |
9,90
(3,4-12,9) |
1,89±1,78 |
3,65 |
3 |
|
|
Fruits |
0,15 |
51(24,97-77,02) |
91,80
(73.5-110) |
3,12±0,28 |
1,26 |
3 |
|
0,23 |
16,66
(12,33-21,23) |
29,88(22.43-37,35) |
2,34±1,32 |
3,29 |
3 |
|
|
0,31 |
13,42(11,21-15,64) |
24,15(12,43-39,15) |
2,57±1,40 |
4,26 |
3 |
|
|
0,39 |
6,00
(3,67-8,23) |
10,81(4,423-13,25) |
1,89±1,78 |
2,63 |
3 |
|
|
X²: Chi square test; CI: Confidence
intervals at 95%; Df: Degree of freedom. Time mortality values were
obtained using Kaplan-Meier estimators (Log-rank method). |
||||||
Table 5.
LT50 and LT90 values of PLEOs against Tc in fumigant toxicity
|
Oil concentrations µL/L air |
LT50
(CI 95%) |
LT90 (CI 95%) |
SLOPE |
X2 |
Df |
||
|
Fumigant toxicity µL/ L air |
|||||||
|
Leaves |
83,33 |
96,60(67,58-125)
|
173,8(125-221,4) |
3,43±2,1 |
4,26 |
3 |
|
|
166,66 |
85,20(52,37-118,12) |
153,3(118,1-189,2) |
2,24±0,5 |
2,83 |
3 |
||
|
250 |
53,60(19,34-87,81) |
96,4(87,81-106,12) |
2,50±1,3 |
3,29 |
3 |
||
|
333,33 |
46,59(15,52-77,27) |
83,8(77,27-143,02) |
2,47±0,4 |
6,75 |
3 |
||
|
Stems |
50 |
61(56,13-65,86) |
109,8(43,4-132,80) |
5,53±1,8 |
5,62 |
3 |
|
|
75 |
51,88(46,31-57,46) |
93,33(75,32-111.23) |
3,36±0,3 |
3,45 |
3 |
||
|
100 |
37,28(30,49-44,08) |
67,14(29,65-75.12) |
2,35±0,0 |
2,11 |
3 |
||
|
125 |
13,65(9,34-17,95) |
24,57(9,21-31,12) |
4,49±1,3 |
4,32 |
3 |
||
|
Fruits |
25 |
73(71,04-74,96) |
131,4(106,12-157,1) |
3,86±1,2 |
3,84 |
3 |
|
|
50 |
54,57(51,06-58,07) |
98,22(69,63-126.83) |
4,30±2,6 |
5,69 |
3 |
||
|
75 |
39,74(35,89-43,60) |
71,53(69,21-73,72) |
2,44±0,4 |
1,79 |
3 |
||
|
100 |
14,66(11,36-17,96) |
29,38(21,19-37.50) |
4,35±1,7 |
4,58 |
3 |
||
|
X²: Chi
square test; Df: Degree of freedom; CI: Confidence intervals at 95%, Time mortality values
were obtained using Kaplan-Meier estimators (Log-rank method). |
|||||||
The obtained outcomes demonstrate
that PLEO exerts a significant insecticidal effect. These findings are in
accordance with previous study [15]. Few
studies [2,15,16], demonstrated the
insecticidal effect of Pl against Tc. However, in other studies [36-37] Tc is a victim model for a large
spectrum of several essential oils such as Artemisia vulgaris and Artemisia
tridentate 36.
In addition, Ebadollahi et al [37] reported
an important insecticidal effect of Lavandula stoechas essential oil and
suggested that it may be used as bio-insecticide against red flour beetle.
Furthermore, PLEO possesses an extensive insecticidal effect when combined to
some essential oils especially, Mentha microphylla and Myrtus
communis against Culex pipiens. The fumigant and contact toxicity
could be explained according to Regnault et al
[38] to oxygenated monoterpenes (e.g.
terpineol, linalool) which induce mortality by inhibition of
acetylcholine-esterase.
3.3.2. Behavioral
locomotor responses and population growth bioassay
The locomotor behavior of insects on the arenas half treated or untreated by essential oils is shown in Figure 1. The results indicate that insects spending more time in the untreated half of arena. Statically, there is a significant difference between the untreated and treated arena in stems LD50 (F=1,25; p<0,01; t-test), as well as between untreated and treated halves of arena for LD50 of fruits (F=2, 65; p<0,001; t-test). However, no significant difference was observed between the LD50 of essential oil from leaves.
Figure
1.
Proportion of time spent by Tc in the untreated and treated half arena
with PLEOs. The mean of 20
repetitions is shown by each histogram bar. The points show significant
differences between the arena halves that was treated with PLEOs and those
untreated (paired t-test p<0, 05).
In this investigation, we have
clearly demonstrated that the essential oils have a neurotoxic effect
immediately after their application, which is manifested by the appearance of
certain symptoms as paralysis, hyperactivity and trembling. These outcomes are
in agreement with those reported by Zhu et al
[39]. The physiological effect of PLEOs,
could be related to the presence of monoterpenoids (terpineol, myrcene, and
D-limonene) acting by disrupting the aminergic [40],
and GABAergic [41] transmission as well as
their inhibition on acetylcholine esterase in the insect nervous system is one
of their possible mechanisms as demonstrated in previous studies [40-41]. These results suggest that PLEOs have effective insecticide
potential against Tc.
The second figure represents the effect of PLEOs on the instantaneous rate of development. The significant differences were noted between the oil doses (F=98,21; p<0,001) and oil types (F=104,51; p<0,001), the instantaneous rate of development declines by increasing concentration. However, the insects treated by essential oil of leaves were less affected, while the other essential oils (stems and fruits) affects the insects considerably. All of the adjustment parameters for the curves presented in Figure 2 are illustrated in Table 6.
Figure
2. Instantaneous
rate of development of Tc exposed to
different essential oils doses of Pl.
Table 6.
Regression
analysis of the curves presented in Figure 2
|
Variable |
Oil |
Model |
Estimated parametrs±S.E |
F |
p |
R2 |
|
|
|
|
|
a |
b |
|||
|
ri |
Leaves |
y = a·exp (−bx) |
0,04±0,001 |
0,006±0,005 |
44,26 |
<0,001 |
0,98 |
|
Stems |
y = a·exp (−bx) |
0,02±0,008 |
0,009±0,001 |
20,15 |
<0,001 |
0,97 |
|
|
Fruits |
y = a·exp (−bx) |
0,05±0,003 |
0,004±0,006 |
32,15 |
<0,001 |
0,99 |
|
4.
Conclusions
The present study
demonstrates that PLEOs have quantitative differences in chemical compounds
between leaves, fruits and stems. The outcomes of this investigation revealed
several volatile compounds in PLEO that were very effective against Tc.
Therefore, the fruits exhibited a higher insecticidal activity followed by
stems then leaves. Regarding the antioxidant potential, the PLEOs possess an
interesting antioxidant capacity. This was validated by DPPH, FRAP and ABTS
assays. As a conclusion, PLEOs could be
used as a powerful antioxidant and eco-friendly solution to control Tc
in stored grains.
Abbreviations
PLEOs: Pistacia lentiscus essential oils
DPPH: Diphenyl-1-picrylhydrazyl
FRAP: ferric reducing ability
power
LD50: Lethal dose
that kills 50%
LD90: Lethal dose
that kills 90%
LT50: Time
required killing 50
LT90: Time
required killing 90
PLFEO: Pistacia lentiscus fruits essential oil
PLSEO: Pistacia lentiscus stems essential oil
PLLEO: Pistacia lentiscus leaves essential oil
Pl:
Pistacia lentiscus
Tc: Tribolium castaneum
Authors’ contributions
Methodology, writing original
draft preparation, C.T.; Conceptualization writing-review, M.B.; Essential oils
analysis and compounds identification, S.M.; Data analysis and acquisition, M.A.;
Validation, writing review & editing, R.A.; Methodology, Validation,
Writing original draft preparation & editing, A.C.
Acknowledgements
A great gratitude for Mr
Abderrazak Regragi, a specialist in animal laboratory management, for his
immense helpful for the authors of this study.
Funding
Self-funding.
Conflicts of interest
There
are no conflicts of interest declared by the authors.
References
1. Babu,
R.M.; Sajeena, A.; Seetharaman, K.; Reddy, M.S. Advances in genetically
engineered (transgenic) plants in pest management—an over view. J. Crop Prot.
2003, 22, 1071–1086.
2. Upadhyay,
N.; Dwivedy, A K.; Kumar, M.; Prakash, B.; Dubey, N.K. Essential oils as
eco-friendly alternatives to synthetic pesticides for the control of Tribolium castaneum (Herbst)
(Coleoptera: Tenebrionidae). J. Essent. Oil-Bear. Plants. 2018, 21, 282–297.
3. Kumar,
P.; Mishra, S.; Malik, A.; Satya, S. Repellent, larvicidal and pupicidal
properties of essential oils and their formulations against the housefly, Musca
domestica. Med. Vet. Entomol. 2011, 25, 302–310.
4. Bakkali,
F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological effects of essential
oils–a review. Food Chem. Toxicol. 2008, 46, 446–475.
5. Priestley,
C.M.; Williamson, E.M.; Wafford, K.A.; Sattelle, D.B. Thymol, a constituent of thyme
essential oil, is a positive allosteric modulator of human GABAA receptors and
a homo‐oligomeric GABA receptor from Drosophila melanogaster. Br. J. Pharmacol.
2003, 140, 1363–1372.
6. Enan, E.E.
Molecular and pharmacological analysis of an octopamine receptor from American
cockroach and fruit fly in response to plant essential oils. Arch. Insect
Biochem. 2005, 59, 161–171.
7. López,
M.D.; Pascual-Villalobos, M.J. Mode of inhibition of acetylcholine-esterase by
monoterpenoids and implications for pest control. Ind. Crops Prod. 2010, 31, 284–288.
8. Bouyahya,
A.; Abrini, J.; Et-Touys, A.; Bakri, Y.; Dakka, N. Indigenous knowledge of the
use of medicinal plants in the North-West of Morocco and their biological
activities. Eur. J. Integr. Med. 2017, 13, 9–25.
9. Jamila,
F.; Mostafa, E. Ethnobotanical survey of medicinal plants used by people in
Oriental Morocco to manage various ailments. J. Ethnopharmacol. 2014, 154, 76–87.
10. Bozorgi,
M.; Memariani, Z.; Mobli, M.; Salehi Surmaghi, M.H.; Shams-Ardekani, M.R.;
Rahimi, R. Five Pistacia species (P.
vera, P. atlantica, P. terebinthus, P. khinjuk, and P. lentiscus): a review
of their traditional uses, phytochemistry, and pharmacology. Sci. World J. 2013.
11. Maxia,
A.; Sanna, C.; Frau, M.A.; Piras, A.; Karchuli, M.S.; Kasture, V.
Anti-inflammatory activity of Pistacia
lentiscus essential oil: involvement of IL-6 and TNF-α. Nat. Prod. Commun. 2011, 6(10), 1934578X1100601033.
12. Giaginis,
C.; Theocharis, S. Current evidence on the anticancer potential of Chios mastic
gum. Nutr Cancer. 2011, 63, 1174–1184.
13. Mharti,
F.Z.; Lyoussi, B.; Abdellaoui, A. Antibacterial activity of the essential oils
of Pistacia lentiscus used in
Moroccan folkloric medicine. Nat. Prod. Commun. 2011, 6, 1934578X1100601024.
14. Catalani,
S.; Palma, F.; Battistelli, S.; Benedetti, S. Oxidative stress and apoptosis
induction in human thyroid carcinoma cells exposed to the essential oil from Pistacia lentiscus aerial parts. PloS
one, 2017, 12, e0172138.
15. Bachrouch,
O.; Jemâa, J.M.B.; Chaieb, I.; Talou, T.; Marzouk, B.; Abderraba, M.
Insecticidal activity of Pistacia lentiscus essential oil on Tribolium
castaneum as alternative to chemical control in storage. Tunis. J. Plant Prot. 2010, 5, 63–70.
16. Bougherra,
H.H.; Bedini, S.; Flamini, G.; Cosci, F.; Belhamel, K.; Conti, B. Pistacia
lentiscus essential oil has repellent effect against three major insect pests
of pasta. Ind Crops Prod. 2015, 63, 249-255.
17. Şahin,
F.; Güllüce, M.; Daferera, D.; Sökmen, A.; Sökmen, M.; Polissiou, M.; Özer, H.
Biological activities of the essential oils and methanol extract of Origanum vulgare ssp. vulgare in the Eastern Anatolia region
of Turkey. Food control. 2004, 15(7), 549-557.
18. Oyaizu,
M. Studies on products of browning reaction anti-oxidative activities of
products of browning reaction prepared from glucosamine. Jpn. J. Nutr. Diet. 1986, 44, 307–315.
19. Kasrati,
A.; Alaoui Jamali, C.; Bekkouche, K.; Wohlmuth, H.; Leach, D.; Abbad, A.
Comparative evaluation of antioxidant and insecticidal properties of essential
oils from five Moroccan aromatic herbs. J. Food Sci. Technol. 2015, 52, 2312-2319.
20. Walthall,
W.K.; Stark, J.D. Comparison of two population‐level ecotoxicological
endpoints: The intrinsic (rm) and instantaneous (ri) rates of increase.
Environ. Toxicol. Chem. 1997, 16, 1068–1073.
21. Braga,
L.S.; Correa, A.S.; Pereira, E.J.G.; Guedes, R.N.C. Face or flee? Fenitrothion
resistance and behavioral response in populations of the maize weevil,
Sitophilus zeamais. J. Stored Prod. Res. 2011, 47, 161–167.
22. Haddi,
K.; Mendonça, L.P.; dos Santos, M.F.; Guedes, R.N.C.; Oliveira, E.E. Metabolic
and behavioral mechanisms of indoxacarb resistance in Sitophilus zeamais (Coleoptera: Curculionidae). J. Econ. Entomol.
2015, 108, 362–369.
23. Corrêa,
A.S.; Pereira, E.J.G.; Cordeiro, E.M.G.; Braga, L.S.; Guedes, R.N.C.
Insecticide resistance, mixture potentiation and fitness in populations of the
maize weevil (Sitophilus zeamais). J.
Crop Prot. 2011, 30, 1655–1666.
24. Pereira,
C.J.; Pereira, E.J.G.; Cordeiro, E.M.G.; Della Lucia, T.M.C.; Tótola, M.R.;
Guedes, R.N.C. Organophosphate resistance in the maize weevil Sitophilus zeamais: magnitude and
behavior. J. Crop Prot. 2009, 28(2), 168-173.
25. Guedes,
N.M.P.; Guedes, R.N.C.; Ferreira, G.H.; Silva, L.B. Flight take-off and walking
behavior of insecticide-susceptible and–resistant strains oil of Sitophilus zeamais exposed to deltamethrin.
Bull. Entomol. Res. 2009, 99, 393–400.
26. Amhamdi,
H.; Aouinti, F.; Wathelet, J.P.; Elbachiri, A. Chemical composition of the
essential of Pistacia lentiscus L.
from Eastern Morocco. Rec. Nat. Prod. 2009, 3(2).
27. Castola,
V.; Bighelli, A.; Casanova, J. Intraspecific chemical variability of the
essential oil of Pistacia lentiscus
L. from Corsica. Biochem. Syst. Ecol. 2000, 28, 79–88.
28. Congiu,
R.; Falconieri, D.; Marongiu, B.; Piras, A.; Porcedda, S. Extraction and
isolation of Pistacia lentiscus L.
essential oil by supercritical CO2. Flavour Fragr. J.
2002, 17, 239–244.
29. Zrira,
S.; Elamrani, A.; Benjilali, B. Chemical composition of the essential oil of Pistacia lentiscus L. from Morocco a
seasonal variation. Flavour Fragr. J. 2003, 18, 475–480.
30. Bouyahya,
A.; Assemian, I.C.C.; Mouzount, H.; Bourais, I.; Et-Touys, A.; Fellah, H.;
Bakri, Y. Could volatile compounds from leaves and fruits of Pistacia lentiscus constitute a novel
source of anticancer, antioxidant, antiparasitic and antibacterial drugs? Ind
Crops. Prod. 2019, 128, 62-69.
31. Aissi,
O.; Boussaid, M.; Messaoud, C. Essential oil composition in natural populations
of Pistacia lentiscus L. From
Tunisia: Effect of ecological factors and incidence on antioxidant and anti-acetylcholine-esterase
activities. Ind. Crops Prod. 2016, 91, 56–65.
32. Yosr, Z.;
Imen, B.H.Y.; Rym, J.; Chokri, M.; Mohamed, B. Sex-related differences in
essential oil composition, phenol contents and antioxidant activity of aerial
parts in Pistacia lentiscus L. during
seasons. Ind. Crops Prod. 2018, 121, 151–159.
33. Barra,
A.; Coroneo, V.; Dessi, S.; Cabras, P.; Angioni, A. Characterization of the
volatile constituents in the essential oil of Pistacia lentiscus L. from different origins and its antifungal and
antioxidant activity. J. Agric. Food Chem. 2007, 55, 7093–7098.
34. Bampouli,
A.; Kyriakopoulou, K.; Papaefstathiou, G.; Louli, V.; Krokida, M.; Magoulas, K.
Comparison of different extraction methods of Pistacia lentiscus var. chia
leaves: Yield, antioxidant activity and essential oil chemical composition. J.
Appl. Res. Med. Arom. Plants. 2014, 1(3), 81-91.
35. Gourine,
N.; Yousfi, M.; Bombarda, I.; Nadjemi, B.; Stocker, P.; Gaydou, E. M.
Antioxidant activities and chemical composition of essential oil of Pistacia atlantica from Algeria. Ind.
Crops Prod. 2010, 31(2),
203-208.
36. Wang, D.;
Collins, P. J.; Gao, X. Optimising
indoor phosphine fumigation of paddy rice bag-stacks under sheeting for control
of resistant insects. J. Stored Prod. Res. 2006, 42, 207–217.
37. Ebadollahi,
A.; Safaralizadeh, M.; Pourmirza, A. Fumigant toxicity of Lavandula stoechas L. oil against three insect pests attacking
stored products. J. Plant Prot. Res. 2010, 50(1).
38. Regnault-Roger,
C.; Hamraoui, A. Fumigant toxic activity and reproductive inhibition induced by
monoterpenes on Acanthoscelides obtectus
(Say) (Coleoptera), a bruchid of kidney bean (Phaseolus vulgaris L.). J. Stored Prod. Res. 1995, 31, 291–299.
39. Zhu, B.C.R.;
Henderson, G.; Yu, Y.; Laine, R.A. Toxicity
and repellency of patchouli oil and patchouli alcohol against Formosan
subterranean termites Coptotermes
formosanus Shiraki (Isoptera: Rhinotermitidae). J. Agric. Food Chem. 2003, 51, 4585–4588.
40. Kostyukovsky,
M.; Rafaeli, A.; Gileadi, C.; Demchenko, N.; Shaaya, E. Activation of
octopaminergic receptors by essential oil constituents isolated from aromatic
plants: possible mode of action against insect pests. Pest Manag. Sci. 2002, 58, 1101–1106.
41. Tong, F.; Coats, J.R. Quantitative structure–activity
relationships of monoterpenoid binding activities to the housefly GABA
receptor. Pest Manag. Sci. 2012, 68, 1122–1129.
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
Abstract
Tribolium
castaneum (Tc) is one of the
principal pests affecting cereals, they provoke considerable quantitative and
qualitative losses of grains. The purpose of this investigation was to compare for the
first time, the antioxidant and insecticidal activities and chemical
composition of aerial parts of Pistacia
lentiscus essential oils (PLEOs). The major components are α-myrcene,
limonene and α-pinene, for leaves, stems and fruits respectively. Concerning
antioxidant activity, the results indicated a high activity of PLEOs with IC 50
of 5, 46±0, 12 mg/ml, 4, 67±0,18 mg/ml and 2, 75±0,12 mg/ml of DPPH assay
compared to the control groups for leaves, stems and fruits respectively. Also,
complementary assays: FRAP and ABTS revealed an important antioxidant capacity
confirming those funded by the DPPH assay. Contact toxicity demonstrated that PLEOs possess strong insecticidal
efficiency against reed flour beetle with LD50 values 0, 77 µL/cm2,
0, 53 µL/cm2 and 0, 49 µL/cm2 for leaves, stems and
fruits respectively. The LT 50 values ranged 42, 80 to 76 hours for
leaves, 5, 5 to 60, 23 hours for stems and 6 to 51 hours for fruits. About
locomotor activity, insects tend to spend more time in the untreated half
arena. However, their instantaneous growth rate was significantly reduced which
is caused by the presence of the essential oil in the arena.
Abstract Keywords
Pistacia lentiscus, GC-MS analysis, antioxidant activity,
fumigant and contact toxicity, behavior and locomotors responses.
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
Editor-in-Chief
This work is licensed under the
Creative Commons Attribution 4.0
License.(CC BY-NC 4.0).


