Short Communication
MaKenna C. Johnson
MaKenna C. Johnson
D. Gary Young Research Institute,
Lehi, UT 84043, USA.
Hsueh-Kung Lin
Hsueh-Kung Lin
D. Gary Young Research Institute, Lehi, UT 84043, USA.
Christopher R. Bowerbank
Christopher R. Bowerbank
D. Gary Young Research Institute, Lehi, UT 84043, USA.
Tyler M. Wilson*
Tyler M. Wilson*
Corresponding
Author
D. Gary Young Research Institute, Lehi, UT 84043, USA.
E-mail: tywilson@youngliving.com;
Tel.: +1-801-669-4501
Received: 2026-03-20 | Revised:2026-04-11 | Accepted: 2026-04-14 | Published: 2026-04-17
Pages: 42-50
DOI: https://doi.org/10.58985/jeopc.2026.v04i01.80
Abstract
Turmeric (Curcuma longa) and
black pepper (Piper nigrum) have a long history of cultivation and economic
value. Both plants have been celebrated for their culinary and medicinal
properties. The current study investigated the use of a novel patented
extraction technique (DeepSpectra®) that employs turmeric essential oil (EO) as
a natural solvent for extracting medicinally important non-volatile compounds
from turmeric rhizomes (curcuminoids) and black pepper berries (piperine).
GC/MS and UPLC-PDA were employed to evaluate the volatile and non-volatile
characteristics of the resulting extracts, respectively. The volatile profile
was largely composed of γ-curcumene (8.6%), α-zingiberene (5.3%), AR-turmerone
(42.1%), and α-turmerone (8.3%). The non-volatile profile contained curcumin
(5.30 mg/mL), demethoxycurcumin (2.94 mg/mL), bisdemethoxycurcumin (3.59
mg/mL), and piperine (1.56 mg/mL). These findings suggest that turmeric EO is a
reliable, natural solvent for extracting non-volatile compounds from raw plant
materials.
Keywords
Black pepper, curcuminoids, DeepSpectra®, gas chromatography, liquid chromatography, mass spectrometry, piperine, turmeric.
1. IntroductionCurcuma longa L.
(turmeric) is a plant species belonging to the Zingiberaceae family [1]. Turmeric has a long history of cultivation,
largely for the harvest of aromatic rhizomes for medicinal, culinary, and
textile (dye) uses [2]. Modern research
has demonstrated that three primary curcuminoids (curcumin, demethoxycurcumin,
and bisdemethoxycurcumin) are responsible for the medicinal properties of rhizomes [3]. In recent years, considerable research has
been conducted on the distillation of turmeric rhizomes for the production of
EO. Studies have shown that turmeric EO demonstrates antioxidant,
antimicrobial, anti-inflammatory, antinociceptive, and herbicidal
properties [4-7]. The EO of turmeric
(specifically C. longa) is largely composed
of sesquiterpenoids [3], with
prominent compounds being AR-turmerone, α-turmerone, and β-turmerone [5-9].
Piper nigrum L.
(black pepper) is a plant species in the Piperaceae family [10] with historical importance in commerce,
particularly in the spice trade [2].
Significant research has been conducted on the therapeutic value of the
non-volatile alkaloid piperine, which is present in black pepper berries. Both
piperine and black pepper EO have been attributed with anti-inflammatory,
antimicrobial, antioxidant, and analgesic properties [11-16]. Black pepper EO is largely composed of (E)-caryophyllene,
α-pinene, β-pinene, δ-3-carene, limonene, and sabinene [14-17].
The current study employs a novel,
patented extraction technique (DeepSpectra®) [18,19] that
uses steam distilled turmeric EO as a natural solvent to extract non-volatile
compounds from a mixture of both ground turmeric and black pepper plant
materials. This approach eliminates the use of harsh chemical solvents (DCM,
methanol, etc.) and provides a means to obtain additional biologically active
and beneficial compounds [20, 21] that
are otherwise not detectable in either turmeric or black pepper EO. The chemical
profiles of turmeric EO and secondarily extracted (aka, DeepSpectra®
extraction) samples were established using GC/MS and UPLC-PDA analyses.
This is the first study to investigate a novel patented extraction technique
for turmeric and black pepper. Future research will further expand the
identification of additional non-volatile compounds present in DeepSpectra®
samples.
2. Materials
and methods
2.1.
Raw materials and extraction methods
Turmeric (C. longa) EO, dried
turmeric rhizomes, and dried black pepper (P. nigrum) berries were
procured from Greenleaf Extractions Pvt. Ltd. (Greenleaf, Kochi, India). Both
turmeric and black pepper plant materials were procured in a pre-ground
condition. The black pepper EO, used as a reference sample, was procured from
Young Living Essential Oils (Young Living, Lehi, UT, USA).
Secondary extraction DeepSpectra®
samples (n = 3) were produced as follows: pre-ground plant
materials of turmeric and black pepper were accurately weighed and added to
turmeric EO in an approximate ratio of 24:11:1 (oil-to-turmeric-to-peppercorn),
mixed in a beaker at 250 rpm for 2 h, and filtered using a 0.22 μm PVDF Luer
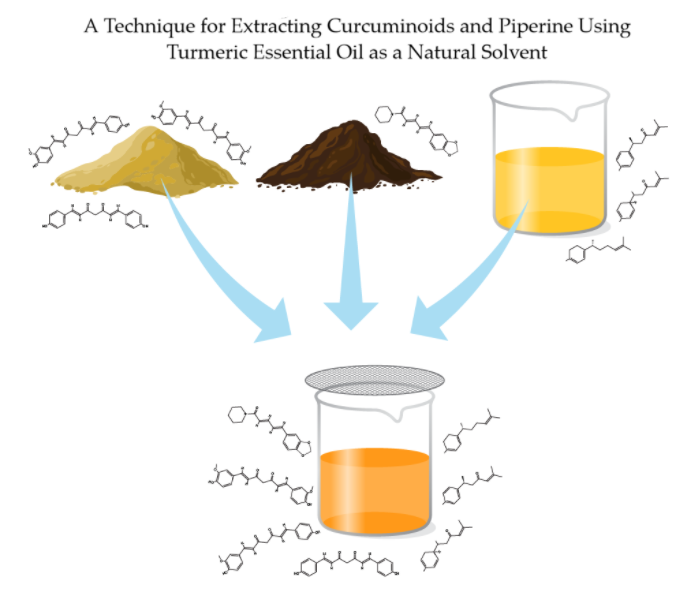
lock filter (Restek Corporation, Bellefonte, PA, USA) (Fig. 1). Exact DeepSpectra®
sample extraction details can be found in Table 1.

Figure 1. Illustration
of the DeepSpectra® extraction process. (1) Dried turmeric rhizome and black
pepper berries are ground into powder, (2) turmeric essential oil, turmeric
powder, and black pepper powder are added together in a specific ratio, (3)
solution is mixed, (4) and the resulting solution (DeepSpectra® extraction) is filtered.
Illustrated by Rick Simonson, Science Lab Studios, Inc. (Kearney, NE, USA).
Table 1. DeepSpectra® (DS)
extraction details, including raw material mass (g) and essential oil mass (g)
used for production of each sample.
Raw Materials
DS
Sample A
DS
Sample B
DS
Sample C
Turmeric
Essential Oil
24.06 g
24.00 g
23.99 g
Turmeric
Rhizome Powder
11.03 g
11.04 g
11.00 g
Black
Pepper Berry Powder
1.01 g
1.05 g
1.00 g
Curcuma longa L.
(turmeric) is a plant species belonging to the Zingiberaceae family [1]. Turmeric has a long history of cultivation,
largely for the harvest of aromatic rhizomes for medicinal, culinary, and
textile (dye) uses [2]. Modern research
has demonstrated that three primary curcuminoids (curcumin, demethoxycurcumin,
and bisdemethoxycurcumin) are responsible for the medicinal properties of rhizomes [3]. In recent years, considerable research has
been conducted on the distillation of turmeric rhizomes for the production of
EO. Studies have shown that turmeric EO demonstrates antioxidant,
antimicrobial, anti-inflammatory, antinociceptive, and herbicidal
properties [4-7]. The EO of turmeric
(specifically C. longa) is largely composed
of sesquiterpenoids [3], with
prominent compounds being AR-turmerone, α-turmerone, and β-turmerone [5-9].
Piper nigrum L.
(black pepper) is a plant species in the Piperaceae family [10] with historical importance in commerce,
particularly in the spice trade [2].
Significant research has been conducted on the therapeutic value of the
non-volatile alkaloid piperine, which is present in black pepper berries. Both
piperine and black pepper EO have been attributed with anti-inflammatory,
antimicrobial, antioxidant, and analgesic properties [11-16]. Black pepper EO is largely composed of (E)-caryophyllene,
α-pinene, β-pinene, δ-3-carene, limonene, and sabinene [14-17].
The current study employs a novel,
patented extraction technique (DeepSpectra®) [18,19] that
uses steam distilled turmeric EO as a natural solvent to extract non-volatile
compounds from a mixture of both ground turmeric and black pepper plant
materials. This approach eliminates the use of harsh chemical solvents (DCM,
methanol, etc.) and provides a means to obtain additional biologically active
and beneficial compounds [20, 21] that
are otherwise not detectable in either turmeric or black pepper EO. The chemical
profiles of turmeric EO and secondarily extracted (aka, DeepSpectra®
extraction) samples were established using GC/MS and UPLC-PDA analyses.
This is the first study to investigate a novel patented extraction technique
for turmeric and black pepper. Future research will further expand the
identification of additional non-volatile compounds present in DeepSpectra®
samples.
2. Materials
and methods
2.1.
Raw materials and extraction methods
Turmeric (C. longa) EO, dried
turmeric rhizomes, and dried black pepper (P. nigrum) berries were
procured from Greenleaf Extractions Pvt. Ltd. (Greenleaf, Kochi, India). Both
turmeric and black pepper plant materials were procured in a pre-ground
condition. The black pepper EO, used as a reference sample, was procured from
Young Living Essential Oils (Young Living, Lehi, UT, USA).
Secondary extraction DeepSpectra® samples (n = 3) were produced as follows: pre-ground plant materials of turmeric and black pepper were accurately weighed and added to turmeric EO in an approximate ratio of 24:11:1 (oil-to-turmeric-to-peppercorn), mixed in a beaker at 250 rpm for 2 h, and filtered using a 0.22 μm PVDF Luer lock filter (Restek Corporation, Bellefonte, PA, USA) (Fig. 1). Exact DeepSpectra® sample extraction details can be found in Table 1.
Figure 1. Illustration
of the DeepSpectra® extraction process. (1) Dried turmeric rhizome and black
pepper berries are ground into powder, (2) turmeric essential oil, turmeric
powder, and black pepper powder are added together in a specific ratio, (3)
solution is mixed, (4) and the resulting solution (DeepSpectra® extraction) is filtered.
Illustrated by Rick Simonson, Science Lab Studios, Inc. (Kearney, NE, USA).
Table 1. DeepSpectra® (DS) extraction details, including raw material mass (g) and essential oil mass (g) used for production of each sample.
|
Raw Materials |
DS
Sample A |
DS
Sample B |
DS
Sample C |
|
Turmeric
Essential Oil |
24.06 g |
24.00 g |
23.99 g |
|
Turmeric
Rhizome Powder |
11.03 g |
11.04 g |
11.00 g |
|
Black
Pepper Berry Powder |
1.01 g |
1.05 g |
1.00 g |
2.2. Analysis methods
To determine volatile compound profiles, EO and DeepSpectra® samples were analyzed, and compounds were identified and quantified by GC/MS using an Agilent 7890B GC/5977B MSD (Agilent Technologies, Santa Clara, CA, USA) and Agilent J&W DB-5, 60 m × 0.25 mm, 0.25 μm film thickness, fused silica capillary column. Operating conditions: 0.1 μL of sample (20% soln. for EO in ethanol), 100:1 split ratio, initial oven temp. of 40 °C with an initial hold time of 5 min, and oven ramp rate of 4.5 °C per min to 310 °C with a hold time of 5 min. The electron ionization energy was 70 eV, scan range was 35–650 amu, scan rate was 2.4 scans per s, source temperature 230 °C, and quadrupole temp. 150 °C. The compounds were identified using the Adams volatile oil library [22] and a Chemstation library search in conjunction with retention indices.
To determine the curcuminoid and piperine content, EO and DeepSpectra® samples were analyzed by UPLC-PDA. Analysis was performed using a Waters ACQUITY UPLC H-Class PLUS system coupled to a Waters Photodiode Array detector (Waters Corporation, Milford, Massachusetts, USA). Analyte separation was achieved using a CORTECS UPLC Shield RP18 (1.6 µm, 2.1 mm X 100 mm) column with a CORTECS Shield RP18 Pre-column (1.6 µm, 2.1 mm X 5 mm).
The chromatographic conditions were as follows: mobile phase A was 0.1% Citric Acid (VWR, ACS grade, item 0529) in ultra-pure water (Milli-Q IQ 7000, 0.22 µm Millipak filter) and mobile phase B was Acetonitrile (J.T. Baker, LC-MS grade, Avantor item 9829-02), and the column temperature was 35 °C. Positive identification was achieved by comparing the retention time and UV-Vis absorbance spectra obtained from the PDA detector. For curcuminoid analysis, 2.0 µL of the sample was injected onto the column and subjected to the mobile phase composition and flow rate described in Table 2. For piperine analysis, 2.0 µL of the sample was injected onto the column and subjected to the mobile phase composition and flow rate described in Table 3.
Table 2. Mobile phase details for curcuminoids (method time, flow rate, concentrations of mobile phases A and B).
Number
Time
(min)
Flow
(mL/min)
A
(%)
B
(%)
Curve
1
Initial
0.3
55
45
Initial
2
10.0
0.3
55
45
6
Table 3. Mobile phase gradient details for piperine (method time, flow rate, concentrations of mobile phases A and B).
Number
Time
(min)
Flow
(mL/min)
A
(%)
B
(%)
Curve
1
Initial
0.3
98
2
Initial
2
1.5
0.3
98
2
6
3
8.0
0.3
20
80
6
4
10.0
0.3
1
99
6
5
10.1
0.3
98
2
6
6
15.0
0.3
98
2
6
The PDA detector conditions for curcuminoid analysis was a UV-channel set at 420 nm. For the curcuminoid analysis (Curcumin, Demethoxycurcumin, and Bisdemethoxycurcumin), EO and DeepSpectra® samples were prepared for analysis by adding 100 µL of sample to 9.90 mL of HPLC-MS grade Methanol (BDH, VWR item BDH85800) with a calibrated pipette in a 15 mL light sensitive centrifuge tube. The samples were inverted several times to mix, sonicated at room temperature for 10 min, and then centrifuged at 6000 RPM for 10 min. Each sample was then filtered (Restek syringe filter, PVDF, 0.22 µm x 30 mm) into an amber HPLC vial. Quantitation of the analytes was achieved by comparing the peak area responses to an established calibration curve (linear regression, minimum R2 value of 0.995) with a range of 10 to 100 µg/mL (ppm). Calibration curves, retention times, and UV-Vis spectra were established by certified reference materials (Cayman Chemical Company, Curcumin, item 81025; Bisdemethoxycurcumin, item 10960; Demethoxycurcumin, item 10961).
2.2. Analysis methods
To determine volatile compound profiles, EO and DeepSpectra® samples were analyzed, and compounds were identified and quantified by GC/MS using an Agilent 7890B GC/5977B MSD (Agilent Technologies, Santa Clara, CA, USA) and Agilent J&W DB-5, 60 m × 0.25 mm, 0.25 μm film thickness, fused silica capillary column. Operating conditions: 0.1 μL of sample (20% soln. for EO in ethanol), 100:1 split ratio, initial oven temp. of 40 °C with an initial hold time of 5 min, and oven ramp rate of 4.5 °C per min to 310 °C with a hold time of 5 min. The electron ionization energy was 70 eV, scan range was 35–650 amu, scan rate was 2.4 scans per s, source temperature 230 °C, and quadrupole temp. 150 °C. The compounds were identified using the Adams volatile oil library [22] and a Chemstation library search in conjunction with retention indices.
To determine the curcuminoid and piperine content, EO and DeepSpectra® samples were analyzed by UPLC-PDA. Analysis was performed using a Waters ACQUITY UPLC H-Class PLUS system coupled to a Waters Photodiode Array detector (Waters Corporation, Milford, Massachusetts, USA). Analyte separation was achieved using a CORTECS UPLC Shield RP18 (1.6 µm, 2.1 mm X 100 mm) column with a CORTECS Shield RP18 Pre-column (1.6 µm, 2.1 mm X 5 mm).
The chromatographic conditions were as follows: mobile phase A was 0.1% Citric Acid (VWR, ACS grade, item 0529) in ultra-pure water (Milli-Q IQ 7000, 0.22 µm Millipak filter) and mobile phase B was Acetonitrile (J.T. Baker, LC-MS grade, Avantor item 9829-02), and the column temperature was 35 °C. Positive identification was achieved by comparing the retention time and UV-Vis absorbance spectra obtained from the PDA detector. For curcuminoid analysis, 2.0 µL of the sample was injected onto the column and subjected to the mobile phase composition and flow rate described in Table 2. For piperine analysis, 2.0 µL of the sample was injected onto the column and subjected to the mobile phase composition and flow rate described in Table 3.
Table 2. Mobile phase details for curcuminoids (method time, flow rate, concentrations of mobile phases A and B).
|
Number |
Time (min) |
Flow (mL/min) |
A (%) |
B (%) |
Curve |
|
1 |
Initial |
0.3 |
55 |
45 |
Initial |
|
2 |
10.0 |
0.3 |
55 |
45 |
6 |
Table 3. Mobile phase gradient details for piperine (method time, flow rate, concentrations of mobile phases A and B).
|
Number |
Time
(min) |
Flow
(mL/min) |
A
(%) |
B
(%) |
Curve |
|
1 |
Initial |
0.3 |
98 |
2 |
Initial |
|
2 |
1.5 |
0.3 |
98 |
2 |
6 |
|
3 |
8.0 |
0.3 |
20 |
80 |
6 |
|
4 |
10.0 |
0.3 |
1 |
99 |
6 |
|
5 |
10.1 |
0.3 |
98 |
2 |
6 |
|
6 |
15.0 |
0.3 |
98 |
2 |
6 |
The PDA detector conditions for curcuminoid analysis was a UV-channel set at 420 nm. For the curcuminoid analysis (Curcumin, Demethoxycurcumin, and Bisdemethoxycurcumin), EO and DeepSpectra® samples were prepared for analysis by adding 100 µL of sample to 9.90 mL of HPLC-MS grade Methanol (BDH, VWR item BDH85800) with a calibrated pipette in a 15 mL light sensitive centrifuge tube. The samples were inverted several times to mix, sonicated at room temperature for 10 min, and then centrifuged at 6000 RPM for 10 min. Each sample was then filtered (Restek syringe filter, PVDF, 0.22 µm x 30 mm) into an amber HPLC vial. Quantitation of the analytes was achieved by comparing the peak area responses to an established calibration curve (linear regression, minimum R2 value of 0.995) with a range of 10 to 100 µg/mL (ppm). Calibration curves, retention times, and UV-Vis spectra were established by certified reference materials (Cayman Chemical Company, Curcumin, item 81025; Bisdemethoxycurcumin, item 10960; Demethoxycurcumin, item 10961).
The PDA detector conditions for piperine analysis was a UV-channel set at 340 nm. For piperine analysis, EO and DeepSpectra® samples were prepared by adding 300 µL of the sample to 9.7 mL of acetonitrile (J.T. Baker, LC-MS grade, Avantor item 9829-02) with a calibrated pipette to a 15 mL light sensitive centrifuge tube. The samples were inverted several times to mix, vortexed for 1 min, and sonicated at room temperature for 10 min. The samples were centrifuged at 6000 RPM for 10 min, and then 100 µL of supernatant was transferred to 9.9 mL of acetonitrile (J.T.Baker, LC-MS grade, Avantor item 9829-02) in a second 15 mL light sensitive centrifuge tube with a calibrated pipette and inverted to mix. Each sample was then filtered (Restek syringe filter, PVDF, 0.22 µm x 30 mm) into an amber HPLC vial. Quantitation of the analytes was achieved by comparing the peak area responses to an established calibration curve (linear regression, minimum R2 value of 0.995) in the range of 0.1 to 1 µg/mL. Calibration curves, retention times, and UV-Vis spectra were established by a certified reference material (Sigma-Aldrich, Piperine, item P49007).
3. Results
The volatile profiles of turmeric EO, black pepper EO, and DeepSpectra® samples (n = 3) are provided in Table 4. DeepSpectra® samples were produced through a novel, patented extraction process that used turmeric EO as a solvent to extract non-volatile compounds from a mixture of turmeric rhizome and black pepper berry plant materials. Prominent compounds (≥ 5.0%) in the turmeric EO and DeepSpectra® samples included γ-curcumene (7.1%, 8.6%), α-zingiberene (4.1%, 5.3%), AR-turmerone (46.6%, 42.1%), and α-turmerone (8.5%, 8.3%). The prominent compounds in the black pepper EO were α-pinene (11.6%), sabinene (9.4%), β-pinene (9.5%), δ-3-carene (7.8%), limonene (13.0%), and (E)-caryophyllene (27.6%).
Table 4. Volatile compounds detected in essential oil (EO) or DeepSpectra® (DS) samples.
Compound name
KI
Turmeric EO
DeepSpectra®
sample
Black
Pepper EO
α-Thujene
924
nd
nd
1.1
α-Pinene
932
0.1
0.1
11.6
Camphene
946
0.1
0.1
0.3
Sabinene
969
tr
tr
9.4
β-Pinene
974
nd
nd
9.5
Myrcene
988
nd
nd
0.8
α-Phellandrene
1002
1.2
1.9
0.7
δ-3-Carene
1008
tr
0.1
7.8
α-Terpinene
1014
0.1
0.2
0.1
p-Cymene
1020
1.2
1.8
0.8
Limonene
1024
0.3
0.4
13.0
1,8-Cineole
1026
1.2
1.9
nd
(E)-β-Ocimene
1044
nd
nd
tr
γ-Terpinene
1054
0.1
0.1
0.1
(Z)-Sabinene
hydrate
1065
nd
nd
0.1
p-Mentha-2,4(8)-diene
1085
nd
nd
0.1
Terpinolene
1086
1.0
1.5
0.2
Linalool
1095
nd
nd
0.2
(E)-Sabinene
hydrate
1098
nd
nd
0.1
Terpinen-4-ol
1174
tr
tr
0.4
p-Cymen-8-ol
1179
tr
tr
nd
Dill
Ether
1184
tr
tr
nd
α-Terpineol
1186
tr
tr
tr
Thymol
1289
tr
tr
nd
δ-Elemene
1335
nd
nd
2.2
α-Cubebene
1348
nd
nd
0.2
Cyclosativene
1369
nd
nd
0.1
unknown
compound 1
*1371
3.1
3.5
nd
α-Copaene
1374
nd
nd
4.2
β-cubebene
1387
nd
nd
0.8
α-Gurjunene
1409
nd
nd
0.1
α-Cedrene
1410
0.2
0.4
nd
(E)-Caryophyllene
1417
0.8
1.1
27.6
β-Ylangene
1419
nd
nd
0.2
α-Guaiene
1437
nd
nd
0.1
(Z)-β-Farnesene
1440
nd
nd
tr
unknown
compound 2
*1444
0.7
0.8
nd
(Z)-Muurola-3,5-diene
1448
nd
nd
tr
Amorpha-4,11-diene
1449
0.9
1.1
nd
α-Humulene
1452
nd
nd
1.4
(Z)-Cadina-1(6),4-diene
1461
nd
nd
tr
γ-Muurolene
1478
nd
nd
0.1
AR-Curcumene
1479
2.5
3.1
nd
Germacrene
D
1480
nd
nd
0.2
γ-Curcumene
1481
7.1
8.6
nd
β-Selinene
1489
nd
nd
0.3
α-Zingiberene
1493
4.1
5.3
nd
α-Muurolene
1500
nd
nd
1.0
β-Bisabolene
1505
1.0
1.3
1.5
Cubebol
1514
nd
nd
0.2
β-Curcumene
1514
0.3
0.4
nd
β-Sesquiphellandrene
1521
1.7
2.0
nd
δ-Cadinene
1522
nd
nd
1.7
(E)-γ-Bisabolene
1529
0.3
0.3
nd
Spathulenol
1577
nd
nd
tr
Caryophyllene
oxide
1582
nd
nd
1.1
Dihydro-AR-turmerone
1595
3.5
2.9
nd
unknown
compound 3
*1609
1.0
1.0
nd
α-Muurolol
1644
nd
nd
0.2
unknown
compound 4
*1648
0.9
0.7
nd
AR-Turmerone
1668
46.6
42.1
nd
α-Turmerone
*1672
8.5
8.3
nd
(Z)-γ-Atlantone
1694
0.7
0.5
nd
β-Turmerone
*1701
2.7
2.4
nd
(E)-γ-Atlantone
1706
0.9
0.7
nd
(6R,7R)-Bisabolene
1740
0.5
0.3
nd
(E)-α-Atlantone
1777
1.3
0.8
nd
The compound name, KI, and relative area % are
reported. KI is the Kovat’s Index value and was previously calculated by Robert
Adams using a linear calculation on a DB-5 column [22].
Values for turmeric and black pepper EO samples resulted from the analysis of
single samples. Values for the DS sample are an average value from three
samples (n = 3). *Either unknown compound or compound not found in
the Adams Library [22]. KI manually calculated.
Several non-volatile compounds were identified in the DeepSpectra® samples; however, these same non-volatile compounds were either possibly detected (but below the limit of quantitation) or not detected in either EO sample (turmeric or black pepper). The non-volatile compounds detected in DeepSpectra® samples were characteristic of turmeric (curcumin, demethoxycurcumin, and bisdemethoxycurcumin) and black pepper (piperine). A summary of the UPLC-PDA findings is provided in Table 5.
Table 5. Non-volatile
compounds detected in DeepSpectra® samples (average).
Compound Name
Value (mg/mL)
Curcumin
5.30
Demethoxycurcumin
2.94
Bisdemethoxycurcumin
3.59
Piperine
1.56
3. Results
The volatile profiles of turmeric EO, black pepper EO, and DeepSpectra® samples (n = 3) are provided in Table 4. DeepSpectra® samples were produced through a novel, patented extraction process that used turmeric EO as a solvent to extract non-volatile compounds from a mixture of turmeric rhizome and black pepper berry plant materials. Prominent compounds (≥ 5.0%) in the turmeric EO and DeepSpectra® samples included γ-curcumene (7.1%, 8.6%), α-zingiberene (4.1%, 5.3%), AR-turmerone (46.6%, 42.1%), and α-turmerone (8.5%, 8.3%). The prominent compounds in the black pepper EO were α-pinene (11.6%), sabinene (9.4%), β-pinene (9.5%), δ-3-carene (7.8%), limonene (13.0%), and (E)-caryophyllene (27.6%).
Table 4. Volatile compounds detected in essential oil (EO) or DeepSpectra® (DS) samples.
|
Compound name |
KI |
Turmeric EO |
DeepSpectra®
sample |
Black
Pepper EO |
|
α-Thujene |
924 |
nd |
nd |
1.1 |
|
α-Pinene |
932 |
0.1 |
0.1 |
11.6 |
|
Camphene |
946 |
0.1 |
0.1 |
0.3 |
|
Sabinene |
969 |
tr |
tr |
9.4 |
|
β-Pinene |
974 |
nd |
nd |
9.5 |
|
Myrcene |
988 |
nd |
nd |
0.8 |
|
α-Phellandrene |
1002 |
1.2 |
1.9 |
0.7 |
|
δ-3-Carene |
1008 |
tr |
0.1 |
7.8 |
|
α-Terpinene |
1014 |
0.1 |
0.2 |
0.1 |
|
p-Cymene |
1020 |
1.2 |
1.8 |
0.8 |
|
Limonene |
1024 |
0.3 |
0.4 |
13.0 |
|
1,8-Cineole |
1026 |
1.2 |
1.9 |
nd |
|
(E)-β-Ocimene |
1044 |
nd |
nd |
tr |
|
γ-Terpinene |
1054 |
0.1 |
0.1 |
0.1 |
|
(Z)-Sabinene
hydrate |
1065 |
nd |
nd |
0.1 |
|
p-Mentha-2,4(8)-diene |
1085 |
nd |
nd |
0.1 |
|
Terpinolene |
1086 |
1.0 |
1.5 |
0.2 |
|
Linalool |
1095 |
nd |
nd |
0.2 |
|
(E)-Sabinene
hydrate |
1098 |
nd |
nd |
0.1 |
|
Terpinen-4-ol |
1174 |
tr |
tr |
0.4 |
|
p-Cymen-8-ol |
1179 |
tr |
tr |
nd |
|
Dill
Ether |
1184 |
tr |
tr |
nd |
|
α-Terpineol |
1186 |
tr |
tr |
tr |
|
Thymol |
1289 |
tr |
tr |
nd |
|
δ-Elemene |
1335 |
nd |
nd |
2.2 |
|
α-Cubebene |
1348 |
nd |
nd |
0.2 |
|
Cyclosativene |
1369 |
nd |
nd |
0.1 |
|
unknown
compound 1 |
*1371 |
3.1 |
3.5 |
nd |
|
α-Copaene |
1374 |
nd |
nd |
4.2 |
|
β-cubebene |
1387 |
nd |
nd |
0.8 |
|
α-Gurjunene |
1409 |
nd |
nd |
0.1 |
|
α-Cedrene |
1410 |
0.2 |
0.4 |
nd |
|
(E)-Caryophyllene |
1417 |
0.8 |
1.1 |
27.6 |
|
β-Ylangene |
1419 |
nd |
nd |
0.2 |
|
α-Guaiene |
1437 |
nd |
nd |
0.1 |
|
(Z)-β-Farnesene |
1440 |
nd |
nd |
tr |
|
unknown
compound 2 |
*1444 |
0.7 |
0.8 |
nd |
|
(Z)-Muurola-3,5-diene |
1448 |
nd |
nd |
tr |
|
Amorpha-4,11-diene |
1449 |
0.9 |
1.1 |
nd |
|
α-Humulene |
1452 |
nd |
nd |
1.4 |
|
(Z)-Cadina-1(6),4-diene |
1461 |
nd |
nd |
tr |
|
γ-Muurolene |
1478 |
nd |
nd |
0.1 |
|
AR-Curcumene |
1479 |
2.5 |
3.1 |
nd |
|
Germacrene
D |
1480 |
nd |
nd |
0.2 |
|
γ-Curcumene |
1481 |
7.1 |
8.6 |
nd |
|
β-Selinene |
1489 |
nd |
nd |
0.3 |
|
α-Zingiberene |
1493 |
4.1 |
5.3 |
nd |
|
α-Muurolene |
1500 |
nd |
nd |
1.0 |
|
β-Bisabolene |
1505 |
1.0 |
1.3 |
1.5 |
|
Cubebol |
1514 |
nd |
nd |
0.2 |
|
β-Curcumene |
1514 |
0.3 |
0.4 |
nd |
|
β-Sesquiphellandrene |
1521 |
1.7 |
2.0 |
nd |
|
δ-Cadinene |
1522 |
nd |
nd |
1.7 |
|
(E)-γ-Bisabolene |
1529 |
0.3 |
0.3 |
nd |
|
Spathulenol |
1577 |
nd |
nd |
tr |
|
Caryophyllene
oxide |
1582 |
nd |
nd |
1.1 |
|
Dihydro-AR-turmerone |
1595 |
3.5 |
2.9 |
nd |
|
unknown
compound 3 |
*1609 |
1.0 |
1.0 |
nd |
|
α-Muurolol |
1644 |
nd |
nd |
0.2 |
|
unknown
compound 4 |
*1648 |
0.9 |
0.7 |
nd |
|
AR-Turmerone |
1668 |
46.6 |
42.1 |
nd |
|
α-Turmerone |
*1672 |
8.5 |
8.3 |
nd |
|
(Z)-γ-Atlantone |
1694 |
0.7 |
0.5 |
nd |
|
β-Turmerone |
*1701 |
2.7 |
2.4 |
nd |
|
(E)-γ-Atlantone |
1706 |
0.9 |
0.7 |
nd |
|
(6R,7R)-Bisabolene |
1740 |
0.5 |
0.3 |
nd |
|
(E)-α-Atlantone |
1777 |
1.3 |
0.8 |
nd |
The compound name, KI, and relative area % are
reported. KI is the Kovat’s Index value and was previously calculated by Robert
Adams using a linear calculation on a DB-5 column [22].
Values for turmeric and black pepper EO samples resulted from the analysis of
single samples. Values for the DS sample are an average value from three
samples (n = 3). *Either unknown compound or compound not found in
the Adams Library [22]. KI manually calculated.
Several non-volatile compounds were identified in the DeepSpectra® samples; however, these same non-volatile compounds were either possibly detected (but below the limit of quantitation) or not detected in either EO sample (turmeric or black pepper). The non-volatile compounds detected in DeepSpectra® samples were characteristic of turmeric (curcumin, demethoxycurcumin, and bisdemethoxycurcumin) and black pepper (piperine). A summary of the UPLC-PDA findings is provided in Table 5.
Table 5. Non-volatile compounds detected in DeepSpectra® samples (average).
|
Compound Name |
Value (mg/mL) |
|
Curcumin |
5.30 |
|
Demethoxycurcumin |
2.94 |
|
Bisdemethoxycurcumin |
3.59 |
|
Piperine |
1.56 |
4. Discussion
The turmeric EO was a light-orange color. Upon extracting compounds from both turmeric rhizome and black pepper berries, the resulting liquid solution was dark, amber-orange color. This initial finding suggests that the turmeric EO is a suitable solvent for extracting additional compounds from both plant materials.
GC/MS analysis resulted in similar volatile profiles for both turmeric EO and DeepSpectra® samples (n = 3); however, there were subtle differences. The average relative abundance of monoterpenoids (10-carbon backbone) and sesquiterpenoids (15-carbon backbone) in the EO samples differed from that in the DeepSpectra samples. Monoterpenoids comprised 5.3% of the turmeric EO and 8.2% of the DeepSpectra® samples. Sesquiterpenoids comprised an average of 89.4% of the turmeric EO and 87.3% of the DeepSpectra® samples. These data suggest that the DeepSpectra® process increases the efficiency of monoterpenoid recovery from plant material samples when extracting compounds from fresh raw materials.
UPLC-PDA analysis of the DeepSpectra® samples resulted in the detection of several curcuminoids and piperine (Fig. 1). However, these same non-volatile compounds were either possibly detected (but below the limit of quantitation) or not detected in either turmeric or black pepper EO samples. The detected curcuminoids in the DeepSpectra® samples included curcumin (aka diferuloylmethane), demethoxycurcumin, and bisdemethoxycurcumin. Additionally, other non-volatile compounds were expected to be co-extracted through DeepSpectra® extraction (Fig. 2), as found in previous studies [20, 21]. However, additional reference standards and resources are required for their identification. These data suggest that the turmeric EO used in the DeepSpectra® process, which is employed as a natural solvent for extracting non-volatile compounds from raw plant materials, is a suitable solvent. Previous reports on the essential oil profile of turmeric [5-9] did not report the presence of any curcuminoids. However, it should be noted that these same analyses were only conducted using gas chromatography, which is typically reserved for analyzing volatile compounds with relatively low molecular weights (i.e., not curcuminoids). DeepSpectra® samples are unique in their chemical composition, which comprises a range of compounds, including monoterpenoids, sesquiterpenoids, curcuminoids, and alkaloids.
Figure 2. UPLC-PDA chromatographic overlay of turmeric essential oil (black) and the DeepSpectra® extraction (red).
The DeepSpectra® extraction employed herein (co-extraction of turmeric and black pepper plant materials simultaneously) was based on research reports that the co-administration of curcuminoids and piperine increases absorption and sustained bioavailability in both animal and human models [23, 24]. Initial research was conducted to achieve approximately a 10:1 ratio of total curcuminoids to piperine in the DeepSpectra® sample. The DeepSpectra® extract, with an approximate 10:1 ratio of curcuminoids to piperine, as well as additional ratios of turmeric and black pepper plant materials, will be produced and evaluated in the future to determine their extraction efficiency and biological activities. Regardless, the current study is the first to establish the use of turmeric EO as a natural and reliable solvent for extracting non-volatile compounds from both turmeric rhizomes and black pepper berries.
5. Conclusions
A novel patented extraction technique (DeepSpectra®) was employed using turmeric EO as a natural solvent for extracting medicinally important non-volatile compounds from turmeric rhizome (curcuminoids) and black pepper berries (piperine). GC/MS and UPLC-PDA were employed to evaluate the volatile and non-volatile characteristics of the resulting extracts, respectively. The volatile profiles of the DeepSpectra® samples was largely composed of γ-curcumene (8.6%), α-zingiberene (5.3%), AR-turmerone (42.1%), and α-turmerone (8.3%). The non-volatile profiles of the DeepSpectra® samples contained curcumin (5.30 mg/mL), demethoxycurcumin (2.94 mg/mL), bisdemethoxycurcumin (3.59 mg/mL), and piperine (1.56 mg/mL). These same non-volatile compounds were either possibly detected (but below the limit of quantitation) or not detected in either turmeric or black pepper EO samples. These findings suggest that turmeric EO is a reliable natural solvent for extracting non-volatile compounds from raw plant materials.
Patents
United States Patent Number: US 12,559,697 B2. Date of Patent: 24 February 2026. Publication Title: METHODS AND SYSTEMS FOR EXTRACTING ADDITIONAL BENEFICIAL LIPID-SOLUBLE COMPOUNDS FROM PLANT MATERIALS IN ENVIRONEMENTIALLY SUSTAINABLE WAYS.
United States Patent Number: US 12,540,293 B2. Date of Patent: 3 February 2026. Publication Title: METHODS AND SYSTEMS FOR EXTRACTING ADDITIONAL BENEFICIAL LIPID-SOLUBLE COMPOUNDS FROM PLANT MATERIALS IN ENVIRONEMENTIALLY SUSTAINABLE WAYS.
Abbreviations
The following abbreviations are used in this manuscript: EO (Essential Oil); DCM (Dichloromethane); DS (DeepSpectra®); GC/MS (Gas Chromatography/Mass Spectrometry); UPLC-PDA (Ultra-Performance Liquid Chromatography – Photodiode Array Detector).
Disclaimer (artificial intelligence)
Author(s) hereby state that no generative AI tools such as Large Language Models (ChatGPT, Copilot, etc.) and text-to-image generators were utilized in the preparation or editing of this manuscript.
Authors’ contributions
Conceptualization, M.C.J., H.K.L., T.M.W.; sample procurement and production, M.C.J., H.K.L., T.M.W.; methodology, M.C.J., T.M.W.; software, M.C.J., T.M.W.; validation, C.R.B.; formal analysis, M.C.J., T.M.W.; data curation, M.C.J., T.M.W.; writing—original draft preparation, T.M.W.; writing—review and editing, M.C.J., H.K.L., C.R.B., T.M.W.; funding acquisition, C.R.B.
Acknowledgements
The authors wish to thank Rick Simonson (Science Lab Studios, Inc.) for the botanical illustration.
Funding
This research was funded by Young Living Essential Oils.
Availability of data and materials
All data are presented within the current manuscript.
Conflicts of interest
The authors declare no conflicts of interest. While the funders (Young Living Essential Oils) hold the patent for DeepSpectra® technology, the funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
1. | The World Flora Online. Available online: https://www.worldfloraonline.org/taxon/wfo-0000365771 (accessed on 26 Feb 2026). |
2. | Bailey, L.H. Manual of Cultivated Plants, revised ed.; Macmillan Publishing Company: New York, NY, USA, pp. 287-289, 317, 1951. |
3. | Li, S.; Yuan, W.; Deng, G.; Wang, P.; Yang, P.; Aggarwal, B. Chemical composition and product quality control of turmeric (Curcuma longa L.). Pharm. Crop. 2011, 2, 28-54. |
4. | Kebede, B.H.; Forsido, S.F.; Tola, Y.B.; Astatkie, T. Free radical scavenging capacity, antibacterial activity and essential oil composition of turmeric (Curcuma domestica) varieties grown in Ethiopia. Heliyon. 2021, 7(2). https://doi.org/10.1016/j.heliyon.2021.e06239 |
5. | Liju, V.B.; Jeena, K.; Kuttan, R. An evaluation of antioxidant, anti-inflammatory, and antinociceptive activities of essential oil from Curcuma longa L. Indian J. Pharmacol. 2011, 43(5), 526-531. https://doi.org/10.4103/0253-7613.84961 |
6. | Stanojević, J.S.; Stanojević, L.P.; Cvetković, D.J.; Danilović, B.R. Chemical composition, antioxidant and antimicrobial activity of the turmeric essential oil (Curcuma longa L.). Adv. Technol. 2015, 4(2), 19-25. |
7. | Ibáñez, M.D.; Blázquez, M.A. Ginger and turmeric essential oils for weed control and food crop protection. Plants. 2019, 8, 59. https://doi.org/10.3390/plants8030059 |
8. | Duong, L.; Mentreddy, S.R.; Satyal, R.; Satyal, P.; Setzer, W.N. Essential oil chemotypes of four Vietnamese Curcuma Species cultivated in North Alabama. Horticulturae. 2022, 8, 360. https://doi.org/10.3390/horticulturae8050360 |
9. | Setzer, W.N.; Duong, L.; Poudel, A.; Mentreddy, S.R. Variation in the chemical composition of five varieties of Curcuma longa rhizome essential oils cultivated in North Alabama. Foods. 2021, 10, 212. https://doi.org/10.3390/foods10020212 |
10. | The World Flora Online. Available online: https://www.worldfloraonline.org/taxon/wfo-0000486494 (accessed on 26 Feb 2026). |
11. | Vijayakumar, R. S.; Surya, D.; Nalini, N. Antioxidant efficacy of black pepper (Piper nigrum L.) and piperine in rats with high fat diet induced oxidative stress. Redox Report. 2004, 9(2), 105–110. https://doi.org/10.1179/135100004225004742 |
12. | Zarai, Z.; Boujelbene, E.; Salem, N.B.; Gargouri, Y.; Sayari, A. Antioxidant and antimicrobial activities of various solvent extracts, piperine and piperic acid from Piper nigrum. LWT-Food Sci. Technol. 2013, 50(2), 634-641. https://doi.org/10.1016/j.lwt.2012.07.036 |
13. | Tasleem, F.; Azhar, I.; Ali, S.N.; Perveen, S.; Mahmood, Z.A. Analgesic and anti-inflammatory activities of Piper nigrum L. Asian Pac. J. Trop. Med. 2014, 7, 461-468. https://doi.org/10.1016/S1995-7645(14)60275-3 |
14. | Jeena, K.; Liju, V.B.; Umadevi, N.P.; Kuttan, R. Antioxidant, anti-inflammatory and antinociceptive properties of black pepper essential oil (Piper nigrum Linn). J. Essent. Oil-Bear. Plant. 2014, 17(1), 1–12. https://doi.org/10.1080/0972060X.2013.831562 |
15. | Bagheri, H.; Manap, M.Y.B.A.; Solati, Z. Antioxidant activity of Piper nigrum L. essential oil extracted by supercritical CO2 extraction and hydro-distillation. Talanta. 2014, 121, 220-228. https://doi.org/10.1016/j.talanta.2014.01.007 |
16. | Singh, S.; Kapoor, I.P.S.; Singh, G.; Schuff, C.; De Lampasona, M.P.; Catalan, C.A.N. Chemistry, antioxidant and antimicrobial potentials of white pepper (Piper nigrum L.) essential oil and oleoresins. Proc. Natl. Acad. Sci., India, Sect. B Biol. Sci. 2013, 83, 357–366. https://doi.org/10.1007/s40011-012-0148-4 |
17. | Liu, L.; Song, G.; Hu, Y. GC–MS Analysis of the essential oils of Piper nigrum L. and Piper longum L. Chromatograph. 2007, 66, 785–790. https://doi.org/10.1365/s10337-007-0408-2 |
18. | United States Patent Number: US 12,559,697 B2. Date of Patent: 24 February 2026. Publication Title: METHODS AND SYSTEMS FOR EXTRACTING ADDITIONAL BENEFICIAL LIPID-SOLUBLE COM-POUNDS FROM PLANT MATERIALS IN ENVIRONEMENTIALLY SUSTAINABLE WAYS. |
19. | United States Patent Number: US 12,540,293 B2. Date of Patent: 3 February 2026. Publication Title: METHODS AND SYSTEMS FOR EXTRACTING ADDITIONAL BENEFICIAL LIPID-SOLUBLE COMPOUNDS FROM PLANT MATERIALS IN ENVIRONEMENTIALLY SUSTAINABLE WAYS. |
20. | Wilson, T.M.; McKay, T.T.; Lin, H.K.; Packer, C.; Bowerbank, C.R. A novel technique for extracting terpenoids from aromatic plant materials. J. Essent. Oil Plant Comp. 2025, 3(1), 60-68. https://doi.org/10.58985/jeopc.2025.v03i01.66 |
21. | Wilson, T.M.; Johnson, M.C.; Lin, H.K.; Jackson, R.B.; Packer, C.; Bowerbank, C.R. Pinus monophylla (Pinaceae) resin: A novel technique for extracting terpenoids from aromatic plant materials. J. Essent. Oil Plant Comp. 2025, 3(2), 69-75. https://doi.org/10.58985/jeopc.2025.v03i02.67 |
22. | Adams, R.P. Identification of essential oil components by gas chromatography/mass spectrometry, 4th ed.; Allured Publishing: Carol Stream, IL, USA, 2007. |
23. | Shoba, G.; Joy, D.; Joseph, T.; Majeed, M.; Rajendran, R.; Srinivas. P.S. Influence of piperine on the pharmacokinetics of curcumin in animals and human volunteers. Planta Med. 1998, 64(4), 353-6. https://doi.org/10.1055/s-2006-957450 |
24. | Suresh, D.; Srinivasan, K. Tissue distribution & elimination of capsaicin, piperine & curcumin following oral intake in rats. Indian J. Med. Res. 2010, 131, 682-91. |
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
Abstract
Turmeric (Curcuma longa) and
black pepper (Piper nigrum) have a long history of cultivation and economic
value. Both plants have been celebrated for their culinary and medicinal
properties. The current study investigated the use of a novel patented
extraction technique (DeepSpectra®) that employs turmeric essential oil (EO) as
a natural solvent for extracting medicinally important non-volatile compounds
from turmeric rhizomes (curcuminoids) and black pepper berries (piperine).
GC/MS and UPLC-PDA were employed to evaluate the volatile and non-volatile
characteristics of the resulting extracts, respectively. The volatile profile
was largely composed of γ-curcumene (8.6%), α-zingiberene (5.3%), AR-turmerone
(42.1%), and α-turmerone (8.3%). The non-volatile profile contained curcumin
(5.30 mg/mL), demethoxycurcumin (2.94 mg/mL), bisdemethoxycurcumin (3.59
mg/mL), and piperine (1.56 mg/mL). These findings suggest that turmeric EO is a
reliable, natural solvent for extracting non-volatile compounds from raw plant
materials.
Abstract Keywords
Black pepper, curcuminoids, DeepSpectra®, gas chromatography, liquid chromatography, mass spectrometry, piperine, turmeric.
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
Editor-in-Chief
This work is licensed under the
Creative Commons Attribution 4.0
License.(CC BY-NC 4.0).


