Short Communication
Isabel P. Lykken
Isabel P. Lykken
D. Gary Young Research Institute, Lehi, UT 84043, USA.
Alma Laney
Alma Laney
Department of Biology, Utah Valley University, Orem, UT 84058, USA.
E-mail: alaney@uvu.edu
Christopher R. Bowerbank
Christopher R. Bowerbank
D. Gary Young Research Institute, Lehi, UT 84043, USA.
Tyler M. Wilson*
Tyler M. Wilson*
Corresponding
Author
D. Gary
Young Research Institute, Lehi, UT 84043, USA.
E-mail: tywilson@youngliving.com; Tel.: +1-801-669-4501
Received: 2026-02-24 | Revised:2026-03-15 | Accepted: 2026-03-17 | Published: 2026-03-25
Pages: 50-56
DOI: https://doi.org/10.58985/jafsb.2026.v04i01.91
Abstract
Essential oils are commonly researched as alternative ingredients to conventional fungicides. The current study investigated the antifungal activity of essential oils with high concentrations of aldehydes (cassia and lemongrass) and phenols (oregano). Plant chemotypes and prominent antifungal compounds were determined by GC/MS analysis (reported as area %). Cassia oil contains (E)-cinnamaldehyde (61.0%) and (E)-o-methoxy cinnamaldehyde (20.3%); lemongrass oil contains neral (30.6%) and geranial (34.8%); oregano oil contains thymol (3.0%) and carvacrol (59.2%). Antifungal activity against Botrytis cinerea (cause of gray mold) was evaluated using a technique for embedding hydrophobic essential oils homogenously into water-based PDA agar media. Oregano demonstrated the highest antifungal activity (MIC 50 μL/15 mL), followed by cassia (MIC 60 μL/15 mL) and lemongrass (70 μL/15 mL). These findings support research into natural alternatives for conventional fungicides.
Keywords
Antifungal, Botrytis cinerea, cassia, essential oil, gray mold, lemongrass, oregano, gas chromatography.
1. Introduction
In agricultural landscapes, the contamination of crops with plant pathogenic fungi (PPF) presents health and economic concerns [1-4]. Substantial health risk to humans and livestock are due to exposure of edible goods to mycotoxins [3-5]. In addition, fungal infections can decrease the quantity and quality of agricultural products, severely impairing crop productivity [1, 3]. Among these PPF, B. cinerea is the source of gray mold disease, and affects over 200 plant species, notably economically important crops including apples, strawberries, grapes, and a variety of vegetables [1, 3, 6-9]. Infection by B. cinerea typically occurs at the field stage; however, due to the fungal species’ capacity to remain dormant until environmental conditions for growth are optimal, it is considered a leading cause of pre- and post-harvest rot [1, 7-9]. Current strategies for the management of gray mold rely primarily on synthetic fungicide applications, promotion of disease resistant crops, and various non-chemical means [3, 7, 8]. Despite their widespread use, these measures are becoming increasingly ineffective against species like B. cinerea, which are known to develop fungicide resistance due to genetic plasticity [1, 8, 9].
In conventional agriculture, synthetic fungicides are applied after flowering and during post-harvest storage [10]. Commercially available fungicides include compounds such as dichloran (nitro derivative), flutriafol (triazole), o-phenylphenol (biphenyl), prochloraz (imidazole), and tolclofos methyl (thiophosphate), which vary in their chemical and crop specificity [3, 5, 11]. Additionally, post-harvest fruits and crops are typically treated with sulfur dioxide to suppress fungal growth [10]. However, the use of synthetic fungicides and sulfur dioxide is not allowed where it is limited by regulatory requirements, or where organic certification is desired [5, 10]. The extensive use of chemical fungicides has been linked to environmental degradation, with growing concern for residues remaining in the soil well beyond the growing season [3, 4, 7, 10-12]. With mounting environmental impact concerns, coupled with industry restrictions, there is a growing demand for sustainable and safer alternatives to synthetic fungicides.
In response, there has been substantial research on plant-derived compounds with antifungal properties. Unlike synthetic fungicides, essential oils consist of complex mixtures of bioactive compounds that may reduce the likelihood of resistance development in fungal populations [3, 4, 9, 10, 12, 13]. The antifungal efficacy of certain essential oil compounds has been linked to the presence of hydroxyl groups in their chemical structure and hydrophobic moieties that interact with fungal membranes [14-16]. Oils rich in phenols (for example, carvacrol) and aldehydes (for example, cinnamaldehyde and citral) have demonstrated notable effectiveness against B. cinerea [7, 12, 15, 16].
The current study investigates the antifungal activity of three essential oils: cassia, lemongrass, and oregano. The study approach, an in vitro petri dish study, involved embedding hydrophobic essential oils into a water-based agar. These findings support the use of natural plant extracts (essential oils) as sustainable alternatives to synthetic fungicides.
2. Materials and methods
2.1. Essential oil preparation and analysis
All essential oils, which were produced by steam distillation, were provided by Young Living Essential Oils (Lehi, UT, USA) and were analyzed by GC/MS to establish the plant chemotype and essential oil quality. Essential oil samples were analyzed, and volatile compounds were identified by GC/MS using an Agilent 7890B GC/5977B MSD (Agilent Technologies, Santa Clara, CA, USA) and Agilent J&W DB-5, 0.25 mm × 60 m, 0.25 μm film thickness, fused silica capillary column. Operating conditions: 0.1 μL of sample (20% soln. for essential oils in methylene chloride), 100:1 split ratio, initial oven temp. of 40 °C with an initial hold time of 5 min., and an oven ramp rate of 4.5 °C per min. to 310 °C with a hold time of 5 min. The electron ionization energy was 70 eV, the scan range was 35–650 amu, the scan rate was 2.4 scans per sec., source temp. 230 °C, and quadrupole temp. 150 °C. Volatile compounds were identified using the Adams volatile oil library [17] using Chemstation library search in conjunction with retention indices. One volatile compound detected in this study, 4-Nonanone, was not found in the Adams volatile oil library. The identification of this compound was made using the 2020 NIST mass spectral library and retention indices were manually calculated using alkane standards. Noted that limonene/1,8-cineole eluted as a single peak in some samples. Their relative amounts were determined by the mass to charge ratio (m/z) 41, 68, 79 (limonene) and 43, 71, 81 (1,8-cineole).
2.2. Fungal identification
Gray mold, which is caused by B. cinerea, was observed on and isolated from post-harvest strawberries exhibiting typical symptoms for this pathogen. Symptomatic strawberries were washed three times with deionized water, soaked in 70% isopropyl alcohol, and the plant tissue was placed on potato dextrose agar (PDA) medium, and incubated at 25 °C (± 2 °C) for 7 days. After the 7-day incubation period, fungal colonies that displayed morphological characteristics of gray mold were selected and transferred to additional PDA medium for genetic identification. To verify the identity of the fungal isolate, DNA was extracted from mycelial scrapings of freshly grown Petri plates using the Plant Isolate DNA extraction kit (IBI Scientific, Dubuque, IA, USA) according to the manufacturer’s recommendations. The DNA was then used as a template for ITS PCR amplification using Apex Taq RED master mix (Apex Bioresearch, Houston, TX, USA) and the primer set ITS1F and TW13 [18]. The PCR amplicons were purified using AMPure XP beads (Beckman Coulter Life Sciences, Indianapolis, IN, USA) according to the manufacturer’s recommendations. This was then subjected to Sanger Sequencing (Psomagen, Inc., Rocksville, MD, USA) to a 3x coverage with both primer sets and assembled into a contig using CAP3 [19]. The contig was then subjected to BLASTN [20] to identify the nearest relative, sharing 100% nucleotide identity to accession #OR544950.1. The contig was deposited in GenBank (accession #PQ460570).
2.3. Fungal pathogen isolation and inoculation
All experiments were conducted under sterile conditions to evaluate the antifungal activity of the selected essential oils against B. cinerea using an agar-based growth inhibition assay. Agar was prepared by combining 10 g of potato dextrose agar and 10 g of malt agar (Millipore Sigma, Sigma Aldrich, St. Louis, MS, USA) with 500 mL of deionized water. The mixture was heated on a stir plate until it reached a rolling boil and became clear and free of debris. The agar was then dispensed into separate vessels in 75 mL increments, and a stir bar was added to each vessel. Bottles were sterilized by heating for 15 min at 15 psi using a pressure cooker.
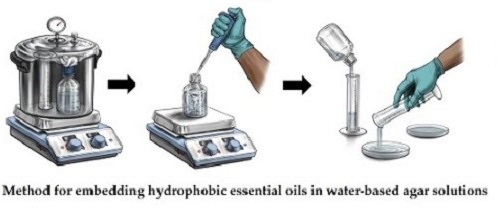
Essential oil treatments were prepared by mixing each oil with Tween 80 (Bulk Apothecary, Aurora, OH, USA) and deionized water at a 1:3:10 ratio. The agar bottles (containing 75 mL of agar solution) were transferred from the pressure cooker to a sterile fume hood containing a stir plate. Maintaining sterile conditions, each agar bottle was placed on a stir plate at 200 rpm, essential oil treatments were accurately measured and added to the bottles, and the solutions were mixed (Fig. 1). This resulted in a homogenously embedded essential oil dose across plates (e.g., 500 µL added per bottle yielded 100 µL per 15 mL plate). Both a non-treatment agar control (water, but no Tween 80 or essential oil) and a vehicle control (Tween 80 and water, but no essential oil) were prepared and processed identically to the treatment plates. Essential oil treatments were tested using 5 replicate plates per dose. The replicate variability of colony diameter at each dose was assessed using the relative standard deviation (RSD). Across all oils and doses, the RSD values were low (consistently < 3%), indicating consistent aliquots and high reproducibility of the method.
Figure 1. Illustration detailing method for directly embedding hydrophobic essential oils in water-based agar solutions.
(1) Lab glassware and agar solution are sterilized using a pressure cooker, (2) essential oil, Tween 80, and water solution are accurately weighed, added to sterile agar solution, and mixed, (3) 15 mL of essential oil-embedded agar solution are poured into petri dish. Illustration by Zach Nielsen.
Following mixing, the agar was poured into Petri dishes in 15 mL aliquots (3). Bottles were returned to the stir plate between pouring to maintain a homogeneous solution. This was repeated for all aliquots, and all plates were left to solidify for 1 h in a ventilated hood at room temperature. Once cooled, and prior to inoculation, the plates were exposed to UV light for 5 min using a UV Clave UV Chamber (Benchmark Scientific Inc., Sayreville, NJ, USA). Plates were inoculated using a sterilized scalpel to cut 0.5 cm × 0.5 cm squares of B. cinerea culture, and placed in the center of each plate. The plates were then sealed and stored at room temperature, and the minimum inhibitory concentration (MIC) was assessed at 7 days post inoculation. The visual presence/absence of inhibition was noted, and colony diameter was measured in centimeters across the widest axis using a standard ruler. The dose ranges were refined based on preliminary inhibition outcomes to efficiently assess the antifungal activity.
3. Results
Essential oils were analyzed by GC/MS to establish both the plant chemotype and essential oil quality. Prominent (> 5%) volatile compounds in cassia include (E)-cinnamaldehyde (61.0%) and (E)-o-methoxy cinnamaldehyde (20.3%); those in lemongrass include neral (30.6%), geraniol (7.6%), geranial (34.8%), and geranyl acetate (5.3%); those in oregano include p-cymene (11.8%), γ-terpinene (6.3%), and carvacrol (59.2%). The profiles of the essential oils of cassia, lemongrass, and oregano are presented in Table 1.
Variable doses of essential oils were tested [7] to evaluate the MIC of essential oils against B. cinerea. This was conducted in 10 μL increments until a minimum inhibitory concentration (MIC) was determined. Cassia, lemongrass, and oregano exhibited antifungal activity, with complete inhibition of B. cinerea growth occurring at doses (per 15 mL agar plate) of 50 μL (oregano), 60 μL (cassia), and 70 μL (lemongrass).
Table 1. Essential oil profiles of cassia (Cinnamomum cassia), lemongrass (Cymbopogon flexuosus), and oregano (Origanum vulgare).
Compound Name | Class | KI | Cassia | Lemongrass | Oregano |
α-pinene | Bicyclic monoterpene | 932 | 0.1 | 0.3 | 1.3 |
camphene | Bicyclic monoterpene | 946 | 0.1 | 1.6 | 0.3 |
benzaldehyde | Aromatic aldehyde/ benzenecarbaldehyde | 952 | 1.2 | nd | nd |
β-pinene | Bicyclic monoterpene | 974 | tr | tr | 1.1 |
6-methyl-5-hepten-2-one | Methyl ketone | 981 | nd | 1.5 | tr |
myrcene | Acyclic monoterpene | 988 | nd | nd | 1.0 |
α-terpinene | Cyclic monoterpene | 1014 | nd | nd | 1.5 |
p-cymene | Alkylbenzene/monocyclic monoterpene | 1020 | 0.1 | tr | 11.8 |
limonene | Cyclic monoterpene | 1024 | tr | 0.4 | 1.0 |
1,8-cineole | Bicyclic monoterpene | 1026 | nd | tr | 1.9 |
γ-terpinene | Monocyclic monoterpene | 1054 | nd | nd | 6.3 |
4-nonanone | Dialkyl ketone | *1067 | nd | 1.1 | nd |
linalool | Monoterpene alcohol | 1095 | nd | 1.2 | 3.1 |
borneol | Bicyclic monoterpene alcohol | 1165 | 0.1 | 0.3 | 1.0 |
neral | Acyclic monoterpene aldehyde | 1235 | nd | 30.6 | nd |
geraniol | Acyclic monoterpene alcohol | 1249 | nd | 7.6 | nd |
geranial | Acyclic monoterpene aldehyde | 1264 | nd | 34.8 | nd |
(E)-cinnamaldehyde | Cinnamaldehyde | 1267 | 61.0 | nd | nd |
thymol | Monoterpene phenol | 1289 | nd | nd | 3.0 |
carvacrol | Monoterpene phenol | 1298 | nd | nd | 59.2 |
geranyl acetate | Monoterpene acetate ester | 1379 | nd | 5.3 | nd |
(E)-caryophyllene | Bicyclic sesquiterpene | 1417 | 0.1 | 3.0 | 2.9 |
coumarin | 1,2-Benzopyrone | 1432 | 4.2 | nd | nd |
(E)-cinnamyl acetate | Cinnamyl ester | 1443 | 6.5 | nd | nd |
γ-cadinene | Bicyclic sesquiterpene | 1513 | 0.1 | 2.5 | tr |
(E)-o-methoxy cinnamaldehyde | Aromatic aldehyde/cinnamic Aldehyde/phenylpropanoid | 1527 | 20.3 | nd | nd |
Compounds detected in any sample at ≥ 1.0% are reported. Values less than 0.1% are denoted as trace (tr), and those not detected as nd. KI is the Kovats Index value and was previously calculated by Robert Adams using a linear calculation on a DB-5 column [17].
4. Discussion
Further distinguished from the plant species level, some plants are defined by chemotypes within the flavor and fragrance industries. These chemotypes are defined by the prominent volatile compounds produced by the plant, which are the basis of the antifungal activity of each essential oil. Prominent compounds for cassia (Cinnamomum cassia) were (E)-cinnamaldehyde (61.0%) and (E)-o-methoxy cinnamaldehyde (20.3%), for lemongrass (Cymbopogon flexuosus) were neral (30.6%) and geranial (34.8%), for oregano (Origanum vulgare) were thymol (3.0%) and carvacrol (59.2%). These volatile profiles are similar to those defined by the International Organization for Standardization (ISO) for cassia, lemongrass, and oregano essential oils [21-23].
The observed antifungal activity of cassia, lemongrass, and oregano is consistent with previous reports that oils largely composed of either phenols or aldehydes exhibit antifungal activity [7, 12, 15, 16]. Oregano oil, which was the most effective oil in this study, is largely composed of carvacrol, a monoterpenoid phenol known for its biological activities. Cassia oil is characterized by high concentrations of the phenylpropanoid (E)-cinnamaldehyde, a widely used antifungal agent, and was the second most effective essential oil against B. cinerea. Lemongrass was the third most effective and, similarly, possessed inhibitory effects that can be attributed to neral and geranial, compounds with reported antifungal activity. These three essential oils have recently been employed as key active ingredients in a patent-pending fungicide [24]. Using a natural solubilizer, fungicides can be formulated with hydrophobic essential oils as the key active ingredients, resulting in 100% natural plant-based ingredients [7].
Several techniques have been employed for investigating the antifungal activity of essential oils, including the direct application of essential oil mixtures to infected plant materials [25, 26], variations of the agar diffusion method [27, 28], or direct embedding of essential oils into agar mixtures prior to inoculation [7, 29]. The latter method typically employs a solubilizer to mix hydrophobic essential oils in an aqueous agar solution. Typically, research methods do not fully disclose how to homogenously mix these solutions while maintaining sterile conditions [7, 29]. The methodology detailed herein provides a reliable approach for homogenously mixing the solution so that (1) Petri dishes are kept sterile until inoculation and (2) potential fungal growth patterns are evenly distributed, both of which can prove difficult. Additionally, a vehicle control was employed to investigate any potential antifungal activity of the solubilizer (Tween 80). Vehicle control solutions were formulated at more than 10x the concentration of solubilizer (800 μL/15 mL agar) used in treatment studies, with no observed fungal growth inhibition at the highest concentrations.
The findings of this study highlight the pathogen-specific nature of essential oil activity and suggest that individual bioactive components target distinct cellular pathways. Incorporating essential oils to create diverse mechanisms of fungal growth inhibition in blended formulations may enhance overall efficacy and reduce the potential for resistance development in fungal pathogens.
5. Conclusions
The current study details the antifungal activity of cassia, lemongrass, and oregano essential oils against B. cinerea, a plant pathogen that causes gray mold. The plant chemotype and prominent antifungal compounds were determined by GC/MS analysis. Prominent (> 5%) volatile compounds in cassia include (E)-cinnamaldehyde (61.0%) and (E)-o-methoxy cinnamaldehyde (20.3%); those in lemongrass include neral (30.6%), geraniol (7.6%), geranial (34.8%), and geranyl acetate (5.3%); those in oregano include p-cymene (11.8%), γ-terpinene (6.3%), and carvacrol (59.2%). The antifungal activity of the essential oils was investigated by means of minimum inhibitory concentrations on PDA media directly embedded with varying concentrations of essential oils. Oregano demonstrated the highest antifungal activity (MIC 50 μL/15 mL), followed by cassia (MIC 60 μL/15), and lemongrass (70 μL/15 mL). These findings support research into natural alternatives for conventional fungicides.
Disclaimer (artificial intelligence)
Author(s) hereby state that no generative AI tools such as Large Language Models (ChatGPT, Copilot, etc.) and text-to-image generators were utilized in the preparation or editing of this manuscript.
Authors’ contributions
Conceptualization, I.P.L., T.M.W.; methodology, T.M.W., A.L..; software, T.M.W.; validation, C.R.B.; investigation and formal analysis, I.P.L., A.L.; resources, T.M.W., C.R.B.; data curation, I.P.L.; writing original draft, I.P.L., T.M.W.; review and editing draft, A.L., C.R.B.; funding acquisition, C.R.B.
Acknowledgements
The authors would like to acknowledge Zach Nielsen for the botanical illustration and extend their gratitude to the D. Gary Young Research Institute and Utah Valley University for supporting this research.
Funding
This research was funded by Young Living Essential Oils.
Availability of data and materials
All data will be made available on request according to the journal policy.
Conflicts of interest
The funders had no role in the design of the study; in the collection, analysis, or interpretation of the data; in the writing of the manuscript; or in the decision to publish the results. The authors declare no conflict of interest.
References
1. | Romanazzi, G.; Feliziani, E. Chapter 4 - Botrytis cinerea (Gray Mold), Postharvest Decay. Academic Press, Cambridge, MA, p. 131-146, 2014. https://doi.org/10.1016/B978-0-12-411552-1.00004-1 |
2. | Rosslenbroich, H.J.; Stuebler, D. Botrytis cinerea — history of chemical control and novel fungicides for its management, Crop Prot. 2000, 19, 557-561. https://doi.org/10.1016/S0261-2194(00)00072-7 |
3. | Islam, T.; Danishuddin; Tamanna, N.T.; Matin, M.N.; Barai, H.R.; Haque, M.A. Resistance mechanisms of plant pathogenic fungi to fungicide, environmental impacts of fungicides, and sustainable solutions. Plants. 2024, 13(19). https://doi.org/10.3390/plants13192737 |
4. | Hu, F.; Tu, X.F.; Thakur, K.; Hu, F.; Li, X.L.; Zhang, Y.S.; Zhang, J.G.; Wei, Z.J. Comparison of antifungal activity of essential oils from different plants against three fungi, Food Chem. Toxicol. 2019, 134, 110821. https://doi.org/10.1016/j.fct.2019.110821 |
5. | Lanier, C.; Richard, E.; Heutte, N.; Picquet, R.; Bouchart, V.; Garon, D. Airborne molds and mycotoxins associated with handling of corn silage and oilseed cakes in agricultural environment, Atmos. Environ. 2010, 44(16), 1980-1986. https://doi.org/10.1016/j.atmosenv.2010.02.040 |
6. | Nakajima, M.; Akutsu, K. Virulence factors of Botrytis cinerea. J. Gen. Plant Pathol. 2014, 80, 15–23. https://doi.org/10.1007/s10327-013-0492-0 |
7. | Wilson, T.M.; Laney, A.; Ruggles, Z.; Carlson, R.E. Essential oils as active ingredients in a plant-based fungicide: An in vitro study demonstrating growth inhibition of gray mold (Botrytis cinerea). Agrochemicals. 2025, 4(3), 11. https://doi.org/10.3390/agrochemicals4030011 |
8. | Adrian, M.; Jeandet, P.; Veneau, J.; Weston, L.A.; Bessis. R. Biological activity of resveratrol, a stilbenic compound from grapevines, against Botrytis cinerea, the causal agent for gray mold. J Chem. Ecol. 1997, 23, 1689–702. https://doi.org/10.1023/B:JOEC.0000006444.79951.75 |
9. | Elmer, P.A.G.; Reglinski, T. Bio suppression of Botrytis cinerea in grapes. Plant Pathol. 2006, 55(2), 155-177. https://doi.org/10.1111/j.1365-3059.2006.01348.x |
10. | Romanazzi, G.; Lichter, A.; Gabler, F.M.; Smilanick, J.L. Recent advances on the use of natural and safe alternatives to conventional methods to control postharvest gray mold of table grapes, Postharvest Biol. Technol. 2012, 63(1), 141-147. https://doi.org/10.1016/j.postharvbio.2011.06.013 |
11. | Blasco, C.; Picó, Y.; Mañes, J.; Font, G. Determination of fungicide residues in fruits and vegetables by liquid chromatography–atmospheric pressure chemical ionization mass spectrometry, J. Chromatogr. A 2002, 947(2), 227-235. https://doi.org/10.1016/S0021-9673(02)00009-2 |
12. | Nazzaro, F.; Fratianni, F.; Coppola, R.; Feo, V.D. Essential oils and antifungal activity. Pharmaceuticals. 2017, 10(4), 86. https://doi.org/10.3390/ph10040086 |
13. | Parikh, L.; Agindotan, B.O.; Burrows, M.E. Antifungal activity of plant-derived essential oils on pathogens of pulse crops. Plant Dis. 2021, 105, 6, 1692-1701. https://doi.org/10.1094/PDIS-06-20-1401-RE |
14. | Hyldgaard, M.; Mygind, T.; Meyer, R.L. Essential oils in food preservation: Mode of action, synergies, and interactions with food matrix components. Front. Microbiol. 2012, 3, 12. https://doi.org/10.3389/fmicb.2012.00012 |
15. | Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol. 2004, 94(3), 223–253. https://doi.org/10.1016/j.ijfoodmicro.2004.03.022 |
16. | Ahmad, A.; Khan, A.; Akhtar, F.; Yousuf, S.; Xess, I.; Khan, L.A.; Manzoor, N. Fungicidal activity of thymol and carvacrol by disrupting ergosterol biosynthesis and membrane integrity against Candida. FEMS Yeast Res., 2011, 11(7), 618–629. https://doi.org/10.1007/s10096-010-1050-8 |
17. | Adams, R.P. Identification of essential oil components by gas chromatography/mass spectrometry, 4th edn.; Allured Publishing: Carol Stream, IL, USA, 2007. |
18. | Asemaninejad, A.; Weerasuriya, N.; Gloor, G.B.; Lindo, Z.; Thorn, R.G. New primers for discovering fungal diversity using nuclear large ribosomal DNA. PLoS One. 2016, 11(7). https://doi.org/10.1371/journal.pone.0159043 |
19. | Huang, X.; Madan, A. CAP3: A DNA sequence assembly program. Genome Res. 1999, 9, 868-877. https://doi.org/10.1101/gr.9.9.868 |
20. | Sayers, E.W.; Bolton, E.E.; Brister, J.R.; Canese, K.; Chan, J.; Comeau, D.C.; Connor, R.; Funk, K.; Kelly, C.; Kim, S.; Madej, T.; Marchler-Bauer, A.; Lanczycki, C.; Lathrop, S.; Lu, Z.; Thibaud-Nissen, F.; Murphy, T.; Phan, L.; Skripchenko, Y.; Tse, T.; Wang, J.; Williams, R.; Trawick, B.W.; Pruitt, K.D.; Sherry, S.T. Database resources of the national center for biotechnology information. Nucleic Acids Res. 2022, 7(50). https://doi.org/10.1093/nar/gkaa892 |
21. | ISO 3216:1997. Oil of cassia, Chinese Type (Cinnamomum aromaticum Nees, syn. Cinnamomum cassia Nees ex Blume). ISO: Geneva, Switzerland, 1997. Available online: https://www.iso.org/standard/8420.html (accessed on 27 January 2026). |
22. | ISO 4718:2004. Oil of lemongrass [Cymbopogon flexuosus (Nees ex Steudel) J.F. Watson]. ISO: Geneva, Switzerland, 2004. Available online: https://www.iso.org/standard/32032.html (accessed on 27 January 2026). |
23. | ISO 13171:2016. Essential oil of oregano [Origanum vulgare L. subsp. hirtum (Link) letsw.]. ISO: Geneva, Switzerland, 2016. Available online: https://www.iso.org/standard/66202.html (accessed on 27 January 2026). |
24. | Carlson, R.E.; Wilson, T.M. Fungicidal compositions comprising essential oils. US Patent 2025/0176562 A1, 5 June 2025. |
25. | Ahmad, Z.; Abbas, H.; Murtaza, T.; Khan, A.U.R.; Ali, A.; Zahid, K.; Tahir, Z.; Mahmood, T.; Habib, A. Assessment of responses of peach cultivars to postharvest pathogen Botrytis cinerea and its mitigation using plant essential oils. Plant Prot. 2023, 7, 2. https://doi.org/10.33804/pp.007.02.4639 |
26. | Karakus, S.; Atıcı, O.; Turan, M.; Azizi, S.; Hajizadeh, H.S.; Kaya, O. Volatile organic compounds produced by some synthetic essential oils as biological fumigants against Botrytis cinerea on apples. Chem. Biol. Technol. Agric. 2023, 10, 136. https://doi.org/10.1186/s40538-023-00505-5 |
27. | Antuhu, Y.L.; Muanpuii, C.V.; Maisnam, R.; Kumari, A.; López-Menchero, J.R.; Coloma, A.G.; Andrés, M.F.; Kaushik, N. Assessing the efficacy of essential oil fumigation in mitigating Botrytis cinerea infection in cherry tomato. BIO Web Conf. 2024, 110, 02008. https://doi.org/10.1051/bioconf/202411002008 |
28. | Christova, P.K.; Dobreva, A.M.; Dzhurmanski, A.G.; Dincheva, I.N.; Dimkova, S.D.; Zapryanova, N.G. The Impact of plant essential oils on the growth of the pathogens Botrytis cinerea, Fusarium solani, and Phytophthora pseudocryptogea. Life. 2024, 14, 817. https://doi.org/10.3390/life14070817 |
29. | Xie, Y.; Zhu, J.; Liu, H.; Lian, H.; Liu, J. In vitro antifungal activity of essential oils against Botrytis cinerea of postharvest grapes. IOP Conf. Ser. Earth Environ. Sci. 2022, 1035, 012008. https://doi.org/10.1088/1755-1315/1035/1/012008 |
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
Abstract
Essential oils are commonly researched as alternative ingredients to conventional fungicides. The current study investigated the antifungal activity of essential oils with high concentrations of aldehydes (cassia and lemongrass) and phenols (oregano). Plant chemotypes and prominent antifungal compounds were determined by GC/MS analysis (reported as area %). Cassia oil contains (E)-cinnamaldehyde (61.0%) and (E)-o-methoxy cinnamaldehyde (20.3%); lemongrass oil contains neral (30.6%) and geranial (34.8%); oregano oil contains thymol (3.0%) and carvacrol (59.2%). Antifungal activity against Botrytis cinerea (cause of gray mold) was evaluated using a technique for embedding hydrophobic essential oils homogenously into water-based PDA agar media. Oregano demonstrated the highest antifungal activity (MIC 50 μL/15 mL), followed by cassia (MIC 60 μL/15 mL) and lemongrass (70 μL/15 mL). These findings support research into natural alternatives for conventional fungicides.
Abstract Keywords
Antifungal, Botrytis cinerea, cassia, essential oil, gray mold, lemongrass, oregano, gas chromatography.
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
Editor-in-Chief
This work is licensed under the
Creative Commons Attribution 4.0
License.(CC BY-NC 4.0).


