Research Article
Ygor Jessé Ramos*
Ygor Jessé Ramos*
Corresponding
Author
Botanical Garden
Research Institute of Rio de Janeiro, Jardim Botânico, Rio de Janeiro, RJ,
22460-030, Brazil.
And
Rio de Janeiro State University, Rio de Janeiro, RJ, Maracanã, 20550-013, Brazil.
E-mail: ygorjesse@jbrj.gov.br
Irene Candido Fonseca
Irene Candido Fonseca
Botanical Garden
Research Institute of Rio de Janeiro, Jardim Botânico, Rio de Janeiro, RJ,
22460-030, Brazil.
George Azevedo Queiroz
George Azevedo Queiroz
Botanical Garden
Research Institute of Rio de Janeiro, Jardim Botânico, Rio de Janeiro, RJ,
22460-030, Brazil.
Daniel de Brito Machado
Daniel de Brito Machado
Botanical Garden
Research Institute of Rio de Janeiro, Jardim Botânico, Rio de Janeiro, RJ,
22460-030, Brazil.
And
Rio de Janeiro State University, Rio de Janeiro, RJ, Maracanã, 20550-013, Brazil.
Claudete da Costa-Oliveira
Claudete da Costa-Oliveira
Botanical Garden Research Institute of Rio de
Janeiro, Jardim Botânico, Rio de Janeiro, RJ, 22460-030, Brazil.
Jéssica Regina Sales Felisberto
Jéssica Regina Sales Felisberto
Botanical Garden
Research Institute of Rio de Janeiro, Jardim Botânico, Rio de Janeiro, RJ,
22460-030, Brazil.
Elsie Franklin Guimarães
Elsie Franklin Guimarães
Botanical Garden
Research Institute of Rio de Janeiro, Jardim Botânico, Rio de Janeiro, RJ,
22460-030, Brazil.
Davyson de Lima Moreira*
Davyson de Lima Moreira*
Corresponding
Author
Botanical Garden
Research Institute of Rio de Janeiro, Jardim Botânico, Rio de Janeiro, RJ,
22460-030, Brazil.
And
Rio de Janeiro State University, Rio de Janeiro, RJ, Maracanã, 20550-013, Brazil.
E-mail: davysonmoreira@jbrj.gov.br
Received: 2023-01-24 | Revised:2023-02-13 | Accepted: 2023-02-17 | Published: 2023-02-24
Pages: 73-79
DOI: https://doi.org/10.58985/jeopc.2023.v01i02.11
Abstract
The aim of this study was to analyze the chemical composition of the
essential oil extracted from the leaves of the species Piper
robustipedunculum Yunck. The essential oil was obtained through
hydrodistillation and analyzed using GC-FID and GC-MS. The results showed that
93.88% of the total composition was composed of 24 compounds. The essential oil
was light yellow in color, with a yield of 0.68%, and was rich in
sesquiterpenes, including both oxygenated (37.09%), such as E-nerolidol
(13.60%), and non-oxygenated sesquiterpenes (45.06%), such as δ-cadinene
(10.72%), α-copaene (9.85%), and valencene (8.34%) as the major components.
This essential oil has the potential for industrial and pharmaceutical
applications, and can aid in future research in ecology, chemotaxonomy, and
chemophenetics related to the Piperaceae species.
Keywords
Aromatic
plant,
chemophenetic,
Piperaceae, E-nerolidol, hydro-distillation,
sesquiterpenes.
1. Introduction
Piperaceae
family belongs to the Piperales order, which is part of the Magnoliideas group.
This pantropical family encompasses approximately 4,100 plant species found
worldwide, with the largest genus being Piper L., containing around
2,000 species [1]. In Brazil, the highest
diversity of this genus is located in the Atlantic and Amazon Forests, housing
347 taxa out of the 466 present in Brazil [1]. The
Northeast region of Brazil records 93 species of this genus.
Several
well-known plants from the Piper genus, such as Black Pepper (Piper
nigrum L.), Long Pepper (Piper longum L.), and Kava-Kava (Piper
methysticum G. Forst), are commonly used as condiments and have various
therapeutic purposes [4, 5]. Ethnobotanical surveys
of medicinal plants emphasize the importance of the Piper genus for its
medicinal properties, including increased gastrointestinal motility,
antifungal, antibacterial, larvicidal, antinociceptive, and antihepatotoxic
activities. The popularity of its medicinal use is linked to the number of Piper
species available [6‑7].
Due to their
similar morphological features, species from this genus can often be found in
the presence of different ethnospecies, including Jaborandi (Piper amalago L., Piper crassinervium
HB & K., Piper mollicomum Kunth and Piper aduncum L.),
Pariparoba (Piper cernuum Vell., Piper mikanianum (Kunth) Steudel
and Piper umbellatum L.), White Betis (Piper rivinoides Kunth., Piper
arboreum Aubl. and Piper tuberculatum Jacq.), among others [8‑9].
The essential
oils extracted from various parts of the Piper genus have demonstrated
remarkable biological properties, including antioxidant, antibacterial,
antifungal, antiprotozoal, antiproliferative, anti-inflammatory, and
neuropharmacological activities [10-12, 7, 13]. This
diversity of activities can be attributed to the extensive chemical diversity
present in Piper's essential oils, which make them valuable tools for
chemophenetic and ecological characterization, particularly for new and
unstudied species. The presence of specialized secondary metabolites in these
oils provides important information for taxonomic descriptions and suit for
defining characteristic of a particular species [14].
Piper
robustipedunculum Yunck (Fig. 1) is a species native to Brazil,
restricted to the Northeastern states. It has morphological similarities with Piper
amplum Kunth, Piper arboreum Aubl. and Piper ilheusense Yunck
which are plants of popular use and coexist in the same region [15-18]. However, this species has no
phytochemical approach in the literature.
With all the above, this work aims to characterize the composition of the essential oil of Piper robustipedunculum Yunck from a natural population of the city of São Vicente Férrer, Pernambuco, Northeastern Brazil.
Figure 1. Habit
of Piper robustipedunculum Yunck.
2. Materials and methods
2.1 Plant material and isolation of
essential oil
Piper
robustipedunculum Yunck leaves were collected (three samples) on
October 22, 2019 at 9 am, in the municipality of São Vicente Férrer, at
coordinates 70 36’51” N and 350 30’ 53” E, at an
elevation of 352 m.d. An
exsiccate was identified and deposited in the Herbarium of the Botany
Department of the National Museum of the Federal University of Rio de Janeiro
under voucher number 45321. This study was assigned in the Brazilian Genetic
Heritage Management Council under the number AE4E953.
The hydrodistillation was the chosen
extraction technique, with Clevenger apparatus type, where 100 g of fresh
leaves was placed in a volumetric flask (2 L) with 700 mL of distilled water
and heated for 2 h. Yielding was calculated in relation to the obtained
essential oil (g) and the amount of fresh leaves (g). The essential oil was
kept in sealed amber flasks under refrigeration -20 oC until
analysis [12‑13].
2.2 Essential oil analysis
The chemical characterization and
quantification of P. robustipedunculum
essential oil was made by
gas chromatography coupled to mass spectrometry (GC-MS) and a gas
chromatography equipped with a flame ionization detector (FID), respectively.
Samples in triplicates were diluted in dichloromethane (HPLC grade, Tedia,
Brazil) before analysis (1 mg/mL).
A 1 μL of the solution was injected
into an HP Agilent GC 6890 coupled to Agilent MS 5973 series mass selective
detectors, splitless mode, wherein the injector temperatures were set at 270°C,
operating at 70 eV in positive mode. A HP-5MS (5%-phenyl-methylpolysiloxane) capillary
column [Agilent J & W; GC Columns (USA)] was used with 30 m x 0.25 mm i.d.
x 0.25 μm
particle size. Chromatography conditions were started at 60 - 240 °C at 3 °C /
min, totaling 60 min. For the separation of the constituents, helium
(~99.9999%) was used as a carrier gas at 1,0 mL/ min, at a rate of 1,0 s sweeps
and mass range of m/z 40 - 600 atomic
mass unit (u) [12‑13].
The GC-FID was achieved in a
chromatograph equipped with a flame ionization detector [HP- Agilent 6890
GC-FID]. A 1 μL of the solution were injected under the same analytical
conditions described above, except for the carrier gas used, which was hydrogen
at flow rate of 1,0 mL/min. The retention times (Rt) of the compounds were
measured in minutes and they were used to calculate their linear retention
index, obtained from the injection of a homologous series of hydrocarbons (C8-C25
de n-alkane, Sigma-Aldrich,
Brazil) under the same sample analytical condition [19‑20,
12‑13].
The mass spectra of the constituents
were compared with those from library (NIST, 98 e WILEY 7n) and with those
published in the literature [21]. Additionally,
when possible, the samples were analyzed in conjunction with authentic
standards (Sigma-Aldrich,
St. Louis, MS, USA).
3. Results and discussion
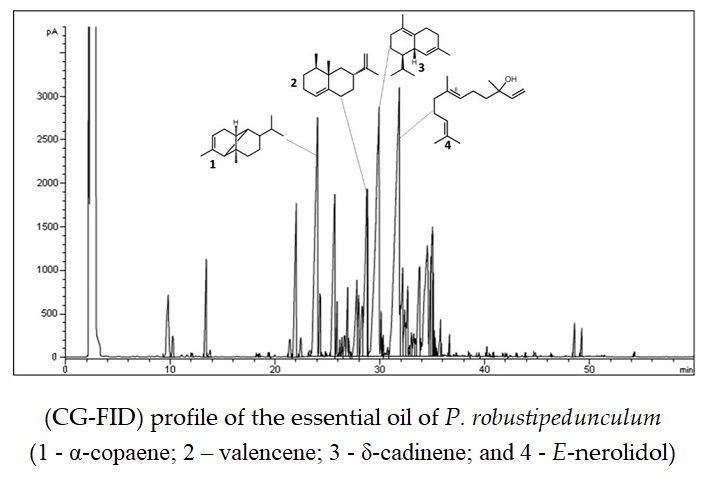
The essential oil (EO) obtained from the leaves of P. robustipedunculum, a native species and endemic to Northeastern Brazil, collected in the region of the Atlantic Forest and studied by GC-MS and GC-FID are presented in Table 1. A chromatographic profile of the essential oil is also presented in Fig. 2. EOs obtained by the same methods in other Piper species also showed high percentage of monoterpenes and sesquiterpenes in leaves and reproductive parts in all investigation periods [14].
Table
1. Aromatic profile of P. robustipedunculum essential
oil from leaves (n = 3) from
Pernambuco, Brazil.
|
RT |
Compounds1 |
RI* |
RI** |
Composition
(%) |
|
9.792 |
limonene |
1026 |
1024 |
1.05 |
|
13.440 |
Z-β-ocimene |
1034 |
1032 |
1.61 |
|
22.002 |
δ-elemene |
1323 |
1335 |
1.62 |
|
24.043 |
α-copaene |
1374 |
1374 |
9.85 |
|
25.679 |
E-caryophyllene |
1419 |
1417 |
4.75 |
|
26.916 |
α-humulene |
1455 |
1452 |
1.05 |
|
27.800 |
E-cadina-1(6),4-diene |
1476 |
1475 |
1.58 |
|
27.972 |
germacrene D |
1483 |
1480 |
2.43 |
|
28.302 |
β-selinene |
1490 |
1489 |
1.12 |
|
28.746 |
valencene |
1500 |
1496 |
8.34 |
|
28.816 |
γ-cadinene |
1515 |
1513 |
4.60 |
|
29.911 |
δ-cadinene |
1524 |
1522 |
10.72 |
|
31.163 |
E-nerolidol |
1560 |
1561 |
13.60 |
|
32.163 |
germacrene
D-4-ol |
1574 |
1574 |
5.16 |
|
32.359 |
gleenol |
1587 |
1586 |
1.02 |
|
32.649 |
viridiflorol |
1593 |
1592 |
1.54 |
|
33.229 |
1-epi-cubenol |
1628 |
1627 |
1.94 |
|
33.362 |
Z-cadin-4-en-7-ol |
1634 |
1635 |
1.04 |
|
33.752 |
epi-α-cadinol |
1640 |
1638 |
2.21 |
|
33.960 |
epi-α-muurolol |
1642 |
1640 |
2.17 |
|
35.010 |
α-muurolol |
1645 |
1644 |
2.12 |
|
34.519 |
α-cadinol |
1657 |
1652 |
5.29 |
|
35.796 |
tricosane |
2299 |
2300 |
3.98 |
|
48.573 |
octacosane |
2798 |
2800 |
4.09 |
|
Total identified |
93.88 |
|||
|
Compound classes (%) |
||||
|
Non-oxygenated
Monoterpene |
2.66 |
|||
|
Non-oxygenated
Sesquiterpene |
45.06 |
|||
|
Oxigenated
sesquiterpenes |
37.09 |
|||
|
Others
(hydrocarbons) |
8.07 |
|||
|
1Compounds an in order of elution on the HP-5MS column;
RT = Retention time in minutes; RI*=
Experimentally determined retention indices for C8 - C20
n-alkanes on the HP-5MS RI column; RI**= Literature Retention Indices [21], %= average compound concentration in the
three samples obtained. |
||||
Figure 2. Chromatographic profile of the flame ionization detector (CG-FID) of the essential oil of P. robustipedunculum from Pernambuco, Brazil. (See compound 1 - α-copaene; 2 – valencene; 3 - δ-cadinene; and 4 - E-nerolidol)
The EO showed light-yellow color and average yield of 0.68% (g/g). This is very important to industrial applications when compared to other commercial species [14, 22‑23]. It was possible to characterize 93.88% of the total EO. Sesquiterpenes (acyclic, monocyclic, bicyclic and tricyclic) were registered as the main fraction, with a total of 82.15%, being non-oxygenated 45.09% and oxygenates 36.09%. E-nerolidol (13.60%), δ-cadinene (10.72%), α-copaene (9.85%), and valencene (8.34%) were identified as major compounds. For Piperaceae species, the high content of sesquiterpenes is in accordance with literature data [24, 7]. The monoterpene class represent only 2.66% of the total. These similar variations of monoterpenes and sesquiterpenes have already been reported by Santos et al. [24] in Brazilian Atlantic Forest for species of the same genus, including Piper arboreum var. arboreum Yunck. and Piper goesii Yunck. The Piperaceae family is known for the frequent occurrence of ethnospecies, as previously mentioned.
Considering taxonomic approaches, in the subclate Isophyllon [25], the taxonomic classification group that belongs to P. robustipedunculum, it is possible to find species that in their EO from leaves compounds produced by acetate-mevalonate or methylerythriol-4-phosphate metabolic pathways.
In 1966, Yuncker [26] pointed out the ease of errors caused by the similarity in morphology among the species within the Isophyllon. Specifically, the medicinal plants Piper amplum Kunth, Piper arboretum Aubl., Piper ilheusense Yunck and Piper vicosanum Yunck are often confused with P. robustipedunculum due to their similar appearance, which can lead to accidental or intentional mislabeling in Northeast Brazil. This can be a problem for quality control and the correct use of these plants [9, 18].
Chemotaxonomically allow for the observation of variations in chemical compositions among species within this group across different studies in the literature. For example, for the EO of Piper ilheuense Yunck it was registered the compounds patchouli alcohol (11.10%), E-caryophyllene (11.80%), gleenol (7.50%) and δ-cadinene (6.90%) [16]. Pereira et al. [27] describe the chemical composition of EO from different species of Piper, in which, Piper amplum contained the iso-leptospermone (26.44%) and α-eudesmol (11.57%), while Piper vicosanum contained α-eudesmol (17.35%) and α-copaene (12.34%). Piper arboreum var. arboreum and Piper arboretum var. hirtelum had a major E-caryophyllene (13.71% and 13.22%) in common, but a significant difference (>5%) in the relative percentage of bicyclogermacrene (18.50 % and 5.32%) and α-cadinol (2.40% and 8.89%), respectively. Studies such as the one presented in this manuscript serve as a means to prevent adulteration and to describe the different chemophenetic phenomena of this species.
The majority of studies on EOs with a high content of E-nerolidol can be found in the Brazilian Atlantic Forest. For example, P. aduncum L. (80.6 – 82.5 % content) [28, 29]; P. claussenianum (Miq.) C. DC. (81.4 – 83.3% content) [30]; Piper diospyrifolium Kunth (10.0% content) [27] and P. gaudichaudianum Kunth (22.1% - 22.4% content) [31-32, 14]. Chan and co-workers [33] state that E-nerolidol is widely used in the industry, including in the production of cosmetics, food, and pharmaceutical products. Piper species that are abundant in E-nerolidol have already been demonstrated to exhibit a variety of biological functions, such as antileishmanial (promastigotes of Leishmania amazonensis, IC50 30.24 µg/ mL) and antifungal (Candida albicans, MIC 0.2 –1.26%) for P. claussenianum [30, 34-35]; and cytotoxic (V79 Chinese hamster lung cells, IC50 4.0 µg/ mL) for P. gaudichaudianum [31].
The compounds δ-cadinene, α-copaene, and valencene are described in the literature for their diverse biological activities when present in the essential oils of Piper [4-5, 7, 11, 14, 17]. These activities include antimicrobial, anti-inflammatory, antioxidant, and anticancer properties. Some studies also suggest that these compounds may possess analgesic and sedative properties, in addition to their use in the fragrance and flavor industry due to their pleasant aroma [14, 17, 22-23].
4. Conclusions
The essential oil of the P. robustipedunculum leaf can be a source of vegetable raw material for future employability in the pharmaceutical, cosmetic, food and pesticide industries, since it is sesquiterpene-rich. Also, the chemical composition of this endemic species from the Atlantic Forest of Northeast Brazil, a biome high threatened by human action, can assist in studying to understand ecological, chemotaxonomics and chemophenetics issues considering Piperaceae species.
Authors’ contributions
Conceptualization, G.A.Q.; Y.J.R., Methodology, Y.J.R., E.F.G., D.L.M.; Formal analysis, G.A.Q., D.B.M., C.C.O., J. R.S.F.; Investigation, Y.J.R., G.A.Q., I.C.F.; Resources, Y.J.R., D.B.M., C.C.O., J.R.S.F., G.A.Q., I.C.F.; Writing – original draft preparation, Y.J.R. and I.C.F.; Writing – review & editing, Y.J.R., I.C.F., D.L.M.
Funding
This research was funded by CNPq (Conselho Nacional de Pesquisas e Desenvolvimento Científico e Tecnológico e Inovação) - Brazil (PROEP n. 407845/2017); FAPERJ (Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro)—Brazil, (APQ1 n. NE26/210.245/2019), (CNE n. 201.211/2022).
Conflicts of interest
The authors declare no conflict of interest. The funding entity had no role in the design of the study, in the collection, analysis, or interpretation of data, in the writing of the manuscript, or in the decision to publish the results.
|
References |
|
|
1. |
Flora do
Brasil. Jardim Botânico do Rio de Janeiro. Available in <http://floradobrasil.jbrj.gov.br/>.
Acesso em 9 outubro 2022. |
|
2. |
Wanke, S.; Samain, M.S.;
Vanderschaeva, L.; Mathieu, G.; Goetghebeur, P.; Neinhuis, C. Phylogeny of
the genus Peperomia (Piperaceae) inferred from the trnk/matk region
(cpDNA). Plant. Biol. 2006, 8, 1, 93-102. https://doi.org/10.1055/s-2005-873060. |
|
3. |
Jaramillo, M.A.; Callejas, R.;
Davidson, C.; Smith, J.F.; Stevens, A.C.; Tepe, E.J. A phylogeny of the
tropical genus Piper using ITS and
the chloroplast intron psbJ–petA. Syst. Bot. 2008, 33, 4, 647-660. https://doi.org/10.1600/036364408786500244. |
|
4. |
Yadav, V.;
Krishnan, A.; Vohora, D.A systematic review on Piper longum L.: Bridging traditional knowledge and
pharmacological evidence for future translational research. J.
Ethnopharmacol. 2019, 247, 112255. https://doi.org/10.1016/j.jep.2019.112255. |
|
5. |
Takooree,
H.; Aumeeruddy, M.Z.; Rengasamy, K.R.; Venugopala, K.N.; Jeewon, R.; Zengin,
G.; Mahomoodally, M.F. A systematic review on black pepper (Piper nigrum L.): from folk uses to
pharmacological applications. Crit.
Rev. Food Sci. Nutr. 2019, 59, 210-243. https://doi.org/10.1080/10408398.2019.1565489. |
|
6. |
Pereira, L.A.; Lima, R.B.; Guimarães E.F.; Almeida, M.Z.; Monteiro,
E.D.C.Q.; Sobrinho, F.D.A.P. Plantas medicinais de uma comunidade quilombola
na Amazônia Oriental: Aspectos utilitários de espécies das famílias
Piperaceae e Solanaceae. Rev.
Bras. Agroecol. 2007, 2, 2, 1385-1388. |
|
7. |
Salehi, B.; Zakaria,
Z.A.; Gyawali, R.; Ibrahim, S.A.; Rajkovic, J.; Shinwari, Z.K.; Valussi, M. Piper species: A comprehensive review
on their phytochemistry, biological activities and applications. Molecules
2019, 24, 7, 1364. https://doi.org/10.3390/molecules24071364. |
|
8. |
Gogosz, A.M.; Boeger,
M.R.T.; Negrelle, R.R.B.; Bergo, C. Anatomia foliar comparativa de nove
espécies do gênero Piper
(Piperaceae). Rodriguésia. 2012, 63, 2, 405-417. https://doi.org/10.1590/S2175-78602012000200013. |
|
9. |
Silva, R.J.F.; Faial,
K.D.C.F.; Mendonça, M.S.D. Pharmacognostical characterization of Piper arboreum var. arboreum and P. tuberculatum (Piperaceae). Acta Amaz. 2016, 46, 2, 195-208. https://doi.org/10.1590/1809-4392201504422. |
|
10. |
Moreira,
D.L.; Souza, P.O.; Kaplan, M.A.C.; Pereira, N.A.; Cardoso, G.L.; Guimarães,
E.F. Effect of leaf essential oil from Piper
solmsianum C. DC. in mice behaviour. An.
Acad. Bras. Ciênc.
2001, 1, 73-78. https://doi.org/10.1590/S0001-37652001000100004. |
|
11. |
Scott,
I.M.; Jensen, H.R.; Philogène, B.J.; Arnason, J.T. A review of Piper spp. (Piperaceae)
phytochemistry, insecticidal activity and mode of action. Phytochem.
2008, 7, 1, 65-75. https://doi.org/10.1007/s11101-006-9058-5. |
|
12. |
Oliveira, G.L.; Cardoso, S.K.;
Lara-Junior, C.R.; Vieira, T.M.; Guimarães, E.F.; Figueiredo, L.S.; Moreira,
D.L.; Kaplan, M.A.C. Chemical study and larvicidal activity against Aedes aegypti of essential oil of Piper aduncum L. (Piperaceae). An. Acad. Bras.
Ciênc. 2013, 85, 4, 1227-1234. https://doi.org/10.21577/1984-6835.20190107. |
|
13. |
Ramos, Y.J.; Moreira,
D.L. Seasonal Study of Essential Oil from Aerial Parts of Peperomia
galioides Kunth (Piperaceae). Rev. Virtual
Quim. 2019, 11, 5, 1540-1550. https://doi.org/10.21577/1984-6835.20190107. |
|
14. |
Ramos,
Y.J.; Gouvêa-Silva, J.G.; Brito Machado, D.; Felisberto, J.S.; Pereira, R.
C.; Sadgrove, N.J.; Moreira, D.L. Chemophenetic and Chemodiversity
Approaches: New Insights on Modern Study of Plant Secondary Metabolite
Diversity at Different Spatiotemporal and Organizational Scales. Rev. Bras. Farmacog. 2022, 1-24. https://doi.org/10.1007/s43450-022-00327-w. |
|
15. |
Guimarães,
E.F.; Monteiro, D. Piperaceae of the Biological Reserve of Poço das Antas,
Silva Jardim, Rio de Janeiro, Brazil. Rodriguésia 2006,
57, 3, 569-589. https://doi.org/10.1590/2175-7860200657312. |
|
16. |
Oliveira,
R.A.; Assis, A.M.A.D.; Silva, L.A.M.; Andrioli, J.L.; Oliveira, F.F. Chemical
profile and antimicrobial activity of essential oil of Piper ilheusense. Chem. Nat. Comp. 2016, 52, 2, 331-333. https://doi.org/10.1007/s10600-016-1634-3. |
|
17. |
Silva, J.K.;
Trindade, R.; Alves, N.S.; Figueiredo, P.L.; Maia, J.G.S.; Setzer W.N.
Essential oils from neotropical Piper species
and their biological activities. Int. J. Mol. Sci. 2017, 18, 12, 2571. https://doi.org/10.3390/ijms18122571. |
|
18. |
Queiroz, G.A.; Guimarães, E.F.; Sakuragui, C.M. First record of Piper
robustipedunculum Yunck. (Piperaceae) in the state of Pernambuco, Brazil. Check List. 2020, 16, 1149. https://doi.org/10.15560/16.5.1149. |
|
19. |
Dool, H.V.D.; Kratz, P.A. Generalization of the retention index
system including linear temperature programmed gas-liquid partition
chromatography. J. Chromatogr. 1963,1,
463-471. https://doi.org/10.1016/ s0021-9673(01)80947-x. |
|
20. |
Ramos,
Y.J.; Brito Machado, D.; Queiroz, G.A.; Guimarães, E.F.; Defaveri, A.C.A.;
Moreira, D. L. Chemical composition of the essential oils of circadian rhythm
and of different vegetative parts from Piper mollicomum Kunth-A
medicinal plant from Brazil. Biochem.
Syst. Ecol. 2020, 92, 104116. https://doi.org/10. 1016/j.bse.2020.104116. |
|
21. |
Adams, R.P. Identification of essential oil components
by gas chromatography/mass spectroscopy. Ed. IV; Allured Bus. Mul,
Carol Stream: Illinois, USA, 2009. |
|
22. |
Filly, A.; Fernandez, X.; Minuti, M.;
Visinoni, F.; Cravotto, G.; Chemat, F. Solvent-free microwave extraction of
essential oil from aromatic herbs: from laboratory to pilot and industrial
scale. Food
Chem. 2014, 150 1,
193-198. https://doi.org/10.1016/j.foodchem.2013.10.139. |
|
23. |
Kemprai, P.; Bora, P.K.; Mahanta,
B.P.; Sut, D.; Saikia, S.P.; Banik, D.; Haldar, S. Piper betleoides C. DC.: Edible source of betel‐scented sesquiterpene‐rich
essential oil. Flavour Fragr. J. 2020, 35, 1, 70-78. https://doi.org/10.1002/ffj.3537. |
|
24. |
Santos, P.R.D.;
Moreira, D.; Guimarães, E.F.; Kaplan, M.A.C. Essential oil analysis of 10
Piperaceae species from the Brazilian Atlantic forest. Phytochem. 2001, 58,
4, 547-551. https://doi.org/10.1016/s0031-9422(01)00290-4. |
|
25. |
Miquel,
F.A.G. Systema Piperacearum.
Kramer, Rotterdam, 1843–1844. |
|
26. |
Yuncker, T.G. New species of
Piperaceae from Brazil. Bol. Inst. Bot. 1966, 3, 1-370. |
|
27. |
Pereira,
R.A.; Ramos Y.J.; Queiroz, G.A.; Guimarães E.F.; Defaveri, A.C.A.; Moreira,
D.L. Chemodiversity of Essential Oils in Piper L.(Piperaceae) Species
from the Restinga of Marambaia Island, Rio de Janeiro-RJ, Brazil. Rev. Virtual Quím. 2021, 13, 5, 1203-1215. https://doi.org/10.21577/1984-6835.20210067. |
|
28. |
Ceole, L.F; Cardoso, M.D; Soares,
M.J. Nerolidol, the main constituent of Piper aduncum essential oil,
has anti-Leishmania braziliensis activity. Parasitol. 2017, 144. 9,1179-1190. https://doi.org/10.1017/S0031182017000452. |
|
29. |
Villamizar, L.H.; Cardoso, M.D.; de
Andrade, J.; Teixeira, M.L.; Soares, M.J. Linalool, a Piper aduncum essential oil component, has selective activity
against Trypanosoma cruzi trypomastigote forms at 4 ◦C. Mem. Inst.
Oswaldo Cruz. 2017,
112, 131–139. https://doi.org/10.1590/0074-02760160361. |
|
30. |
Marques, A.M.; Barreto, A.L.S.;
Batista, E.M.; Curvelo, J.A.R.; Velozo, L.S.M.; Moreira, D.L.; Guimarães,
E.F.; Soares, R.M.A.; Kaplan, M.A.C. Chemistry and biological activity of
essential oils from Piper claussenianum
(Piperaceae). Nat. Prod. Commun. 2010, 5, 1837–1840. https://doi.org/10.1177/1934578X1000501131. |
|
31. |
Péres, V.F.; Moura, D.J.; Sperotto,
A.R.M.; Damasceno, F.C.; Caramão, E.B.; Zini, C.A.; Saffi J. Chemical
composition and cytotoxic, mutagenic and genotoxic activities of the
essential oil from Piper
gaudichaudianum Kunth leaves. Food Chem. Toxicol. 2009, 47, 2389–2395. https://doi.org/10.1016/j.fct.2009.06.035. |
|
32. |
Sperotto, A.R.M.; Moura, D.J.; Péres,
V.F.; Damasceno, F.C.; Caramão, E.B.; Henriques, J.A.P.; Saffi, J. Cytotoxic
mechanism of Piper gaudichaudianum Kunth
essential oil and its major compound nerolidol. Food Chem. Toxicol. 2013, 57, 57–68. https://doi.org/10.1016/j.fct.2013.03.013. |
|
33. |
Chan, W.K.; Tan, L.T.H; Chan, K.G.;
Lee, L.H.; Goh, B.H. Nerolidol: A Sesquiterpene Alcohol with Multi-Faceted
Pharmacological and Biological Activities. Molecules. 2016,
21, 5, 529. https://doi.org/10.3390/ molecules21050529. |
|
34. |
Marques, A.M.; Barreto, A.L.S.;
Curvelo, J.A.D.R.; Romanos, M.T.V.; Soares, R.M.D.A.; Kaplan, M.A.C.
Antileishmanial activity of nerolidol-rich essential oil from Piper claussenianum. Rev. Bras. Farmacogn. 2011, 21, 5, 908-914. https://doi.org/10.1590/S0102-695X2011005000157. |
|
35. |
Curvelo, J.A.R.; Marques, A.M.;
Barreto, A.L.S.; Romanos, M.T.V.; Portela, M.B.; Kaplan, M.A.C.; Soares,
R.M.A.; A novel nerolidol-rich essential oil from Piper claussenianum modulates Candida
albicans biofilm. J. Med.
Microbiol. 2014. 63, 5,
697–702. https://doi.org/ 10.1099/jmm.0.063834-0. |
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
Abstract
The aim of this study was to analyze the chemical composition of the
essential oil extracted from the leaves of the species Piper
robustipedunculum Yunck. The essential oil was obtained through
hydrodistillation and analyzed using GC-FID and GC-MS. The results showed that
93.88% of the total composition was composed of 24 compounds. The essential oil
was light yellow in color, with a yield of 0.68%, and was rich in
sesquiterpenes, including both oxygenated (37.09%), such as E-nerolidol
(13.60%), and non-oxygenated sesquiterpenes (45.06%), such as δ-cadinene
(10.72%), α-copaene (9.85%), and valencene (8.34%) as the major components.
This essential oil has the potential for industrial and pharmaceutical
applications, and can aid in future research in ecology, chemotaxonomy, and
chemophenetics related to the Piperaceae species.
Abstract Keywords
Aromatic
plant,
chemophenetic,
Piperaceae, E-nerolidol, hydro-distillation,
sesquiterpenes.
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
Editor-in-Chief
This work is licensed under the
Creative Commons Attribution 4.0
License.(CC BY-NC 4.0).


