Research Article
Thaís Aparecida dos Santos Oliveira
Thaís Aparecida dos Santos Oliveira
Faculdade de
Filosofia, Ciências e Letras de Ribeirão Preto, Universidade de São Paulo,
Ribeirão Preto, SP, Brazil.
Jackson Breno Amaral Silva
Jackson Breno Amaral Silva
Faculdade de
Filosofia, Ciências e Letras de Ribeirão Preto, Universidade de São Paulo,
Ribeirão Preto, SP, Brazil.
Julia Grassi Barco
Julia Grassi Barco
Faculdade de
Filosofia, Ciências e Letras de Ribeirão Preto, Universidade de São Paulo,
Ribeirão Preto, SP, Brazil.
Milton Groppo
Milton Groppo
Faculdade de
Filosofia, Ciências e Letras de Ribeirão Preto, Universidade de São Paulo,
Ribeirão Preto, SP, Brazil.
Sara Lemes de Souza
Sara Lemes de Souza
Instituto de
Ciências Biomédicas, Universidade Federal de Uberlândia, Uberlândia, MG,
Brazil.
Carlos Henrique Gomes Martins
Carlos Henrique Gomes Martins
Instituto de
Ciências Biomédicas, Universidade Federal de Uberlândia, Uberlândia, MG,
Brazil.
Antônio Eduardo Miller Crotti*
Antônio Eduardo Miller Crotti*
Corresponding
Author
Faculdade de Filosofia, Ciências e Letras de Ribeirão Preto, Universidade de São Paulo, Ribeirão Preto, SP, Brazil.
E-mail: millercrotti@ffclrp.usp.br, Tel.: +(16) 3515-3747
Received: 2022-12-13 | Revised:2023-01-23 | Accepted: 2023-01-23 | Published: 2023-01-30
Pages: 32-38
DOI: https://doi.org/10.58985/jeopc.2023.v01i02.06
Abstract
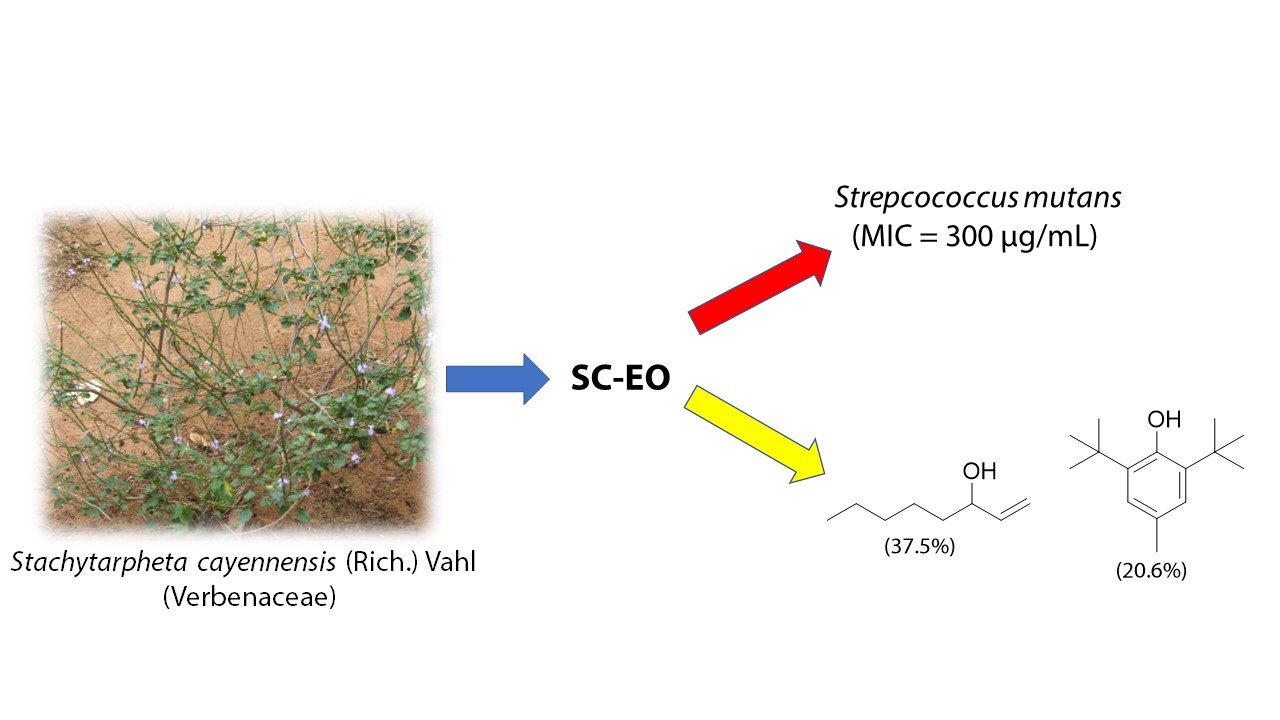
The
antibacterial activity of the essential oil from the leaves of Stachyterpheta
cayenensis (Verbenaceae) (SC-EO) grown in Brazil against a panel of cariogenic
bacteria was investigated using the broth microdilution method. SC-EO displayed
strong activity against Streptococcus mutans, S. mitis, S. salivarus, and S.
sanguinis, with minimal inhibitory concentration (MIC) values of 300, 300, 400,
and 400 µg/mL, respectively. Oct-1-en-3-ol (37.8%), 2,6-di-tert-butyl-4-methylphenol
(20.9 to 20.6%), (E)-phytol (8.6 to 8.3%), eugenol (5.1 to 4.8%), and borneol
(4.9 to 4.6 %) were identified as the main chemical compounds of SC-EO. To the
best of our knowledge, this is the first report on the antibacterial activity
of the essential oil of S. cayenensis.
Keywords
Coast redwood,
heartwood, sap-wood, monoterpene enantiomers, α-pinene, fatty alcohol, diterpenoid.
1. Introduction
Dental caries is a major public health problem worldwide. It is characterized by the demineralization of the tooth's hard tissues, cavitation, and tooth loss [1]. This pathology is associated with acidogenic and aciduric bacteria that adhere to the tooth surface and form a structurally and functionally organized biofilm (dental plaque) [2]. Biofilm removal by brushing and flossing is the most efficient procedure to prevent dental caries [3]. However, most people fail to control biofilm at a sufficient level through mechanical removal only. Therefore, the use of chemicals incorporated into mouth rinses as a complementary measure is also necessary and has been demonstrated to be of great value to diminish the tooth surface biofilm [3]. Currently, chlorhexidine (CHX) is considered to be the gold anti-cariogenic standard [4], however, the regular use of oral care products containing this chemical is often associated with several side effects [5]. In this scenario, the search for new potential chemotherapeutic agents that can be incorporated into dental products has escalated in recent years.
Essential oils (EOs) are mixtures of volatile compounds produced by plants as part of their secondary metabolism [6]. These compounds are mainly monoterpenes, sesquiterpenes, and phenylpropanoids, which can occur as functionalized derivatives such as alcohols, ketones, aldehydes, and esters [7]. EOs and their compounds are known to display antibacterial activities against a wide range of bacteria, including cariogenic bacteria [8, 9]. However, the antibacterial activity of EOs is related to their chemical composition, which depends on intrinsic (e.g., genetics and morphology) and extrinsic (e.g., humidity, soil composition, stage of the vegetative cycle, geographic variations, and seasonal variations) factors [10]. Thus, it is crucial to determine the EO composition each time it is obtained from a plant collected in different places or conditions [11-13].
Stachytarpheta cayennensis (Rich.) Vahl (Verbenaceae) is an herbaceous plant commonly found in tropical and subtropical America. In Brazil, S. cayennensis is popularly known as “gervão roxo”, “vassourinha de botão” or “gervão do campo” [14]. The infusion and decoction of its leaves are used in folk medicine to treat malaria, inflammation, pain, fever, hypertension, stress, diabetes, constipation, and hepatic and renal disorders [14]. Recently, the popular use of S. cayennensis for the treatment of COVID-19 in Jamaica has been also reported [15]. In the literature, extracts of S. cayennensis have been reported to display anti-inflammatory [16, 17], antinociceptive [17], antiulcero-genic [16], hypoglycemic [18], immunomodulatory [19], sedative and anxiolytic [20], anticancer [21], anti-malarial [22], antileishmanial [23], and antimicrobial [16, 24, 25]. However, data on the antimicrobial activities of the essential oil of S. cayennensis are still scarce [25, 26].
As part of our ongoing research on the antibacterial activities of essential oils (EOs) and other natural products as potential leads for incorporation into dental products [27-30], here we report the antibacterial activity of the essential oil from Stachytarpheta cayennensis leaves grown in Brazil Southeast against a panel of cariogenic bacteria. The chemical composition of the essential oil is also reported.
2. Materials and methods
2.1 Plant material, EO extraction, and Gas chromatography-mass spectrometry (GC-MS) analysis
Stachytarpheta cayennensis (Rich.) Vahl (Verbenaceae) was collected at “Sítio 13 de Maio” near the city of Franca (20o26’S 47o27’W 977 m, State of São Paulo, Brazil) in April 2021. A voucher specimen (SPFR 10005) was deposited at the Herbarium of Departamento de Biologia, Faculdade de Filosofia, Ciências e Letras de Ribeirão Preto, Universidade de São Paulo, São Paulo, Brazil (Herbarium SPFR).
Fresh leaves (1,200 g) of S. cayennensis were divided into three samples (3 x 400 g) and accommodated into three 1-L round bottom flasks containing 500 mL distilled water each. The flasks were coupled to a Clevenger-type apparatus and submitted to hydrodistillation for four hours. After the manual collection of the essential oil of S. cayennensis (SC-EO), traces of water were removed by freezing the collected sample below 0oC, followed by the transfer of the unfrozen SC-EO to a new vial. The SC-EO yield (w/w) was calculated from the weight of the fresh leaves.
SC-EO was diluted in ethyl ether (Sigma-Aldrich, St. Louis, MO, USA) at a concentration of 1.0 mg/mL and analyzed by gas chromatography-mass spectrometry (GC-MS) on a Shimadzu QP2010 Plus (Shimadzu Corporation, Kyoto, Japan) system equipped with an OAC-20i autosampler under the previously reported conditions [30]. The column temperature was programmed to increase from 60oC to 240oC at 3oC/min and held at 240oC for 5 min [30]. Helium (99.999%) was used as carrier gas at a flow rate of 1.0 mL/min. The injector and ion-source temperatures were set at 240oC and 280oC, respectively. The injection volume was 0.1 µL and a split ratio of 1:10 was used. The mass spectrometer operated in the electron ionization (EI) mode at 70 eV. Mass spectra were recorded with a scan interval of 0.5 s over the mass range of 40-600 Da. Quantification of each SC-EO constituent was estimated from GC-MS chromatograms by internal normalization (%) and expressed as the average of three replicates. The chemical compounds of SC-EO were identified on the basis of their retention indices (RI) on an Rtx-5MS (30 m x 0.25 mm x 0.25 µm) capillary column (Restek Co., Bellefonte, PA, USA) relative to a homologous series of n-alkanes (C8-C20) under the same operating conditions as well as computer matching with the Wiley 7, NIST 08 and FFNSC 1.2 spectral libraries. Moreover, the constituents were determined by comparison of their mass spectra with those reported in the literature.
2.2. Bacterial strains and antimicrobial assays
The minimum inhibitory concentration (MIC) values of SC-EO were determined by using the broth micro-dilution method in 96-well microplates. The following standard strains from the ATCC were employed: Streptococcus mutans (ATCC 25175), Streptococcus mitis (ATCC 49456), Streptococcus salivarius (ATCC 25975), Streptococcus sanguinis (ATCC 10556), Streptococcus sobrinus (ATCC 33478), Enterococcus faecalis (ATCC 4082), and Lactobacillus casei (ATCC 11578). Individual 24-hour colonies from blood agar (Difco Labs, Detroit, MI, USA) were suspended in 10.0 mL tryptic soy broth (Difco Labs, Detroit, MI, USA). Suspensions of each microorganism were standardized as previously described [31]. Samples of SC-EO were dissolved in DMSO (Merck, Darmstadt, Germany) and diluted in tryptic soy broth (Difco Labs, Detroit, MI, USA) to achieve concentrations in the range of 400 to 20 µg/mL. The final DMSO concentration was 5% (v/v), and this solution was used as the negative control. One inoculated well was included to control the adequacy of the broth for organism growth. One non-inoculated well free of the antimicrobial agent was also included to ensure medium sterility. Chlorhexidine dihydrochloride (CHD) (Sigma-Aldr-ich, St. Louis, MO, USA) was dissolved in tryptic soy broth (Difco Labs, Detroit, MI, USA) and used as a positive control at concentrations ranging from 59 to 0.115 µg/mL. The microplates (96 wells) were sealed with plastic film and incubated at 37oC for 24 h as described above. After that, 30 µL of 0.02% resazurin (Sigma-Aldrich, St. Louis, MO, USA) aqueous solution was poured into each microplate reservoir, to indicate microorganism viability. The MIC values were determined as the lowest concentration of SC-EO capable of inhibiting microorganism growth [32].
3. Results and discussion
The antibacterial activity of EOs and their constituents have been extensively reported and reviewed in the literature [8, 9, 33]. Many EOs have displayed promising activities against cariogenic bacteria, such as those obtained from Cinnamomum verum [34], Plectranthus neochilus [35], and Salvadora persica [36].
Recently, studies on the combination between EOs and herbal toothpastes have also been carried out [37]. On the other hand, a few studies have reported the antibacterial activity of the essential oil of S. cayennensis. Onofre and coworkers obtained the EO of S. cayennensis grown in Brazil Southwest and assessed its antibacterial activity against the pathogenic bacteria Escherichia coli ATCC-25922, Staphylococcus aureus ATCC-25923 and Pseudomonas aeruginosa ATCC-9027 using the agar diffusion method [25]. Duarte and coworkers investigated the antibacterial activity of the EO of S. cayennensis grown in Brazil Southeast against different Eschericia coli serotypes [26]. However, to date, the antibacterial activity of the EO of S. cayennensis against cariogenic bacteria has not been reported.
The antibacterial activity of SC-EO against a representative panel of cariogenic bacteria was investigated in terms of their MIC values (Table 1).
Table 1. In vitro antibacterial activity (MIC, in µg/mL) of the essential oil of S. cayenensis (SC-EO).
Microorganism | SC-EO | CHD |
Streptococcus mutans | 300 | 0.92 |
Streptococcus mitis | 300 | 0.36 |
Streptococcus sanguinis | >400 | 0.36 |
Streptococcus salivarus | 400 | 0.92 |
Streptococcus sobrinus | 400 | 0.92 |
Lactobacillus casei | >400 | 0.18 |
Enterococcus faecalis | >400 | 0.36 |
CHD: chlorhexidine dihydrochloride. The assays were performed in three replicates assays for each microorganism. | ||
SC-EO was not active against S. sanguinis, L. casei, and E. faecalis in the range of concentrations tested in this study (MIC > 400 µg/mL). On the other hand, SC-EO displayed activity against S. mutans (MIC = 300 µg/mL), S. mitis (MIC = 300 µg/mL), S. sobrinus (MIC = 400 µg/mL), and S. salivarus (MIC = 400 µg/mL). According to the literature [8], these MIC values denote a strong antibacterial activity against cariogenic bacteria. This is also an interesting result because very few natural compounds are known to inhibit S. mutans, which is one of the primary causative agents of dental caries [38].
SC-EO was obtained in 0.10% (w/w) yield. The chemical composition of SC-EO was determined by GC-MS. A representative GC-MS chromatogram of SC-EO is shown in Fig. 1.
Figure 1. Representative GC-MS chromatogram of SC-EO.
A total of thirteen compounds were identified, as listed in Table 2. The major compounds were identified as being oct-1-en-3-ol (37.8 to 37.5 %), 2,6-di-tert-butyl-4-methylphenol (20.9 to 20.6 %), (E)-phytol (8.6 to 8.3 %), eugenol (5.1 to 4.8 %), and borneol (4.9 to 4.6%). This chemical composition significantly differs from the chemical composition reported by Lima and coworkers for the EOs isolated from S. cayennensis leaves grown in Brazil North [39]. The authors identified methyl 7-methylcyclopentan[c]pyran-4-carboxylate (48.4%) and citronellol (89.5%) as the major compounds in the EOs isolated by hydrodistillation followed by extraction with ethyl acetate and hexane, respectively [39]. Moreover, none of the compounds identified in SC-EO were previously reported by the authors. α-Pinene and oct-1-en-3-ol were detected as minor compounds in the EO of Stachytarpheta gesnerioides grown in Brazil Southeast, but its major compound – the sesquiterpene guaiol (56.5%) was not detected in SC-EO [40]. Differences in the EO composition can be due to geographic factors, which affect growth conditions, climate, altitude, and soil type, as well as to agricultural methods and practices, developmental stage, and harvesting time [41]. It is well-established that the antibacterial activity of EOs cannot be assignable to a single mechanism of action only [6].
Table 2. Chemical composition of the essential oil from Stachytarpheta cayennensis leaves (SC-EO).
Compound | RT (min) | RIexp | RIlit | %RA |
α-Thujene | 5.78 | 930 | 931 | 0.9 |
α-Pinene | 5.98 | 937 | 939 | 3.6 |
1-Octen-3-ol | 7.11 | 977 | 978 | 37.5 |
Octan-1-ol | 7.60 | 995 | 993 | 2.0 |
Limonene | 8.73 | 1031 | 1031 | 2.0 |
Eucalyptol | 8.83 | 1034 | 1033 | 3.6 |
Borneol | 13.76 | 1163 | 1165 | 4.6 |
Cinnamaldehyde | 14.00 | 1263 | 1266 | 1.2 |
p-Vinyl-phenol | 18.80 | 1305 | 1309 | 3.9 |
Eugenol | 22.40 | 1355 | 1356 | 4.8 |
(E)-Caryophyllene | 24.69 | 1417 | 1418 | 2.2 |
2,6-di-tert-Butyl-4-methylphenol | 28.50 | 1526 | 1527 | 20.6 |
(E)-Phytol | 44.48 | 1947 | 1949 | 8.3 |
Monoterpene hydrocarbons |
|
|
| 6.5 |
Oxygenated monoterpenes |
|
|
| 8.2 |
Sesquiterpene hydrocarbons |
|
|
| 2.2 |
Oxygenated diterpenes |
|
|
| 8.3 |
Phenylpropanoids |
|
|
| 6.0 |
Others |
|
|
| 64.0 |
Not identified |
|
|
| 4.8 |
RT: retention time in an Rtx-5MS column; RIexp: retention indices relative to n-alkanes C8-C20 on Rtx-5MS capillary column; RIlit: retention indices from the literature (https://webbook.nist.gov/); RA: relative area (peak area relative to the total peak area in the GC-MS chromatogram), an average of three replicates. | ||||
In Gram-positive bacteria, lipophilic EO components can easily diffuse across the cell wall and act upon they are within the cytoplasm [8]. Indeed, the anti-cariogenic potential of some SC-EO compounds has been demonstrated. The anti-biofilm activity and the inhibitory effect of β-caryophyllene on biofilm have been recently demonstrated [42]. Eugenol was reported to inhibit the acid production by S. mutans, reduce the synthesis of water-insoluble glucans by glucosyltransferases, and suppress the adherence of S. mutans to saliva-coated hydroxyapatite beads, besides reducing the incidence and severity of carious lesions in rats [43]. Limonene significantly reduces caries lesions caused by S. sobrinus as compared to the control group [44]. However, the antibacterial activity of SC-EO cannot be assigned to the activity of its compounds only, since possible additive or synergistic interactions between the SC-EO compounds can also occur. These interactions are beneficial for antibacterial activity due to the difficulties bacteria have in developing resistance to them [45].
4. Conclusions
SC-EO displayed strong activity against S. mutans, S. mitis, S. salivarus, and S. sobrinus, with MIC values of 300, 300, 400, and 400 µg/mL, respectively. The chemical composition of SC-EO significantly differs from previous reports in the literature, with the predominance of 1-octen-3-ol and 2,6-di-tert-butyl-4-methylphenol, which were not detected in the EO isolated from other S. cayennensis specimens. This is the first report on the antibacterial activity of the essential oil from the leaves of S. cayennensis up to date and highlights an interesting anti-cariogenic potential that could be further exploited for the development of new oral care products.
Authors’ contributions
Conceptualization, A.E.M.C., C.H.G.M.; Methodology, T.A.S.O, J.B.A.S., J.G.B.; Investigation, T.A.S.O, J.B.A.S., J.G.B., S.M.S., M.G; Writing – original draft preparation, T.A.S.O., J.B.A.S., J.G.B.; Writing – review & editing, A.E.M.C., C.H.G.M.; Supervision, A.E.M.C., C.H.G.M.; Funding acquisition, A.E.M.C., C.H.G.M.
Acknowledgements
The authors thank the Brazilian foundations FAPESP for the financial support, and CNPq for fellowships.
Funding
This work was supported by the Fundação de Amparo à Pesquisa do Estado de São Paulo [grant number 2007/54241-8].
Conflicts of interest
The authors declare no conflict of interest.
References
1.
Karadaglioglu, O.I.; Alagoz, L.G.; Herbal
products against dental caries. Ann.
Dent. Spec. 2020, 8, 55-61.
2.
Yuan, J.; Yuan, W.Q.; Guo, Y.Y.; Wu, Q.;
Wang, F. Xuan, H.Z. Anti-biofilm activities of Chinese poplar propolis
essential oil against Streptococcus
mutans. Nutrients 2022, 14, 3290.
https://doi.org/10.3390/nu14163290.
3.
Takenaka, S.; Ohsumi, T. Noiri, Y.
Evidence-based strategy for dental biofilms: Current evidence of mouthwashes on
dental biofilm and gingivitis. Jpn.
Dent. Sci. Rev. 2019, 55, 33-40. https://doi.org/10.1016/j.jdsr.2018.07.001.
4.
Balagopal, S. Arjunkumar, R.
Chlorhexidine: the gold standard antiplaque agent. J. Pharm. Sci. Res. 2013,
5, 270-274.
5.
Brookes, Z.L.S.; Bescos, R.; Belfield,
L.A.; Ali, K. Roberts, A. Current uses of chlorhexidine for management of oral
disease: a narrative review. J. Dent.
2020, 103, 103497. https://doi.org/10.1016/j.jdent.2020.103497
6.
Nazzaro, F.; Fratianni, F.; De Martino,
L.; Coppola, R. De Feo, V. Effect of essential oils on pathogenic bacteria. Pharmaceuticals 2013, 6, 1451-1474. https://doi.org/10.3390/ph6121451.
7.
Dhifi, W.; Bellili, S.; Jazi, S.;
Bahloul, N. Mnif, W. Essential oils’ chemical characterization and
investigation of some biological activities: a critical review. Medicines. 2016, 3, 25. https://doi.org/10.3390/medicines3040025.
8.
Oliveira, T.A.S.; Santiago, M.B.;
Santos, V.H.P.; Silva, E.O.; Martins, C.H.G. Crotti, A.E.M. Antibacterial
activity of essential oils against oral pathogens. Chem. Biodiv. 2022, 19,
e202200097. https://doi.org/10.1002/cbdv.202200097.
9.
Freires, I.A.; Denny, C.; Benso, B.; De
Alencar, S.M. Rosalen, P.L. Antibacterial activity of essential oils and their
isolated constituents against cariogenic bacteria: a systematic review. Molecules. 2015, 20, 7329-7358. https://doi.org/10.3390/molecules20047329.
10.
Morais, S.M.; Cossolosso, D.S.; Silva,
A.A.S.; Moraes-Filho, M.O.; Teixeira, M.J.; Campelo, C.C.; Bonilla, O.H.;
Paula-Júnior, V.F. Vila-Nova, N.S. Composition, in vitro and in silico
antileishmanial evaluation, antioxidant and cytotoxicity activities. J. Braz. Chem. Soc. 2019, 30, 2404-2412. https://doi.org/10.21577/0103-5053.20190155.
11.
Arigó, A.; Rigano, F.; Micalizzi, G.;
Dugo, P. Mondello, L. Oxygen heterocyclic compound screening in Citrus
essential oils by linear retention index approach applied to liquid
chromatography coupled to photodiode array detector. Flavour Fragr. J. 2019,
34, 349-364. https://doi.org/10.1002/ffj.3515.
12.
Jalili, M. Sharifi, A. Chemical
composition and antibacterial activities of the essential oil and extract of Cirsium congestum. J. Food Biosci.
Technol. 2023, 13, 53-60.
13.
Mariyammal, V.; Sathiageetha, V.;
Amalraj, S.; Gurav, S.S.; Amiri-Ardekan, E.; Jeeva, S. Ayyanar, M. Chemical
profiling of Aristolochia tagala
Cham. leaf extracts by GC-MS analysis and evaluation of its antibacterial
activity. J. Indian Chem. Soc. 2023, 100, 100807. https://doi.org/10.1016/j.jics.2022.100807.
14.
Penido, C.; Costa, K.A.; Futuro, D.O.;
Paiva, S.R.; Kaplan, M.A.C.; Figueiredo, M.R. Henriques, M.G.M.O.
Anti-inflammatory and anti-ulcerogenic properties of Stachytarpheta cayennensis (LC Rich) Vahl. J. Ethnopharm. 2006,
104, 225-233. https://doi.org/10.1016/j.jep.2005.09.006.
15.
Pieroni, A.; Vanderbroek, I.;
Prakofjewa, J.; Bussmann, R.W.; Paniagua-Zambrana, N.Y.; Maroyi, A.; Torri, L.;
Zocchi, D.M.; Dam, A.T.K.; Khan, S.M.; Ahmad, H.; Yesil, Y.; Huish, R.; Pardo-de-Santayana,
M.; Mocan, A.; Hu, X.; Boscolo, O. Sõukand, R. Taming the pandemic? The
importance of homemade plant-based foods and beverages as community responses
to COVID-19. J. Ethnobiol. Ethnomed.
2020, 16, 1-9. https://doi.org/10.1186/s13002-020-00426-9.
16.
Okoye, T.C.; Akah, P.A.; Okoli, C.O.;
Ezike, A.C. Mbaoji, F.N. Antimicrobial and antispasmodic activity of leaf
extract and fractions of Stachytarpheta
cayennensis. Asian Pac. J. Trop. Med. 2010, 2010, 189-192. https://doi.org/10.1016/s1995-7645(10)60006-5.
17.
Schapoval, E.E.S.; de Vargas, M.R.W.;
Chaves, C.G.; Bridi, R.; Zuanazzi, J.A. Henriques, A.T. Antiinflammatory and
antinociceptive activities of extracts and isolated compounds from Stachytarpheta cayennensis. J.
Ethnopharm. 1998, 60, 53-59. https://doi.org/10.1016/s0378-8741(97)00136-0.
18.
Adebajo, A.C.; Olawode, E.O.; Omobuwajo,
O.R.; Adesanya, S.A.; Begrow, F.; Elkhawad, A.; Akanmu, M.A.; Edrada, R.;
Proksch, P.; Schmidt, T.J.; Klaes, M. Verspohl, E.J. Hypoglycaemic constituents
of Stachytarpheta cayennensis leaf. Planta Med. 2007, 73, 241-250. https://doi.org/10.1055/s-2007-967125.
19.
Okoye, T.C.; Akah, P.A.; Ezike, A.C.;
Uzor, P.F.; Odoh, U.E.; Igboeme, S.O.; Onwuka, U.B. Okafor, S.N.
Immunomodulatory effects of Stachytarpheta
cayennensis leaf extract and its synergistic effect with artesunate. BMC Complement. Altr. Med. 2014, 14, 376. https://doi.org/10.1186/1472-6882-14-376.
20.
Olayiwola, G.; Ukponmwan, O. Olawode, D.
Sedative and anxiolytic effects of the extracts of the leaves of Stachytarpheta cayennensis in mice. African J. Trad. Compl. Alt. Med. 2013, 10, 568-579.
21.
Seshadri, V.D.; Kandasamy, K.; Al
Osaimi, M.; Gabr, G.; Kamal, M.; Poyil, M.; Oyouni, A.A.A.; Altayar, M.A.;
Jalal, M.M.; Albloshi, A.M.K.; Bawazir, W.M. Mohideen, A.P. Stachytarpheta cayennensis-mediated
copper nanoparticles shows anticancer activity in both in vitro and in vivo
models. Appl. Nanosci. 2022, (early access). https://doi.org/10.1007/s13204-022-02546-y.
22.
Gkokon, J.E.; Ettebong, E. Antia, B.S. In vivo antimalarial activity of
ethanolic leaf extract of Stachytarpheta
cayennensis. Indian J. Pharmacol. 2008,
40, 111-113. https://doi.org/10.4103/0253-7613.42303.
23.
Maquiaveli, C.D.; Sa, A.M.O.E.; Vieira,
P.C. da Silva, E.R. Stachytarpheta
cayennensis extract inhibits promastigote and amastigote growth in Leishmania amazonensis via parasite
arginase inhibition. J. Ethnopharm.
2016, 192, 108-113. https://doi.org/10.1016/j.jep.2016.07.044.
24.
De Souza, P.A.; Silva, C.G.; Machado,
B.R.P.; de Lucas, N.C.; Leitão, G.G.; Eleutherio, E.C.A.; Ortiz, G.M.D.
Benchetrit, L.C. Evaluation of antimicrobial, antioxidant and phototoxic
activities of extracts and isolated compounds from Stachytarpheta cayennensis (Rich.) Vahl, Verbenaceae. Rev. Bras. Farmacogn. 2010, 20, 922-928. https://doi.org/10.1590/s0102-695x2010005000042.
25.
Onofre, S.B.; Kagimura, F.Y. Mattiello,
S.P. Antimicrobial activity of essential oils obtained from Stachytarpheta cayennensis, (Rich.)
Vahl. (Verbenaceae) collect ed in the South-west region of Paraná -Brazil. J. Med. Plants Res. 2014, 8, 1347-1353. https://doi.org/10.13140/RG.2.1.4332.7846.
26.
Duarte, M.C.T.; Leme, E.E.; Delarmelina,
C.; Soares, A.A.; Figueira, G.M. Sartoratto, A. Activity of essential oils from
Brazilian medicinal plants on Escherichia
coli. J. Ethnopharmacol. 2007,
111, 197-201. https://doi.org/10.1016/j.jep.2006.11.034.
27.
Melo, N.I.; Carvalho, C.E.; Fracarolli,
L.; Cunha, W.R.; Veneziani, R.C.S.; Martins, C.H.G. Crotti, A.E.M.
Antimicrobial activity of the essential oil of Tetradenia riparia (Hochst.) Codd. (Lamiaceae) against cariogenic
bacteria. Braz. J. Microbiol. 2015, 46, 519-525. https://doi.org/10.1590/s1517-838246246220140649.
28.
Aguiar, G.P.; Carvalho, C.E.; Dias,
H.J.; Reis, E.B.; Martins, M.H.G.; Wakabayashi, K.A.L.; Groppo, M.; Martins,
C.H.G.; Cunha, W.R. Crotti, A.E.M. Antimicrobial activity of selected essential
oils against cariogenic bacteria.
Nat. Prod. Res. 2013, 27, 1668-1672. https://doi.org/10.1080/14786419.2012.751595.
29.
Carvalho, E.S.; ayres, V.F.S.; Oliveira,
M.R.; Corrêa, G.M.; Takeara, R.; guimarães, A.C.; Santiago, M.B.; Oliveira,
T.A.S.; Martins, C.H.G.; Crotti, A.E.M. Silva, E.O. Anticariogenic activity of
three essential oils from Brazilian Piperaceae. Pharmaceuticals. 2022,
15, 972. https://doi.org/10.3390/ph15080972.
30.
Oliveira, T.A.S.; Vieira, T.M.;
Esperandim, V.R.; Martins, C.H.G.; Magalhães, L.G.; Miranda, M.L.D. Crotti,
A.E.M. Antibacterial, antiparasitic, and cytotoxic activities of chemical
characterized essential oil of Chrysopogon
zizanioides roots.
Pharmaceuticals 2022, 15, 967. https://doi.org/10.3390/ph15080967.
31.
Ferreira, M.A.; Carvalho, T.C.; Turatti,
I.C.C.; Furtado, N.A.J.C.; Martins, C.H.G.; Lopes, N.P.; Cunha, W.R. Crotti,
A.E.M. Antimicrobial activity of Aegiphilla
sellowiana against oral pathogens.
Rev. Bras. Farmacogn. 2010, 20,
246-249. https://doi.org/10.1590/s0102-695x2010000200018.
32.
Andrews, J.M. Determination of minimum
inhibitory concentrations. J.
Antimicrob. Chemother. 2001, 48
(supl. 1), 5-16. https://doi.org/10.1093/jac/dkf083.
33.
Winska, K.; Maczka, W.; Lyczko, J.;
Grabarczyk, M.; Czubaszek, A. Szummy, A. Essential oils as antimicrobial
agents—myth or real alternative? Molecules. 2019, 24, 2130. https://doi.org/10.3390/molecules24112130.
34.
Choi, O.; Cho, S.K.; Kim, J.; Park, C.G.
Kim, J. In vitro antibacterial
activity and major bioactive components of Cinnamomum
verum essential oils against cariogenic bacteria, Streptococcus mutans and Streptococcus
sobrinus. Asian Pac. J. Trop. Med. 2016,
6, 308-314. https://doi.org/10.1016/j.apjtb.2016.01.007.
35.
Crevelin, E.J.; Caixeta, S.C.; Dias,
H.J.; Groppo, M.; Cunha, W.R.; Martins, C.H.G. Crotti, A.E.M. Antimicrobial
activity of the essential oil of Plectranthus
neochilus against cariogenic bacteria.
Evid. Based. Compl. Alt. Med. 2015,
2015, 102317. https://doi.org/10.1155/2015/102317.
36.
Khan, M.; Alkhathlan, H.Z. Khan, S.T.
Antibiotic and antibiofilm activities of Salvadora
persica L. essential oils against Streptococcus
mutans: a detailed comparative study with chlorhexidine digluconate. Pathogens 2020, 9, 66. https://doi.org/10.3390/pathogens9010066.
37.
Karadaglioglu, O.I.; Ulusoy, N.; Baser,
K.H.C.; Hanoglu, A. Sik, H. Antibacterial activities of herbal toothpastes
combined with essential oils against Streptococcus
mutans. Pathogens 2019, 8, 20. https://doi.org/10.3390/pathogens8010020.
38.
Saleem, M.; Nazir, M.; Ali, M.S.;
Hussair, H.; Lee, Y.S.; Riaz, N. Jabbar, A. Antimicrobial natural products: an
update on future antibiotic drug candidates.
Nat. Prod. Rep. 2010, 27, 238-254. https://doi.org/10.1039/b916096e.
39.
Lima, P.C.T.; Niculau, E.S.; Silva, W.F.
Óleo essencial in natura da planta Stachytarpheta cayennensis (verbenaceae):
caracterização química por GC-MS, in Ciências agrárias: O avanço da ciência no
Brasil - volume 1, J.O.F. Melo, Editor. 2021, Editora Científica Digital:
Campinas, SP. p. 111-125. https://doi.org/10.37885/210604906.
40.
Silva, P.S.; Suzuki, E.Y.; Moreira,
A.P.; Barbosa-Raposo, N.R.; Almeida-Alves, T.M. Facio-Viccini, L. Stachytarpheta gesnerioides Cham.:
chemical composition of hexane fraction and essential oil, antioxidant and
antimicrobial activities. Bol.
Latinoam. Caribe Plantas Med. Arom. 2012,
11, 542-548.
41.
Moghaddam, M.; Mehdizadeh, L. Chemistry
of essential oils and factors influencing their constituents, in Handbook of
Food Bioengineering, Soft Chemistry and Food Fermentation, A.M. Grumezescu and
A.M. Holban, Editors. 2017, Academic Press. p. 379-419.
42.
Yoo, H.J. Jwa, S.K. Inhibitory effects
of beta-caryophyllene on Streptococcus
mutans biofilm. Arch. Oral Biol.
2018, 88, 42-46. https://doi.org/10.1016/j.archoralbio.2018.01.009.
43.
Xu, J.S.; Li, Y.; Cao, X. Cui, Y. The
effect of eugenol on the cariogenic properties of Streptococcus mutans and dental caries development in rats. Exp. Therap. Med. 2013, 5, 1667-1670. https://doi.org/10.3892/etm.2013.1066.
44.
Liu, Y.; Liu, P.W.; Wang, I.; Shi, Y.F.;
Chen, J.; Wang, H.J. Zhang, X.Y. Inhibitory effects of citrus lemon oil and
limonene on Streptococcus sobrinus -
Induced dental caries in rats. Arch.
Oral Biol. 2020, 118, 104851. https://doi.org/10.1016/j.archoralbio.2020.104851.
45. Dorman, H.J.D. Deans, S.G. Antimicrobial agents from plants: antibacterial activity of plant volatile oils. J. Appl. Microbiol. 2000, 88, 308-316. https://doi.org/10.1046/j.1365-2672.2000.00969.x.
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
Abstract
The
antibacterial activity of the essential oil from the leaves of Stachyterpheta
cayenensis (Verbenaceae) (SC-EO) grown in Brazil against a panel of cariogenic
bacteria was investigated using the broth microdilution method. SC-EO displayed
strong activity against Streptococcus mutans, S. mitis, S. salivarus, and S.
sanguinis, with minimal inhibitory concentration (MIC) values of 300, 300, 400,
and 400 µg/mL, respectively. Oct-1-en-3-ol (37.8%), 2,6-di-tert-butyl-4-methylphenol
(20.9 to 20.6%), (E)-phytol (8.6 to 8.3%), eugenol (5.1 to 4.8%), and borneol
(4.9 to 4.6 %) were identified as the main chemical compounds of SC-EO. To the
best of our knowledge, this is the first report on the antibacterial activity
of the essential oil of S. cayenensis.
Abstract Keywords
Coast redwood,
heartwood, sap-wood, monoterpene enantiomers, α-pinene, fatty alcohol, diterpenoid.
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
Editor-in-Chief
This work is licensed under the
Creative Commons Attribution 4.0
License.(CC BY-NC 4.0).


