Research Article
Opeyemi Nudewhenu Avoseh*
Opeyemi Nudewhenu Avoseh*
Corresponding author
Department of
Chemistry, Faculty of Science, Lagos State University, Ojo, Lagos, Nigeria.
E-mail:
opeyemi.avoseh@lasu.edu.ng, seavoseh@gmail.com; Tel.: +2349036037221.
Department of Chemistry, Faculty of Science, Lagos State University, Ojo, Lagos, Nigeria.
E-mail: opeyemi.avoseh@lasu.edu.ng, seavoseh@gmail.com; Tel.: +2349036037221.
Isiaka Ajani Ogunwande
Isiaka Ajani Ogunwande
Foresight
Institute of Research and Translation, Ibadan, Nigeria.
E-mail: isiaka.ogunwande@gmail.com
Amonah Temitope Arije
Amonah Temitope Arije
Department of
Chemistry, Faculty of Science, Lagos State University, Ojo, Lagos, Nigeria.
And
Department of
Chemistry, East Tennesse State University, Brown Hall Room 469, Johnson City,
TN 37614-1700, USA.
E-mail: amonaharije@gmail.com
Fanyana Moses Mtunzi
Fanyana Moses Mtunzi
Department of
Chemistry, Faculty of Applied and Computer Sciences, Vaal University of
Technology, Vanderbijl Park, South Africa.
E-mail: Fanyana@vut.ac.za
Roberta Ascrizzi
Roberta Ascrizzi
Dipartimento di
Farmacia, Università di Pisa, Italy.
E-mail: roberta.ascrizzi@gmail.com
Guido Flamini
Guido Flamini
Dipartimento di
Farmacia, Università di Pisa, Italy.
E-mail: guido.flamini@farm.unipi.it
Received: 2022-12-21 | Revised:2023-01-18 | Accepted: 2023-01-19 | Published: 2023-01-26
Pages: 26-31
DOI: https://doi.org/10.58985/jeopc.2023.v01i01.05
Abstract
This study was designed to determine the chemical constituents of
essential oil from the leaves of Alternanthera
sessilis (L.)
R. Br. Ex DC. (Amaranthaceae) grown
in Nigeria. The essential oil was isolated using
hydrodistillation method. The constituents of A. sessilis oil were
characterized using gas chromatography-flame ionization detector (GC-FID) and
gas chromatography-mass spectrometry (GC-MS). The yield of the essential oil
was 0.21% (v/w), calculated on a dry weight basis. A total of seventeen compounds
accounting for 95.6% of the oil content were identified. The oil was devoid of any of the monoterpene compounds. The main constituents of
the oil were hexahydrofarnesyl
acetone (35.6%), β-caryophyllene (16.1%) and n-heptadecane (10.7%). Terpenoid compounds were being reported for the
first time in A. sessilis.
Keywords
Alternanthera sessilis, essential oil, hexahydrofarnesyl acetone, β-caryophyllene,
n-heptadecane.
1. Introduction
Alternanthera is a diverse genus (of about 80 to 200 species, and the second largest in subfamily Gomphrenoideae of the Amaranthaceae. The highest diversity of this genus occurs in South America, but many species also occur in the Caribbean, Central America and Mexico. Alternanthera sessilis (L.) R. Br. Ex DC. A plant of the family Amaranthaceae occurs throughout the tropical and subtropical regions of the World [1]. It has been introduced to other parts of the world including Nigeria. The synonyms of A. sessilis include Alternanthera triandra Lam., Alternanthera prostrata Don. And Achyranthus triandra Roxb [2-3]. A. sessilis is a perennial herb with prostrate stems, rarely ascending, often rooting at the nodes. The leaves are obovate to broadly elliptic, occasionally linear-lanceolate, 1-15 cm long and 0.3-3 cm wide. The petioles are glabrous to sparsely villous of about 1-5 mm long. The shiny white and glabrous flowers are spikes, bract and bracteoles 0.7-1.5 mm long while the sepals are 2.5-3 mm long. The plant flowers from December until March. The plant grows wild, but is also cultivated for food, herbal medicines, as an ornamental plant. The leaves and young shoots are consumed as vegetables [4].
There are several reports describing the in-vitro and in-vivo pharmacological studies on A. sessilis. A study reported that both aqueous and ethanolic extracts of aerial parts of A. sessilis possess significant nootropic potential [3,5]. A. sessilis will inhibit the cytotoxic nature of the pathogen causing ocular diseases. Various extracts of A. sessilis were proved to exhibited antimicrobial [6-7], anti-cataract [4,6], wound healing [6, 8], antioxidant [6,9], analgesic [10-11], hepatopro-tective [12-13], anti-cancer [12,14], hematinic [13,15], anti-hyperglycemic [14,16], analgesics [14, 16], anti-diarrhoeal [15,17], anti-inflammatory [18] and anti-diabetic [19]. A. sessilis is a promising tool for enhancing production of potent α-glucosidase inhibitors [20].
The young shoots of A. sessilis contain 3β-Oβ-D-glucopyranosyluronic acid, 2β-Oβ-D-glucopyrano-syloleanolic acid, stigmasterol and β-sitosterol [21-22]. Phytochemical studies yielded β-carotene, ricinoleic acid, myristic, palmitic, stearic, oleic and linoleic acids, α-spiraterol and uronic acid, 2, 4-methylene cycloartanol, cycloeucalenol, choline, oleanolic acid, lupeol, campesterol and 5-α-stigmasta-7-enol [23-24]. The phenolic compounds present in A. sessilis were ferulic acid, catechin, vanillic acid, and epigallo-catechin, gallic acid and chlorogenic acid [9, 25-26]. The polyphenols isolated from A. Sessilis were (+)-catechin, rutin, ellagic acid, and quercetin [10,27]. Moreover, propane-diyl-bis-hexahydro-isochromene, an antifungal compound was also isolated from A. sessilis leaves [28]. The allelopathic potential of A. sessilis was attributed to the content of vanillic acid, gallic acid and chlorogenic acid [25]. Crude extract of A. sessilis analyzed by GC/MS afforded (Z,Z)-9,12,-octadecadienoic acid (25.33%), vitamin A aldehyde (11.94%) and 12-bromododecanoic acid (11.37%) as major compounds [29]. Analysis of hydro alcoholic and acetone extracts of stems by GC-MS gave the major phytoconstituents as methoxy-bis (cyclopenta-diene), 5,10-dihexyl-5,10-dihydroindolo [3,2-b]indole-2,7-dicarbaldehyde and 1,2-bis[3,4-dimethoxy ben-zyl]-1,2-bis (methoxymethyl) ethane [30]. The fatty acids content of A. sessilis includes ascorbic acid, stearic acid, lignoceric acid, behenic acid, palmitic acid and oleic acid [31]. A mixture of diasteriomers of ionone which showed low antimicrobial activity against Pseudomonas aeruginosa and Trichophyton mentagrophytes were also isolated from the plant [32]. Other compounds such as 11-eicosenoic acid, methyl ester, (E)-9-octadecenoic acid ethyl ester and 20-oxo-heneicosanoic acid methyl ester were also detected from A. sessilis.
The major components of essential oil of leaves of A. sessilis analyzed by the GC-MS were found to be 1,1,1,5,5,5-hexamethyl-3,3-bis[trimethylsilyl)oxy]tris-iloxane (15.43%), S,S-dioxide trans-2-methyl-4-N-pen-tylthiane (11.27%), didodecylphthalate (10.62%) and tetrahydro-2,5-dimethoxy furan (10.01%) [33]. However, 1,1,1,5,5,5-hexamethyl-3,3-bis [trimethyl-silyl) oxy]trisiloxane (17.76%), trans-4-ethyl-5-octyl-2, 2-bis(trifluromethyl)-1,3-dioxolane (11.12%) and tet-rahydro-2,5-dimethoxy furan (9.10%) were the major components of the flower oil [33]. The leaf essential oil of A. sessilis was reported to possess radical scavenging activity [33].
In continuation of our on-going study aimed at the characterization of the chemical constituents and biological activities of essential oils from Nigerian plants [34] we report herein the volatile compounds identified in the leaves of A. sessilis.
2. Materials and methods
2.1 Collection of A. sessilis leaves
The leaves of A. sessilis (220 g) were collected from Lagos State University, Nigeria. The collection of the plant samples was done during the month of April 2018. The plant was identified by Mr Adeniji, K.A. of the Forestry Research Institute, Ibadan with voucher number FHI 112139; and a voucher specimen was deposited at the herbarium. Prior to hydrodistillation process the samples were air-dried under laboratory shade for two weeks (27ºC) to reduce the moisture contents. In addition, sediments and other unwanted materials were separated from the samples.
2.2 Hydrodistillation of the essential oil fromA. sessilis
Dried samples were pulverized into coarse powder in locally made grinder prior to hydrodistillation. In this experiment, 220 g of pulverized samples were used. The pulverized leaves of A.sessilis were loaded into a 5 L flask. Distilled water was added until the sample was covered completely. The sample was then subjected to hydrodistillation for 3 h in Clevenger-type apparatus according to an established protocol [35] to obtained essential oil which was stored under refrigeration (4oC) in weighed sample bottle as described in previous studies [34-35].
2.3 Chemical analysis of the oil
In the analysis of the chemical constituents of the essential oil, gas chromatography-flame ionization detector (GC-FID) and gas chromatography coupled with mass spectrometry (GC/MS) were used. For the GC-FID, an HP-5890 Series II gas chromatograph that has two capillary columns (HP-Wax and HP-5), both of dimension 30 m x 0.25 mm and film thickness of 0.25 μm was used for the analysis. The GC was temperature program at 60ºC and isothermally held for 10 min, rising at 5ºC/min to 220ºC. The injector and detector temperatures were both maintained at a temperature of 250ºC. Nitrogen was used as a carrier gas at a flow rate of 2 mL/min. The GC was equipped with a dual detector (FID). The method of splitting at ratio of 1:30 was employed to inject the essential oil samples (0.5 mL) into GC. Quantification was done by external standard method using calibration curves generated by running GC analysis of representative compounds.
During the analysis of the essential oil by gas chromatography-mass spectrometry (GC/EIMS), a Varian CP-3800 gas-chromatograph equipped with a HP-5 capillary column of dimension 30 m x 0.25 mm and a film thickness 0.25 μm was used. The gas chromatograph was coupled to a Varian Saturn 2000 ion trap mass detector. The analytical conditions employed include injector and transfer line temperatures of 220ºC and 240ºC, respectively, while the oven temperature was programmed from 60ºC to 240ºC at 3ºC/min. In this analysis, the carrier gas was helium at a flow rate of 1mL/min. The volume of essential oil injected into the GC was 0.2 μL (10% n-hexane solution) at a split ratio of 1:30. In addition, the mass spectra were recorded at 70 eV while the acquisition mass range was recorded at m/z 30-300 with a scan rate of 1 scan/sec.
The identification of the constituents was based on comparison of their retention times with those of authentic samples, comparing their linear indices relative to a series of n-alkanes (C6-C36). Further identifications were also made possible by the use of a homemade library of mass spectra built up from pure substances and components of known oils [36].
and MS literature data as described in our previous report [34]. Moreover, the molecular weights of all the identified substances were confirmed by GC-CIMS, using MeOH as CI ionizing gas.
3. Results and discussion
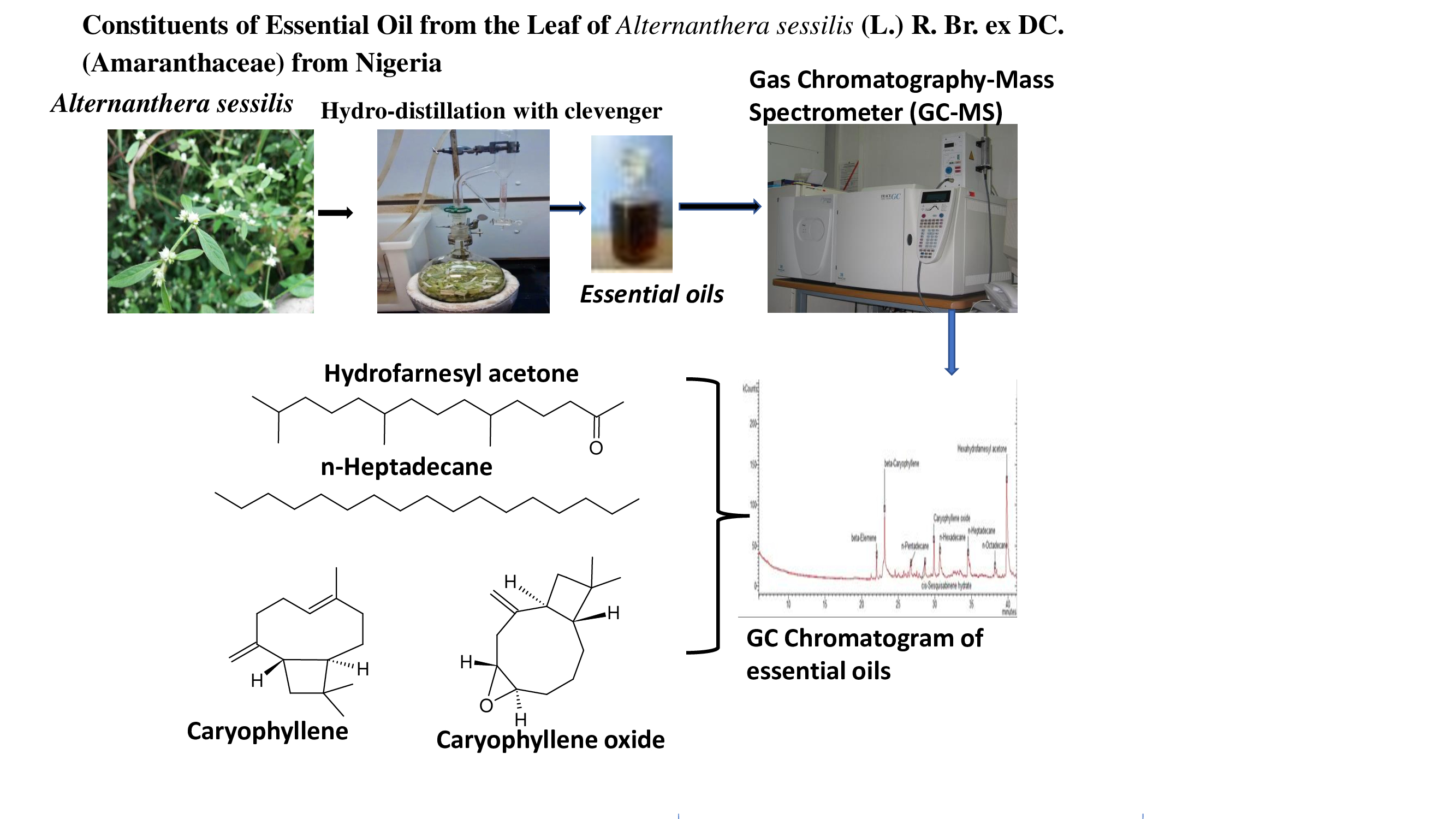
The average yield of essential oils was 0.21% ± 0.01 (v/w), calculated on a dry weight basis. The oil sample was colourless. The identities, retention indices and percent compositions of the oils are shown in Table 1 and Fig. 1.
Table 1. Constituent of essential oil of Alternanthera sessilis
Sl. No | Compoundsa | LRIb | LRIc | Relative abundanced | ||
1 | β-Elemene | 1392 | 1390 | 3.6 | ||
2 | n-Tetradecane | 1400 | 1400 | 0.3 | ||
3 | β-Caryophyllene | 1420 | 1419 | 16.1 | ||
4 | α-Humulene | 1456 | 1454 | 0.9 | ||
5 | γ-Himachalene | 1475 | 1474 | 0.3 | ||
6 | Germacrene D | 1478 | 1477 | 1.0 | ||
7 | (E, Z)-α-Farnesene | 1490 | 1489 | 0.4 | ||
8 | n-Pentadecane | 1500 | 1500 | 3.9 | ||
9 | (E)-γ-Bisabolene | 1537 | 1537 | 0.7 | ||
10 | cis-Sesquisabinene hydrate | 1545 | 1550 | 3.0 | ||
11 | Caryophyllene oxide | 1581 | 1583 | 6.4 | ||
12 | n-Hexadecane | 1600 | 1600 | 6.4 | ||
13 | Humulene epoxide II | 1607 | 1610 | 1.7 | ||
14 | (E)-Sesquilavandulol | 1625 | 1624 | 0.1 | ||
15 | n-Heptadecane | 1700 | 1700 | 10.7 | ||
16 | n-Octadecane | 1800 | 1800 | 3.6 | ||
17 | Hexahydrofarnesyl acetone | 1845 | 1842 | 35.6 | ||
Total | 95.6 |
| ||||
Sesquiterpene hydrocarbons (Sr. No. 1, 3-7, 9) | 23.9 |
| ||||
Oxygenated sesquiterpenes (Sr. No. 10, 11, 13, 14) | 11.2 |
| ||||
Apocarotenoids (Sr. No. 17) | 35.6 |
| ||||
Non-terpene derivatives (Sr. No. 2, 8, 12, 15, 16) | 24.9 |
| ||||
aElution order on HP-5; b LRI, Linear retention indices on HP-5column; cLiterature retention indices; dStandard deviation are insignificant and excluded from the Table to avoid congestion; Sr. No serial number.
Seventeen compounds representing 95.6% of the total oil contents were identified by GC/MS. Sesquiterpene hydrocarbons (23.9%), oxygenated sesquiterpenes (11.2%), apocarotenoid (35.6%) and aliphatic hydro-carbons (24.9%) were the representative class of compounds identified in the oil. Monoterpene compounds were not identified from the oil. The main constituents of the oil were hexahydrofarnesyl acetone (35.6%), β-caryophyllene (16.1%), n-heptadecane (10.7%), caryophyllene oxide (6.4%) and n-hexa-decane (6.4%). The authors are aware of only a report on the analysis of A. sessilis essential oil. It can be deduced that the said analysis was not performed with conventional instrument while the identification of the compounds seemed faulty. This was due to the fact that non-terpenoid rather than terpenes were detected in the oil samples.
The major components of essential oil of leaves in previously report [33] such as 1,1,1,5,5,5-hexamethyl-3,3-bis[trimethylsilyl)oxy]trisiloxane, S,S-dioxide-trans-2-methyl-4-N-pentylthiane didodecylphthalate, and tetrahydro-2,5-dimethoxy furan, as well as trans-4-ethyl-5-octyl-2,2-bis(trifluromethyl)-1,3-dioxolane and tetrahydro-2,5-dimethoxy furan that were present in the flower oil, were not identified in the present oil under investigation. In addition, all the major and minor compounds of the present study were not previously as part of the constituents of A. sessilis. Moreover, scanty information exists on the chemical constituents and biological activities of essential oils from some other Alternanthera species analyzed from other parts of the world. 2,6-Di-t-butyl-4-methylphenol (42.16%), as well as geraniol (9.15%), aristolene (6.15%), γ-eudesmol (10.58%), and geranyl tiglate (8.21%) were the major constituents of the leaves of Alternanthera brasiliana (L.) Kuntze [37]. It can be postulated that the present data represents the first comprehensive attempt at the characterization of the volatile constituents of A. sessilis. Nevertheless, the amount and the composition of the bioactive substances may vary among different Alternanthera species, and according to different factors such as the extraction methods, the geographic and the growing conditions, the harvest time etc. [38].
Figure. 1. GC chromatogram and representative compounds of Alternanthera sessilison HP-5 column
4. Conclusions
The chemical constituents of essential oils from the leaf of A. sessilis from Nigeria were being reported for the first time. The major constituents of the essential oil were identified as hexahydrofarnesyl acetone, β-caryophyllene and n-heptadecane. None of the monoterpene compounds was present in the oil sample. The data presented herein were found to differ completely from a previous study on the oil sample.
Authors’ contributions
Conceptualization, O.N.A.; Methodology, I.A.O., O.N.A; Software, R.A; Validation, I.A.O; Formal analysis, F.M.M., R.A.; Investigation, A.T.A., O.N.A.; Resources, P.S.; Data curation, I.A.O; Writing–original draft preparation, A.T.A.; Writing – review & editing, R.A., O.N.A.; Project administration, O.N.A.
Acknowledgements
The authors would like to express their appreciation to the Department of Biochemistry, Lagos State University, for their technical assistance during the animal study.
Funding
No funding was received for this research.
Conflicts of interest
The authors declare that they have no conflict of interests.
References
1.
Tanaka, Y.;
Van Ke, N. Edible Wild Plants of Vietnam: The Bountiful Garden. Thailand:
Orchid Press. p. 21, 2007.
2.
Jayawardana, Y. D.; .
Sarananda, K. H.; Senarathne, S. M.; Ratnayake, R.H. The effect of variety and
fertilizer application on functional properties of ‘Mukunuwenna’ ‘’Alternanthera
sessilis,” J. Food Agric. 2018, 11(2), 9-18. https://doi.org/10.4038/jfa.v11i2.5213.
3.
Iamonico, D.;
Sánchez-Del Pino, I. Taxonomic revision of the genus Alternanthera
(Amaranthaceae) in Italy. Plant
Biosyst., 2016: 150,
2, 333-342. https://doi.org/10.11646/phytotaxa.199.1.1.
4.
Güzel, Y. A new
invasive weed record for Turkey: Alternanthera sessilis (Amaranthaceae)
. Plant Prot. Bull., 2017, 57, 1, 65–72. https://doi.org/10.16955/bitkorb.299057.
5.
Kumar, M.S.; Rani, S.G.;
Kumar, S.K.; Astalakshmi, N. Screening of aqueous ethanolic extracts of aerial
parts of Alternanthera sessilis Linn R Br Ex Dc for nootropic activity. J. Pharm. Sci. Res. 2011, 3, 1294-1297.
6.
Sobha, K.;
Vayunandana, R.G.; Ratna, K.A.; Verma, M.K. An investigation into phytochemical
constituents, antioxidant, antibacterial and anti-cataract activity of Alternanthera
sessilis, a predominant wild leafy vegetable of South India. Biocatal. Agric. Biotechnol. 2017, 10,
197-203. https://doi.org/10.1016/j.bcab.2017.03.008.
7.
Suganya, D.;
Banupriya, R.; Uma Maheswari, A.; Elumalai, S. Studies on biological activity
of aqueous extract of Alternanthera sessilis (Linn) for developing
potential herbal drug formulation of ocular diseases. Med. Aromat. Plants. 2019, 8, 327. https://doi.org/10.35248/2167-0412.19.8.327.
8.
Muniandy, K.;
Sivapragasam, G.; Woan, S.T.; Suresh, S.K.; Norhaizan, M.E.; Govindasamy, C.;
Al-Numair, S.K.; Palanisamy, A. In vitro wound healing potential of stem
extracts of Alternanthera sessilis. Evidence-Based Complement. Alter. Med.
2018: Article
ID 3142073, 13. https://doi.org/10.1155/2018/3142073.
9.
Azizah, O.;
Amin, I.; Fouad, A.R. Antioxidant properties of Alternanthera sessilis red and green. Acta Hortic. 2015, 1106, 131-136. https://doi.org/10.17660/actahortic.2015.1106.20.
10. Mondal, H.; Sanjib, S.; Khalijah, A.; Hemayet, H.;
Abdulwali, A.; Md Khirul, I.; Ismet, A.J.; Samir, K.S.; Md Golam, H.; Jamil,
A.S.; Shaikh, J.U. Central-stimulating and analgesic activity of the ethanolic
extract of Alternanthera sessilis in
mice. BMC Complement. Altern. Med. 2014, 398.
https://doi.org/10.1186/1472-6882-14-398.
11. Sushree, S.M.; Arjun, K.; Jayant, C.; Pritam, M.;
Rohini, K.R.; Indrapal, R. Analgesic
activity of hydroethanolic extract of Alternanthera sessilis in
mice. J. Pharmacog. Phytochem. 2018, 7, 1836-1839.
12. Bhuyan, B.; Baishya, K.; Rajak, P. Effects of Alternanthera sessilis on liver function
in carbon tetrachloride induced hepatotoxicity in Wister rat model. Ind. J. Clin. Biochem. 2018, 33, 190-195.
https://doi.org/10.1007/s12291-017-0666-1.
13. Borthakar, A.; Mohan, P.; Lahon, L.C.; Kalita, D.J.;
Rahman, T.; Sarmah, A. Evaluation of hepatoprotective property of Alternanthera
sessilis in CCl4 induced hepatotoxicity in rats. Int. J. Curr. Microbiol. Appl. Sci. 2017, 6,
3271-3279. https://doi.org/10.20546/ijcmas.2017.608.390.
14. Gothai, S.; Muniandy, K.; Mohd, E.N.; Subbiah, S.K.;
Arulselvan, P. Anticancer potential of Alternanthera sessilisextract on HT-29 human colon cancer cells. Asian Pac. J. Trop. Biomed. 2018, 8,
394-402. https://doi.org/10.4103/2221-1691.239427.
15. Arollado, E.C.; Marina, O.O. Hematinic activity of Alternanthera sessilis (L.) R. Br.
(Amaranthaceae) in mice and rats. E-Int.
Scient. Res. J. 2010, 2, 1-7.
16. Ahamed, I.H.; Mohammad, F.; Shahnaz, R.; Rownak, J.;
Mohammed, R. A preliminary evaluation of antihyperglycemic and analgesic
activity of Alternanthera sessilis
aerial parts. BMC Complement. Altern.
Med. 2014, 14,
169. https://doi.org/10.1186/1472-6882-14-169.
17.
Kumar, Y.S.;
Das S. Evaluation of anti-diarrhoeal property of crude aqueous extract of Alternanthera sessilis Linn. J. Pharm. Innov. 2013, 3, 110-115. https://doi.org/10.4314/biokem.v16i2.32580.
18.
Subhashini,
T., Krishnaveni, B.; Srinivas, R.C. Anti-inflammatory activity of the leaf
extract of Alternanthera sessilis. HYGEIA J. Drugs Med. 2010, 2, 54-57.
19. Das, M.; Kumar, A.D.; Mastanaiah, K.; Das, A.
Evaluation of anti-diabetic activity of ethanolic extract of Alternanthera
sessilis Linn. in streptozotocin-induced diabetic rats. Int. J. Pharm. Sci. Res. 2015, 7,
1027-1032. https://doi.org/10.48047/ecb/2023.12.si5.094.
20.
Chai, T.T.;
Khoo, C.S.; Tee, C.S.; Wong, F.C. Alpha-glucosidase inhibitory and antioxidant
potential of antidiabetic herb Alternanthera sessilis: comparative
analyses of leaf and callus solvent fractions. Pharmacog. Mag. 2016, 12, 253-258.
21. Kapundu, M.; Lami, N.; Delaude, C. Analysis of
saponin from Alternanthera sessilis. Bull. Soc. Roy. Sci. Liege. 1986, 55, 605-606.
22.
Sinha, P.;
Arora, V.K.; Wahi, S.P. Chemical investigation on Alternanthera sessilis.
Ind. Drug. 1984, 1,
139-140.
23.
Walter, T.M.;
Merish, S.; Tamizhamuthu, M. Review of Alternanthera
sessilis with reference to traditional Siddha medicine. Int. J. Pharmacog. Phytochem. Res. 2014, 6, 249-254.
24. Aamir, S.; Abdul Q.; Rafia R.;
Farwa N.; Muhammad R.S. A review of bioactivity guided medicinal uses and
therapeutic potentials of noxious weed (Alternanthera sessilis). Int. J.
Chem. Biol. Sci. 2018,14, 95-103.
25. Mehmood, A.; Tanveer, A.; Nadeem, M.A.; Zahir, Z.A.
Comparative allelopathic potential of metabolites of two Alternanthera species against germination and seedling growth of
rice. Planta Daninha Vicosa-MG.
2014, 32, 1-10. https://doi.org/10.1590/s0100-83582014000100001.
26. Gupta, R.; Singh, H.K. Detection and quantification
of gallic acid in Alternanthera sessilis
and Clerodendrum infortunatum by
HPTLC. Int. J. Pharm. Pharmacol. 2016, 4,
467-471. https://doi.org/10.15406/ppij.2016.04.00097.
27. Vani, M.; Rahaman, S.K.A.; Rani, A.P. Detection and
quantification of major phytochemical markers for standardization of
Talinum portulacifolium, Gomphrena serrata, Alternanthera sessilis and
Euphorbia heterophylla by HPLC. Pharmacog.
J. 2018, 10, 439-446. https://doi.org/10.5530/pj.2018.3.72.
28. Sundar, R.D.V.; Ravi, L.; Mythili, S. Discovery of
new anti-fungal phytochemical PDHC (propane-diyl-bis-hexahydro-isochromene)
isolated from Alternanthera sessilis
leaves. Int. J. Pharm. Sci. Res.
2019, 10, 1136-1147.
29. Khan, M.S.; Yusufzai, S.K.; Ying, L.Y.; Zulnashriq,
W. GC-MS based chemical profiling and evaluation of antioxidant potential of
leaves and stems of Alternanthera
sessilis red from Sabah, Malaysia. Int.
J. Pharm. Pharm. Sci. 2018, 10, 4-9. https://doi.org/10.22159/ijpps.2018v10i7.25204.
30. Mamillapalli, V.; Abdul Rahaman, S.K.; Avula, P.R. In vivo antiasthmatic
studies and phytochemical characterization on the stem extracts of Alternanthera
sessilis L. using guinea pigs model. J.
Entomol. Zool. Stud. 2017, 5, 1160-1171.
31. Arif, M.; Nusrat, J.; Tanvir, M.M.; Azizur, R.
Investigation of some constituents of two plants (Alternanthera
philoxeroides and Alternanthera sessilis) of Amaranthaceae family. Dhaka Univ. J. Sci. 2010, 58, 327-328.
32. Ragasa, C.Y.; Tremor, N.;
Redeont, J.A. Ionone derivatives from Alternanthera sessilis. J. Asian Nat. Prod. Res. 2002, 4,
109-115. https://doi.org/10.1080/10286020290027380.
33. Khan, M.S.; Yusufzai, S.K.;
Kaun, L.P.; Shah, M.D.; Idris, R. Chemical composition and antioxidant activity
of essential oil of leaves and flowers of Alternanthera sessilis red
from Sabah. J. App. Pharm. Sci.
2016, 6, 157-161. https://doi.org/10.7324/japs.2016.601222.
34. Eresanya, O.I.; Avoseh, N.O.;
Ogunwande, I.A.; Lawal, O.A.; Giwa-Ajeniya, A.O. Chemical constituents of
essential oil from the leaves of Phyllanthus muellerianus (O. Kuntze)
Exell. J. Essent. Oil Bearing Plants.
2019, 22, 3, 865-870. https://doi.org/10.1080/0972060x.2019.1623721.
35. British
Pharmacopoeia. HM Stationary Office, London, Vol. II, 1990.
36. National
Institute of Science and Technology. Chemistry Web Book Data. Data from NIST
Standard Reference Database 2018; pp. 69.
37. Wakabayashi, K.A.L.; de Melo, N.I.; de Paula Aguiar, G.; de
Carvalho, C.E.; Rafael, C.R.; André L.L.M; Groppo Jr., M.; Rodrigo, C.S.V.;
Antônio, E.M.C. Chemical composition of the essential oil from the leaves of Alternanthera
brasiliana (L.) Kuntze (Amaranthaceae). Investigação. 2010, 10, 82-85.
38. Sharifi-Rad,
M.; Elena, M.V.; Bahare, S.; Sharifi-Rad, J.; Karl, R.M.; Seyed, A.A.; Farzad,
K.; Salam, A.I.; Dima, M.; Zainul, A.Z.; Sharifi-Rad, M.; Zubaida, Y.;
Marcello, I.; Adriana, B.; Daniela, R.
Plants of the genus Zingiber as source of antimicrobial agents:
from tradition to pharmacy. Molecules.
2017, 22,
2145-2162. https://doi.org/10.20944/preprints201711.0102.v1.
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
Abstract
This study was designed to determine the chemical constituents of
essential oil from the leaves of Alternanthera
sessilis (L.)
R. Br. Ex DC. (Amaranthaceae) grown
in Nigeria. The essential oil was isolated using
hydrodistillation method. The constituents of A. sessilis oil were
characterized using gas chromatography-flame ionization detector (GC-FID) and
gas chromatography-mass spectrometry (GC-MS). The yield of the essential oil
was 0.21% (v/w), calculated on a dry weight basis. A total of seventeen compounds
accounting for 95.6% of the oil content were identified. The oil was devoid of any of the monoterpene compounds. The main constituents of
the oil were hexahydrofarnesyl
acetone (35.6%), β-caryophyllene (16.1%) and n-heptadecane (10.7%). Terpenoid compounds were being reported for the
first time in A. sessilis.
Abstract Keywords
Alternanthera sessilis, essential oil, hexahydrofarnesyl acetone, β-caryophyllene,
n-heptadecane.
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
Editor-in-Chief
This work is licensed under the
Creative Commons Attribution 4.0
License.(CC BY-NC 4.0).


