Review Article
Jazeela Abdulkhader
Jazeela Abdulkhader
Department of Pharmacology, Government Medical
College, Kannur-670503, Kerala, India.
Ayisha Ummer
Ayisha Ummer
Department of Pharmacology, Government Medical
College, Kannur-670503, Kerala, India.
Shifla Kareem
Shifla Kareem
Department of Pharmacology, Government Medical
College, Kannur-670503, Kerala, India.
Greeshma Sebastian
Greeshma Sebastian
Department of Pharmacology, Government Medical
College, Kannur-670503, Kerala, India.
Jaishna Jayachandran Nambiar
Jaishna Jayachandran Nambiar
Department of Pharmacology, Government Medical
College, Kannur-670503, Kerala, India.
Minil Mukundan
Minil Mukundan
Department of Pharmacology, Government Medical
College, Kannur-670503, Kerala, India.
Aiswarya Lakshmi
Aiswarya Lakshmi
Department of Pharmacology, Government Medical
College, Kannur-670503, Kerala, India.
Appil Gopalakrishnan
Appil Gopalakrishnan
Department of Pharmacology, Government Medical
College, Kannur-670503, Kerala, India.
Siju Ellickal Narayanan
Siju Ellickal Narayanan
Department of Pharmacology, Government Medical College, Kannur-670503, Kerala, India.
E-mail: sijuellickal@rediffmail.com, Tel: +90-3323238220, Fax: +90-3323238225
Received: 2023-06-07 | Revised:2023-07-15 | Accepted: 2023-07-18 | Published: 2023-08-10
Pages: 42-56
DOI: https://doi.org/10.58985/jpam.2023.v01i02.06
Abstract
Alzheimer's disease (AD) is the
dominant source of dementia worldwide among numerous neurodegenerative
disorders, with no definitive and enduring cure to date despite prolonged
research efforts. AD is believed to be caused due to the accumulation of
extracellular Aβ plaques and intracellular neurofibrillary tangles consisting
of hyper phosphorylated τ-protein in various parts of the human brain, such as
the cortex and limbic system. The development of drugs has been
significantly benefited by Ayurveda medicinal plants. Presently, there are more
than a hundred new medicines already in clinical development. The primary
emphasis of the review is on exploring multiple medicinal plants as a means of
preventing symptoms associated with Alzheimer’s disease. A study of the
phytochemical properties in various parts of plants has revealed several
significant chemical compounds, including lignans, flavonoids, tannins,
polyphenols, triterpenes, sterols, and alkaloids. These agents exhibit a
diverse range of pharmacological functions that can aid in the prevention and
treatment of various conditions, such as inflammation, amyloid deposits,
cholinesterase inhibition, hyperlipidemia, and oxidative stress.
Keywords
Alzheimer’s disease, medicinal
plants, phytochemical constituents, neuroprotective
herbs, therapeutic effects, flavonoids.
1. Introduction
Alzheimer
disease (AD) is a neurodegenerative disorder of the brain which is mostly
affecting old age people. The characteristics of the disorder are memory loss,
behavior deterioration, thought slowness and performance impairment [1]. The term Alzheimer Disease was
discovered by a German neuropathologist and psychiatrist named Alois Alzheimer
in 1906 [2]. Approximately five million people with age 65 years or older
and 200000 people younger than 65 years are affected by AD [3]. Alzheimer’s disease cannot be cured completely, but
symptomatic treatment may improve memory and other problems related with
dementia. Traditional medicine is practiced worldwide as memory enhancer since
ancient times. Natural therapy including herbs and medicinal plants has been
used in the treatment of memory deficits such as dementia, amnesia, as well as
Alzheimer’s disease for a long time [4]. Inflammation of brain tissues, increase
in the level of free radicals and deficiency of acetylcholine are the major
factors responsible for the generation of AD.
1.1 Pathophysiology
The
pathology in AD is neuronal degeneration and loss of synapses in the
hippocampus, cortex, and subcortical structures [5]. This loss results in gross atrophy of the affected regions,
resulting in loss of memory, inability to learn new information, mood swings,
executive dysfunction, etc [6].
The
histopathological characteristic features seen in the brain of patients with AD
are
1.
Senile
plaques contain extracellular aggregates of amyloid-beta (Aβ), a peptide
synthesized by breakage of Aβ precursors (genetic locus 21q21–22). Abnormal
deposits of Aβ are also seen in blood vessels.
2.
Intracellular
aggregations of neurofibrillary tangles (NFTs), dense bundles of abnormal fibres
in the cytoplasm of neurons which consist of an altered form of the hyper
phosphorylated microtubules associated protein (τ) [7].
Aβ plaques develop initially in basal,
temporal and orbitofrontal, neocortexregions of the brain and in later stages
progress throughout the neocortex, hippocampus, amygdala, diencephalon, and
basal ganglia. Aβ is found throughout
the mesencephalon, lower brain stem, and cerebellar cortex in critical cases [8].
This
concentration of Aβ triggers τ-tangle formation, which is found in the locus
coeruleus and transentorhinal and entorhinal areas of the brain. In the
critical stage, it spreads to the hippocampus and neocortex. For establishing the cause of AD, number of hypotheses is proposed.
The cholinergic hypothesis, which is the oldest theory, describes acetylcholine
(ACh) deficiency as one of the causative
factor for AD. Currently available therapies for AD management are based on
this hypothesis. The β-amyloid hypothesis, the most cogent hypothesis provides
the basis for the development of new therapeutic strategies for AD treatment [9].
According to
(based on) cholinergic hypothesis, acetylcholine deficiency is one of the
causative factors for AD [10]. So treatment for AD is done based
on this hypothesis.
Treatment
option for AD is mainly targeting the following, cholinergic dysfunction,
amyloid β neurotoxicity, oxidative damage and inflammation [11].Various studies showed that
non-steroidal anti-inflammatory drugs and antioxidants diminish the development
of AD [12]. FDA approved drugs for the
treatment and to alleviate the symptoms of AD are donepezil, rivastigmine,
galantamine (acetylcholinesterase inhibitors) and memantine (NMDA receptor antagonist).Cholinesterase inhibitors acts by
improving cognition in patients with mild to moderate AD while for severe AD
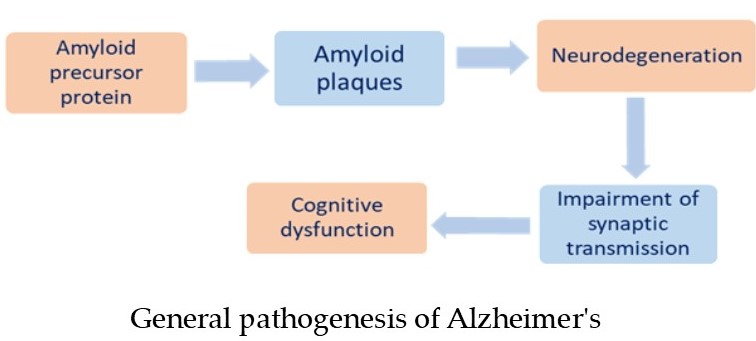
cases Memantine is more appropriate [13]. General pathogenesis of Alzheimer's disease
presented in Fig 1.
Figure 1.
General pathogenesis of Alzheimer's
disease
1.2 Herbs/medicinal
plants
Traditional
medicines have been practiced for treating and curing various diseases and illnesses
since ancient times. It is due to the fact that the phytochemicals present in herbs
and medicinal plants have significant impact on treating diseases and improves
health with the least side effects. The herbs which have been used for the
treatment of Alzheimer’s disease have the ability to improve brain functions [14]. The exact mechanism of action of
medicinal plants against AD is not known, but they exert their protective
effects against cognitive impairment through antioxidant and anti-inflammatory
activities and specific action on AChE, β-amyloid fibril formation and tau aggregation
[15]. Possible mechanism of action of herbal drugs shown in Fig 2.
Figure 2. Herbal drugs possible mechanism of action
2. Materials and methods
2.1 Search
strategy and selection
The current
study was conducted for finding plants that gives
anti-Alzheimer’s activity. The published studies were searched by
using terms like ‘anti-Alzheimer’s activity of various plants’,
‘anti-cholinesterase activity’, ‘evaluation of anti-Alzheimer’s activity’,
‘neuro-protective herbs’, ‘Indian medicinal herbs for anti-Alzheimer’s
activity’, ‘cholinesterase inhibitory activity’, ‘medicinal plant with
anti-Alzheimer’s activity’, ‘effect of medicinal plants on Alzheimer’s
disease’, and ‘anti-Alzheimer’s activity of fruit’ through Google, Google Scholar,
PubMed.
3.
Results and discussion
The plants that
give anti-Alzheimer’s activity were selected and study their phytochemical
constituents that give different activity. We have enlisted the selected plants
that give anti-Alzheimer’s activity below.
3.1 Satureja cuneifolia
3.1.1
General description
Satureja
cuneifoliais a plant (Fig. 3.) of
the Lamiaceae family, which is used in Ayurveda and herbal treatment for
centuries [16].
3.1.2 Active constituents
The methanolic and
water extract of Satureja cuneifolia contains both flavonoids and
phenolic compounds like rutin, kaempferol-3-Orutinoside and fumaric acid, and
the results show that methanolic extract is rich in phenolic compounds, which
highlights the anti-oxidant and antiradical property in the plant.
3.1.3 Pharmacological properties
Antioxidant,
antidiabetic and anti-Alzheimer’s disease activity.
The extracts were
analysed by LC-MS/MS method and total phenolic contents were determined. By
some enzyme inhibition method as in vitro, it has been found that the
methanolic and water extract of Saturejacuneifolia enhances the anti-Alzheimer and ant diabetic
activity [17]. It has been found that water and methanolic plant extract
have the inhibitory property of
3.2 Moringa oleifera
3.2.1 General description
In
the family Moringaceae, Moringa oleifera (Fig.
4.) is the common species amongst the 13
cultivars. It’s commonly used in Africa and Asia as a food or food additive,
due to its Phytochemical and pharmacological property [19].
3.2.2 Active constituents
The
phytochemicals present in the plant are phenolic compounds, carotenoid,
polyphenol, flavonoid, saponin, terpene and glycoside compounds [20].
3.2.3 Pharmacological properties
Aqueous,
methanol, ethyl acetate and hexane extract of leaf, seed, and root of Moringa
oleifera shows pancreatic lipase and acetylcholinesterase inhibition properties,
as well as it shows antioxidant activity (DMPD+ radical scavenging activity,
nitrite scavenging activity and ferric reducing power). Also, it’s used to treat asthma, heart disease,
anemia, swelling and wounds [82].
The
modified method of Ingkaninan et al. was employed for assaying
acetylcholinesterase activity and absorbance was taken at 405nm. Aqueous leaf
extract (IC50 = 3.26 ± 0.26 mg/ml) and hexane root extract (IC50 = 0.08 ± 0.00
mg/ml) exhibited the highest antilipase and antiacetylcholinesterase activity
respectively. The result shows that Moringa oleifera shows antilipase,
anticholinesterase, and antioxidant properties [21].
3.3 Phyllanthus acidus
3.3.1 General description
Phyllanthus acidus (Fig. 5.), belonging to the family Euphorbiaceae, is a tree that
has been used in traditional medicine to treat pain, inflammatory, oxidative
stress related disorders and is also important to promote intellect and enhance
memory.
3.3.2 Active
constituents
Phyllanthusacidus contain chemical
constituents like phenolic compounds and flavonoids [22].
3.3.3 Pharmacological properties
The in vitro study of methanolic fruit extract of Phyllanthus
acidus shows it has a considerable amount of antioxidant activity as well
as anti-acetylcholinesterase and anti-butyrylcholinesterase activity, so it
shows its effectiveness against Alzheimer's disease and other neurodegenerative
disorders [23]. Anti-diabetic activity, burning micturition, enhancing
immunity and to treat anemia are the other uses [82].
Antioxidant potential and neuroprotective activities were evaluated by assessing total phenol content (FCR assay), total flavonoid content, total antioxidant capacity, Fe3+ reducing power capacity, DPPH (2, 2-diphenyl-1-picrylhydrazyl) and hydroxyl radical scavenging capacity, lipid peroxidation inhibition activity & metal chelating activity, acetylcholinestrase (AChE) and butyrylcholinestrase (BChE) inhibitory activities were performed using Ellman’s method [24].
3.4 Clitoria ternatea
3.4.1 General description
Clitoria ternatea (Fig.
6.) belongs to the family fabaceae, have been widely used as a brain tonic
and is believed to promote memory and
intelligence [25]. Hence the present study was conducted to determine the
Alzheimer’s activity of Clitoria ternatea.
3.4.2 Active constituents
The
alcoholic extract of Clitoria ternatea contains phytochemicals like
flavonoids, alkaloids, tannins, phytosterol,
phenol and saponins [26].
3.4.3 Pharmacological activities
The test for determining the activity of acetyl cholinesterase was done by Ellmann’s method; used for monitoring cholinesterase activity and also Ach hydrolysis by acetyl cholinesterase. From the study, it was found that aqueous extract of Clitoria ternatea has a higher activity when compared to neostigmine standard. So the aqueous extract of Clitoria ternatea can be used for Alzheimer’s disease.
3.5. Crocus sativus
3.5.1 General description
Crocus sativus (Fig.
7.) also called saffron belongs
to the family Iridaceae, having great properties on memory and
cognitive deficiency [27].
3.5.2 Active constituents
The
Phytochemicalconstituents present in the Crocus sativus are picrocrocin,
kaempferol, safranal, phenol, flavonoids and crocetin. These phytochemicals
have the capacity to elucidate anti-Alzheimer’s and memory enhancer properties [28].
3.5.3 Pharmacological activities
C.sativus possess Nerve sedative, analgesic, anti-Alzheimer’s
activity. Also saffron has the ability to cross the blood brain barrier, so it
can be considered in therapeutic approaches of other neurological disorders.
From the study, it has been shown that the Crocus sativus has the greater effect on Alzheimer’s disease and also other neurological effects. The phytochemical constituents and pharmacological property it has a greater value in the treatment of Alzheimer’s disease [29].
3.6 Annona squamosa
3.6.1 General description
The
plant Annona squamosa Linn (Fig.
8.) belongs to the family Annonaceae.
3.6.2 Active constituents
The
secondary metabolites present in the plants were anthocyanidins, flavones,
flavonols, and alkaloids [30]. Among phenolic compounds, flavonoids enhance antioxidant
properties in plant extracts. Vitamin C is considered a powerful antioxidant
which eliminates non-radical reactive species. Carotenoids, found in the pulp
and seed of A. squamosa shows antioxidant activity by suppression of
superoxide O2 which can result in cell damage. Carotenoid consumption provides
prevention of degenerative diseases.
3.6.3 Pharmacological activities
Antioxidant,
anti-inflammatory, antidiabetic and anti-acetylcholinesterase activity. Fruit is
sweet, flavorous, enriches the blood, increases muscular strength, cooling, sedative
to heart and relieve vomiting. Root cathartic, drastic purgative. Astringent
bark is used as an antidiarrhoeic cure in Cambodia. Leaves are used as
insecticide.
The total phenolic, flavonoids and vitamin C content of methanolic extracts of pulp and seed of Annona squamosal L was determined by the Folin–Ciocalteu method, aluminium chloride colorimetric method, 2,6-dichlorophenol-indofenol titration method respectively. Evaluation of antioxidants was conducted by using the ABTS, Fe3+ reduction, β-carotene protection, DPPH and 2-DR protection methods. The assay for inhibition of acetylcholinesterase activity was determined using the method described by Ellman [31].
3.7
Glycyrrhiza glabra
3.7.1 General
description
Glycyrrhiza glabra is a plant (Fig.
9.) of the Leguminosae
family which is used in Ayurveda and herbal treatment for centuries [32].
3.7.2 Active constituents
The aqueous root extract of Glycyrrhiz aglabra contains triterpene, saponins, polysaccharides,
flavonoids, mineral salts, amino acids, pectins, simple sugars and
various other substances.
3.7.3 Pharmacological
properties
It has antimalarial, expectorant, diuretic, laxative, antispasmodic, anti-inflammatory, antioxidant, anti-ulcer and sedative properties. Glycyrrhizin and its aglycone, glycyrrhetinic acid, enhance anti-inflammatory activity [33].
The aqueous extract of the root of Glycyrrhiza glabra
was administered orally in 1-month-old male Wistar albino rats for six
successive weeks by Four doses (75, 150, 225, and 300 mg/kg). The antioxidant
and anti-inflammatory activities of Glycyrrhiza
glabra exhibit memory enhancement effect. It concluded that the aqueous root
extract of Glycyrrhiza glabra reveals memory
enhancement effects in the management of impaired learning, dementia,
Alzheimer's disease, and other neurodegenerative disorders [34].
3.8 Withania somnifera
3.8.1
General description
Ashwagandha,
or Withania somnifera (Fig. 10),
is a Solanaceae family herb, often called Indian winter cherry or Indian
ginseng, and has been used for thousands of years in India to benefit health [35].
3.8.2 Active constituents
Withasomniferin
A, withasomidienone, withasomniferols A to C,withaferin A, and withanone are
among the most interesting steroidal compounds in Ashwagandha. withanamides antagonise neuronal cell death triggered
by amyloid plaques. It also contains alkaloids,
high amounts of iron and amino acids including tryptophan, also contain
lignans, tannins, polyphenols, sterols and flavanoids.
3.8.3 Pharmacological
properties
So it has antioxidant activity, free radical scavenging
activity, anti-inflammatory, ant-amyloidogenic, anti-cholinesterase and
hypolipidemic activities [36]. Also used to anxiety, depression, and fatigue [82].
Aqueous extracts of this herb have been found to
increase cognition-enhancing and memory-improving effects. Methanolic extracts
of Withania somnifera reversed amyloid
peptide-induced memory deficit in mice. SoWithania somnifera is used in the treatment of Alzheimer’s
disease [37].
Figure 3. to Figure 10.
3.9 Celastrus paniculatus
3.9.1 General description
Celastrus paniculatus
(Fig.
11.) is a woody liana commonly known as black
oil plant. it is found in Maldives,
Australia, China, Cambodia, Malaysia, Taiwan, Nepal, Thailand and in the
Pacific Islands. Celastrus paniculatus is plant
belonging to the family Celastraceae [38].
3.9.2 Active
constituents
The seed of C. paniculatus
contains sesquiterpene alkaloids, monounsaturated and polyunsaturated fats, sesquiterpene ester triterpenoids, volatile
oil, polyalcohol esters, sterols and fatty
acids.
3.9.3 Pharmacological
properties
The oil obtained from the seed of Celastrus paniculatus enhances sedative and anticonvulsant properties.
The methanolic extract of Celastrus paniculatus seed exhibited analgesic and anti-inflammatory properties in mice and rats [39]. Celastrus paniculatus seed oil enhances memory processes in rats. A methanolic extract of Celastrus paniculatus seed oil enhances the free radical scavenging effects. Invitro cholinesterase enzyme inhibition assay and scavenging of DPPH, ONOO-, reducing power and inhibition of total ROS generation assay were used for studying anti-Alzheimer’s disease and antioxidant effect of crude methanolic extract various organic soluble fractions of Celastrus paniculatus seed. The EtOAc fraction had major (pp0.001) inhibitory effects on cholinesterases [40].
3.10 Ginkgo biloba
3.10.1
General description
Ginkgo
biloba
(Fig. 12.) is a living fossil that belongs
to the family Ginkgoaceae, widely cultivated in
China and Japan. It’s herbal medicine that has
been used in China since ancient times. Ginkgo
biloba extract (GBE) has been widely used to
treat Alzheimer's disease.
3.10.2 Active
constituents
The various phytochemical constituents present in the extract
are ginkgo flavonoids, including quercetin, kaempferol, and isorhamnetin,
terpene lactones, consisting of ginkgolides A, B, and C and bilobalide, and
ginkgolic acids [41].
3.10.3 Pharmacological
properties
Flavonoids enhance the antioxidant effects. The flavonoids
and terpenoids present in the extract of Ginkgo
biloba are responsible for anti-oxidation, anti-inflammation,
and anti-apoptosis; protection against mitochondrial dysfunction,
amyloidogenesis, and Aβ aggregation which exhibit anti Alzheimer’seffect [42].
3.11
Coriandrum sativum
3.11.1 General description
Coriander (Coriandrum
sativum) (Fig. 13.) is an aromatic
plant of Umbellifera or Apiaceae. [43]. Green
coriander contains high level of water, thiamine, zinc and dietary fibres and
fewer amounts of saturated fats, and cholesterol. Seeds are rich in vitamins,
lipids and minerals, such as calcium, phosphorus, sodium, zinc, potassium and
magnesium. The fresh leaves can be used for garnishing but also used in many
foods.
3.11.2 Active constituents
The
main components of essential oil are linoleic and linolenic acids. The phenolic compounds provide antioxidant
activity for C. sativum exhibit hepatoprotectiveactivity [44]. C.
sativum's
hepatoprotective activity is attributed to its phenolic compounds, which
possess antioxidant properties.
3.11.3 Pharmacological properties
The
seeds and leaves of the plant have been reported to possess a range of
beneficial properties such as antioxidant, diuretic, cholesterol lowering,
anxiolytic, sedative-hypnotic and anticonvulsant activities.
The
present study evaluated the impact of inhaling volatile oil (at concentrations
of 1% and 3% per day) over the course of 21 days, on spatial memory performance
in a rat model of Alzheimer’s disease induced by A
3.12 Bacopa monnieri
3.12.1
General description
Bacopa
monnieri (Fig. 14.),
also known as Brahmi, is an herb from the Scorophulariacae plant family
that has been mentioned in the Indian traditional medicine (ayurveda)
literature for it’s medicinal properties in treating anxiety, intellect and
memory disorders [46].
3.12.2
Active constituents
It
contains alkaloids, glycosides, sapogenin, saponins and flavonoids.
3.12.3
Pharmacological properties
Antioxidant,
free radical scavenging, anti-Alzheimer’s activity. Also used to control mental
stress, anxiety, lower blood pressure, and anti-epilepsy activity [82].
Research
has demonstrated that EBm promotes mechanisms for scavenging free radicals and
safeguards cells in the pre-frontal cortex, hippocampus and striatum against
cytotoxicity and DNA damage associated with Alzheimer’s disease. Several animals
and in vitro studies have revealed that EBm exhibits antioxidant and free
radical scavenging properties [47].
3.13 Peruvian maca
3.13.1 General description
Peruvian maca (lepidiummeyenii) (Fig.
15.) belongs to the family Brassicaceae
is a root native to the Andean region known for its high fibre and nutrient
content including vitamin C, copper, and iron [48]. Additionally, this fruit contains bioactive compounds that
provide numerous benefits to individuals seeking a healthy diet.
3.13.2 Active constituents
Maca root is abundant in essential amino acids, fatty acids
and minerals especially iron, calcium and copper.
3.13.3 Pharmacological
properties
Anti-oxidant,
anti-cancer, anti-inflammatory and antidepressant.
Maca has been shown to provide a wide range of health
benefits, including but not limited to regulation of sexual dysfunction,
neuroprotection, memory enhancement, anti-depressant effect, antioxidant
properties, anti- cancer properties, anti-inflammatory properties and skin
protection. Maca extract contains Macamides which are considered to be the
active compounds responsible for nuro-protective effect by inhibiting FAAH [49, 50].
Figure 11 to Figure
14
3.14. Phyllanthus
emblica
3.14.1 General description
Phyllanthus emblica Linn (Fig.
16.) or Emblica officinalis
Gaertn, commonly referred to as amla or Indian gooseberry, [51] is considered the most significant medicinal plant in the
Ayurvedic traditional system of medicine. The Indian gooseberry belongs to the
family Phyllanthaceae.
3.14.2 Active constituents
Compounds such as tannoids, tannins, vitamin C, and
flavonoids.
3.14.3 Pharmacological
properties
Amla has been reported to exhibit various activities such as
radiomodulatory, chemomodulatory, chemopreventive, free radical scavenging,
antioxidant, anti-inflammatory, antimutagenic, and immune modulatory effects [52]. The fruit of this plant is known to exhibit strong
antioxidant properties.
Amnesia was induced by administering Scopolamine (1 mg kg-1,
IP), and the memory was assessed through the elevated plus-maze and passive
avoidance tests. Piracetam (200 mg kg-1, IP) was used as a reference nootropic
agent. The findings of the study revealed that the EO extract was effective in
reversing the amnesia induced by scopolamine, indicating its potential as a
memory enhancer with anti-oxidant and anti-cholinesterase activity. The study
suggests that EO extract could be useful in the treatment of cognitive
impairments induced by cholinergic dysfunction [53].
3.15. Acorus calamus
3.15.1 General description
Acorus calamus Linn(Acoraceae) (Fig.
17.), commonly known as
Vacha, is a herb used in traditional Indian medicine to treat a variety of
health conditions. Acorus calamus L. is a perennial monocot plant from [54] the Acoraceae family, and its rhizomes have been
extensively used in traditional medicine to treat various ailments, including
mental ailments, chronic diarrhoea, and fever, among others.
3.15.2 Active constituents
Volatile oil (1.5-3.5%), starch, resin, and tannin, eugenol
and asarone. Volatile oil contains asaraldehyde [83].
3.15.3 Pharmacological properties
Acorus calamus leaves, rhizomes, and essential oil have been found to
possess several biological activities, such as antispasmodic and carminative
properties [54]. The extract of Acorus calamus Linn (Acoraceae),
commonly known as calamus, and its constituent α-asarone have demonstrated
significant reduction in the production of reactive oxygen species (ROS)
induced by l-glutamate. Additionally, the extract and α-asarone were able to
suppress the phosphorylation of protein kinase RNA-like ER kinase (PERK)
induced by tunicamycin [55].
These findings suggest that A. calamus extract and
α-asarone may offer protection to hippocampal cells against oxidative and ER
stress by reducing ROS production and suppressing PERK signaling, respectively.
α-Asarone shows promise as a potential therapeutic agent for treating
neurodegenerative disorders, such as Alzheimer's disease [56].
3.16 Panax ginseng
3.16.1 General description
Panax Ginseng (Fig.
18.) belongs to the
family Araliaceae. Ginseng is a memory boosting herb which is commonly
used in China, Japan and Korea.
3.16.2 Pharmacological properties
The beneficial
pharmacological effects of ginseng include antioxidant, anti-inflammatory,
anticancer and vasorelaxative effects [57].
3.16.3 Active constituents
The key components present in ginseng extract are Ginsenosides and gintonin, they possess
anti-AD effects. Ginsenosides
(a derivative of triterpenoid) are the first isolated chemical from ginseng
which inhibits aggregation of amyloid β (Aβ) and removes Amyloid-β from
neurons. It reduces the symptoms of AD by inhibiting the activity of
Anticholinesterase [58].
According to
recent reports, ginsenosides and gintonin are responsible for anti-AD activity
via inhibition of Aβ-induced neurotoxicity and reactive oxidative stress,
stimulation of soluble amyloid precursor protein α (sAPPα) formation (but not Aβ),
anti-inflammatory effects, and enhancement of cholinergic systems, hippocampal
neurogenesis, and cognitive functions by conducting in vitro and in vivo
studies. Further study should be needed to explore the benefits of ginseng
extract [59].
3.17 Uncaria
tomentosa
3.17.1 General description
Uncaria tomentosa (Fig.
19.) Also known as
Cat’s claw belongs to the family Rubiaceae. It is preferred as raw
material for cat’s claw.
3.17.2 Active constituents
Alkaloids
are the major constituent responsible for pharmacological activities. The primary chemical constituents
found in Cortex Uncariae are tetracyclic indole alkaloids such isorhynchophylline
and rhynchophylline, along with indole alkaloids like speciophylline,
mitraphylline, pteropodine, uncarine F, and isomitraphylline.
3.17.3 Pharmacological properties
The known
pharmacological activities of U.tomentosa are anti-inflammatory,
antioxidant, immune modulator, antitumor, antidepressant and neuroprotective
activity.
It
has been reported that uncaria tomentosa can alleviate cognitive
impairments in AD [60]. A similar plant of the family known as uncariarhynchophylla
also possess the same activity since both contain similar chemical
constituents. A study was conducted to compare the anti-AD activities of
methanolic extracts of these plants and it has proved that they significantly
improved the learning and memory impairments in STZ-induced rats [61]. A study was carried out for evaluating the
anticholinesterase activity of the aqueous extract of Uncaria tomentosa.
The extract showed maximum inhibition of the enzyme. So it reveals that U. tomentosa
possessespotent anti-AChE activity. Oxindole alkaloids present in aqueous
extract are responsible for anti-AChE activity [62].
3.18 Curcuma longa
3.18.1 General description
Curcuma longa (Fig. 20.) belongs to the family Zingiberaceae.
It consists of dried
as well as fresh rhizomes and is a common spice in South
Asia
3.18.2 Active
constituents
Turmeric contains about 5 per cent of volatile oil, resin, abundant
Zingiberaceous starch grains and curcuminoids [83].The
active constituent present in Curcuma Longa is Curcumin
(difeuloylmethane) [63].
3.18.3 Pharmacological
properties
Curcumin is a powerful anti-inflammatory agent and antioxidant so it has
been used to treat inflammation of skin and muscles in the Indian and Chinese
systems of medicine. Curcumin can penetrate blood-brain barrier as it is a
small molecule. Curcumin inhibits the production and accumulation of Aβ
polypeptide. A diet containing significant amounts of curcumin may be the
reason for the lower risk of AD in older Indians, since it was revealed that
the prevalence of AD in adults aged 70 to 79 in India is 4.4 times lower than
that in the United States [64].
Studies showed that curcumin protects neurons from degeneration. Curcumin
is incorporated with PLGA-PEG-PLGA thermo-sensitive hydrogel and given as an intramuscular
injection in aluminium chloride induced rat model, the system sustained the
release of curcumin loaded micelle for 20 days and showed good biocompatibility
and biodegradability which effectively prevent development and progression of
AD. No significant toxicity has been
reported [65]
3.19 Centella asiatica
3.19.1 General description
Centella asiatica (Fig.
21.) also known as Jalbrahmi or Mandukparni belongs to the family Apiaceae
[66]. In the Ayurvedic
system of medicine, the leaves of Centella have been used as a memory enhancer.
It has a potential to prevent various memory-related disorders and also
practiced in African and Chinese system of medicine.
3.19.2 Active
constituents
Asiatic acid, asiaticosides, madasiatic acid, madecassoside, (main
constituents) brahmoside, brahminoside, isothankuniside, thankuniside and
centelloside are the important phytochemicals present Centella asiatica [68].
Ethanolic extract of C. Asiatica has been
shown to have antioxidant, AChE inhibitory and anti-diabetic activity which may
lead to the finding of a more effective agent for the treatment and management
of AD and Diabetes and related other disorders.
The enzymes acetyl cholinesterase (AChE) and butyryl cholinesterase
(BChE) were measured to determine the anti-Alzheimer activity [69].
3.19.3 Pharmacological
properties
Pharmacological activities of the plant includes antioxidant,
anti-inflammatory, neuroprotective, antidepressant, nootropic, anticonvulsant,
sedative and immune stimulant activity [67].
The results of the
present research revealed that the C. Asiatica extract might have a substantial inhibitory effect on ChEs. C. Asiatica may be
able to boost levels of the neurotransmitter acetylcholine and hence enhance
synaptic transmission in the AD brain by blocking the enzymes AChEs and BChEs.
Through the scavenging of reactive free radicals and ROS, which would otherwise
play a significant role in the production of neurofibrillary tangles and
neurotic plaques, the plant extract's anti-oxidant activity also, suggested
that it has neuro protective effects in AD. A recent study revealed that
extract of Centellaasiatica selectively decreased the levels of amyloid
beta in the hippocampus of animal models with Alzheimer disease [70].
3.20 Convolvulus pluricaulis
3.20.1 General
description
Convolvulus pluricaulisis (Fig.
22.) is a perennial herb belongs to the family convolvulaceae. It is
also known as Shankpushpi [71]. It has
been used traditionally for curing variety of disorders like nausea, bed
urination, ulcers, hypertension, hyperthyroidism, convulsions and neurodegenerative
disorders [72].
3.20.2 Active constituents
C.pluricaulis contains bioactive compounds such as cinnamic acid, pentanoic acid,
ascorbic acid, vitamin E, phthalic acid, squalene, silane, decanoic acid,
linoleic acid, b-sitosterol, tropane alkaloids, and kaempferol [73].
3.20.3 Pharmacological properties
Shankpushpi is a powerful anti-inflammatory, anticonvulsant, anxiolytic,
and antioxidant.
Convolvulus pluricaulis has been shown to have a
variety of pharmacological actions, including the improvement of learning and
memory in both young and old rats in recent studies. Chronic unpredictable mild stress
induced rat when exposed to methanolic extract of C.pluricaulis resulted
in remarkable changes in inflammatory cytokines, liver enzymes, serotonin, and
noradrenaline levels. Hence C.pluricaulis may turn
out to be an efficacious medication for depression and neuroinflammation [74.]. In a Drosophila model of
Alzheimer's disease, Convolvulus Pluricaulis alleviated human
microtubule-associated protein tau-induced neurotoxicity.
Figure 15 to Figure
22
3.21 Eclipta
alba
3.21.1 Gneral description
Eclipta alba/false daisy (Fig. 23.) is an
annual herbaceous plant belonging to the family Asteraceae. It is found
throughout India, abundant in marshy places and available in all seasons [83].
Synonyms- Bhringraj and Karisilakanni [75].
3.21.2 Active
constituents
The major chemical constituents present in E.alba are alkaloids,
flavonoids, glycosides, triterpenoides, polyacetylenes, coumestrans, sigmasterols,
etc [76].
3.21.3 Pharmacological
properties
Anti-inflammatory,
antidiabetic, antiproliferative, hypolipedemic
Clinical studies on pharmacological actions such hepatotoxicity,
proliferative, diabetic, hypolipedemic, etc. have been conducted. To
investigate the anti-inflammatory effects of an oral methanolic extract, albino
Wistar rats have been used [77]. Studies
proved that methanolic extract of E. alba
ameliorates mitochondrial dysfunction and oxidative stress by employing
DPPH and ABTS radical scavenging assays for evaluating in vitro antioxidant activity.
According to this study, Ecliptaalba reduces scopolamine-induced lipid
peroxidation, the total neuroprotective effect of the extract was found to be
more than 50%, showing the potential advantages of indigenous plants in
ameliorating oxidative stress-induced mitochondrial dysfunction and enhances mitochondrial
activity in the rat brain [78].
3.22 Salvia officinalis
3.22.1 General description
Salvia officinalis L. (Fig.
24.) belongs to the family Lamiaceae/Labiatae. The genus Salvia
includes around 900 species and is one of the largest Genus of Lamiaceae family.
S.officinalis is native to Middle East and Mediterraneanareas.
3.22.2 Active
constituents
1,8cineole, camphor, α- andβ -thujone, vridiflorol,
and α-pinene are among the major chemical constituents found in S.
officinalis.
3.22.3 Pharmacological
properties
It has been used for the treatment of various disorders like
inflammation, hyperglycemia, gout, rheumatism, diarrhea, ulcer, seizures,
dizziness and paralysis in the folk medicine of Asia and Latin America [79].
Many studies have been undertaken in recent years to examine the
pharmacological activities of S. officinalis, and it has been reported
that it possesses anti-inflammatory, antibacterial, antioxidant hypoglycemia
and hypolipidaemic, anti-nociceptive, antimutagenic, and antidementia activities
[80]. The
pharmacological actions of S. officinalis are speculated to include
effects that are anti-mutagenic, anti-cancer, anti-inflammatory, and that
improve memory and cognition. Essential oils obtained from S. officinalis
and other species of salvia is known to have anticholinesterase activity and
improve mood and cognitive performance [81].
Figure
23 and Figure 24
The herbal drugs and their activities are shown in
Table 1.
Table 1. Herbal drugs and activity
Herbs |
Family |
Chemical
composition |
Pharmacological
activity |
|
Saturejacuneifolia |
Lamiaceae |
Flavonoids, phenolic compounds |
Anti-Alzheimers, antidiabetic. |
|
Moringaoleifera |
Moringaceae |
phenolic compounds, carotenoid,
polyphenol, flavonoid, saponin, terpene and glycoside |
antilipase, anticholinesterase, and
antioxidant, Anti-Alzheimers activity. |
|
Phyllanthusacidus |
Euphorbiaceae |
Phenolic compounds, flavonoids. |
Antioxidant, acetylcholinesterase and anti-butyrylcholinesterase. |
|
Clitoriaternatea |
Fabaceae |
Flavonoids, alkaloids, tannins,
phytosterol, phenol and saponins. |
Anxiolytic, anti-inflammatory, anti-microbial,
anti-cholineseterase. |
|
Crocus sativus |
Iridaceae |
Crocetin, flavonoids, phenol,
picrocrocin, kaempferol. |
Nerve sedative, analgesic,
anti-Alzheimer’s activity. |
|
Annonasquamosa |
Annonaceae |
Phenolic compounds, flavonoids,
carotenoids. |
Antioxidant, anti-inflammatory,
anti-acetylcholinesterase. |
|
Glycyrrhizaglabra |
Leguminosae |
Triterpene, saponins, flavonoids,
pectins, amino acids. |
Antimalarial, antispasmodic,
anti-inflammatory, anti-oxidant,
anti-Alzheimer’s activity. |
|
Withaniasomnifera |
Solanaceae |
Alkaloids, Iron, Amino acids. |
Antioxidant, free radical scavenging,
anti-Alzheimer’s activity. |
|
Celastruspaniculatus |
Celastraceae |
Alkaloids, volatile oil, sterol,
fatty acids, triterpenoids. |
Antioxidant, anti-Alzheimers. |
|
Ginkgo biloba |
Ginkgoaceae |
Flavonoids, terpenoids, ginkgolic
acids. |
Anti-free radical, antioxidant,
anti-inflammation, anti-apoptosis, anti-Alzheimer’s activity. |
|
Coriander sativum |
Apiaceae |
Phenolic compounds, essential oil. |
Antioxidants, anti-anxiety,
anticonvulsant, anti-Alzheimer’s. |
|
Bacopamonnieri |
Scrophulariaceae |
Alkaloids, glycosides, flavonoids,
saponins. |
Antioxidant, free radical scavenging,
anti-Alzheimer’s activity |
|
Peruvian maca |
Brassicaceae |
Amino acids, fatty acids, minerals. |
Anti-oxidant, anti-cancer,
anti-inflammatory, antidepressant, Anti-Alzheimers. |
|
Phyllanthusemblica |
Phyllanthaceae |
Flavonoids, tannins, vitamin C,
tannoids. |
Antioxidant, anti-inflammatory, anti-
cholinesterase. |
|
Acoruscalamus |
Acoraceae |
Asarone, shyobunone. |
Antispasmodic, carminative,
anti-Alzheimer’s. |
|
Panax ginseng |
Araliaceae |
Ginsenosides, gintonin. |
Antioxidant, anti-inflammatory,
anticancer, Anti-Alzheimers. |
|
Uncariatomentosa |
Rubiaceae |
Tetracyclic indole alkaloids, indole
alkaloids, oxindole alkaloids. |
Anti-oxidant, antitumor, neuro
protective, antidepressant. |
|
Curcuma longa |
Zingiberaceae |
Curcumin |
Anti-inflammatory, antioxidant, anti-
Alzheimer’s. |
|
Centellaasiatica |
Apiaceae |
Asiaticoside, madasiatic acid,
brahmoside, thankuniside, centelloside. |
Antioxidant, anti-inflammatory,
neuroprotective, antidepressant, anticonvulsant. |
|
Convolvulus pluricaulis |
Convolvulaceae |
Ascorbic acid, vitamin E, linoleic
acid, b-sitosterol, tropane alkaloids. |
Anti-inflammatory, anticonvulsant,
anxiolytic, antioxidant, Anti-Alzheimers. |
|
Ecliptaalpa |
Asteraceae |
Alkaloids, flavonoids, glycosides,
triterpenoids, coumestrans, sigmasterols. |
Anti-inflammatory, antidiabetic,
antiproliferative, hypolipedemic, Anti-Alzheimers. |
|
Salvia officinalis |
Lamiaceae |
1,8 cineole, camphor, |
Anti-inflammatory, antioxidant, antidementia,
anti-cociceptive, hypolipedemic, Anti-Alzheimers. |
4. Conclusions
The management of Alzheimer’s disease is a big challenge for the medical science, because there are few choices and only a few drugs are approved by the US FDA. Since ancient times traditional medicines are practiced for memory loss worldwide. Based on that the plants that give anti-Alzheimer’s activity was selected and study their phytochemical constituents that give the different activity. We have enlisted the selected plants that give anti-Alzheimer’s activity. Due to the limited availability of journals, libraries, and recent research papers after 2022, the current study was unable to gather a significant amount of information about herbs for the treatment of Alzheimer’s disease.
Authors’ contributions
Collect literature, J.A.; A.K.P.; S.K.;
G.S. and J.J.N.; Materialize and typed the review, M.M.; A.A.G. and S.E.N
Acknowledgements
Declared none.
Funding
No fund Received
Availability of data and materials
All
relevant data are within the paper and its supporting information files.
Additional data will be made available on request according to the journal
policy.
Conflicts of interest
Authors have declared that no competing
interests exist.
References
1.
Akram,
M.; Nawaz, A. Effects of medicinal plants on Alzheimer’s disease and memory
deficits. Neural Regen. Res. 2017, 12, 660-670. doi:
10.4103/1673-5374.205108.2.
Hippius, H.; Neundörfer, G. The discovery
of Alzheimer’s disease. Dialogues Clin. Neurosci. 2003, 5, 101–108.3.
Livingston, G.; Huntley, J.; Sommerlad,
A. Dementia prevention, intervention and care. Lancet Comm. 2020, 396, 413-446. 4.
Shal, B.; Ding, W.; Ali, H.; Kim, Y.S.; Khan,
S. Anti-neuroinflammatory potential of natural products in attenuation of Alzheimer’s
disease. Front. Pharmacol. 2018, 9, 548.5.
Overk, C.R.; Masliah, E. Pathogenesis of
synaptic degeneration in alzheimer’s disease and lewy body disease. Biochem.
Pharmacol. 2014, 88, 508-516. doi: 10.1016/j.bcp.2014.01.015. 6.
Gregory, J.; Vengalasetti, Y.V.; Bredesen,
D.E.; Rao, R.V. Neuroprotective herbs for the management of Alzheimer’s disease.
Biomolecules. 2021, 11, 4. doi: 10.3390/biom11040543.7.
Hoyer, S. Oxidative energy metabolism in
Alzheimer brain. Studies in early-onset and late-onset cases. Mol. Chem.
Neuropathol. 1992, 16, 207-224. doi: 10.1007/BF03159971. 8.
Tiwari, S.; Atluri, V.; Kaushik, A.; Yndart,
A.; Nair, M. Alzheimer’s disease: pathogenesis, diagnostics, and therapeutics. Int.
J. Nanomedicine. 2019, 14, 5541-5554. doi: 10.2147/IJN.S200490.9.
Mehla, J.; Gupta, P.; Pahuja, M.; Diwan,
D.; Diksha, D. Indian medicinal herbs and formulations for Alzheimer’s disease,
from traditional knowledge to scientific assessment. Brain Sci. 2020,
10, 12. doi: 10.3390/brainsci10120964. 10.
Chen, Z.R.; Huang, J.B.; Yang, S.L.; Hong,
F.F. Role of cholinergic signaling in Alzheimer’s disease. Molecules. 2022, 27,
1816. doi: 10.3390/molecules27061816.11.
Tönnies, E.; Trushina, E. Oxidative stress,
synaptic dysfunction, and Alzheimer’s disease. J. Alzheimers Dis. 2017,
57, 1105-1121. doi: 10.3233/JAD-161088. 12.
Prasad, K.N.; Cole, W.C.; Prasad, K.C. Risk
factors for Alzheimer’s disease: role of multiple antioxidants, non-steroidal
anti-inflammatory and cholinergic agents alone or in combination in prevention
and treatment. J. Am. Coll. Nutr. 2002, 21, 506-522. doi:
10.1080/07315724.2002.10719249.13.
Grossberg, G.T. Cholinesterase inhibitors
for the treatment of Alzheimer’s Disease. Curr. Ther. Res. Clin. Exp.
2003, 64, 216-235. doi: 10.1016/S0011-393X(03)00059-6. 14.
Arpita Roy. Role of medicinal plants
against Alzheimer’s disease. Int. J. Complement. Altern. 2018, 11, 205-208. doi:
10.15406/ijcam.2018.11.00398.15.
Phani Kumar G.; Farhath Khanum. Neuroprotective
potential of phytochemicals. PMC. 2012,
12, 81-90. doi: 10.4103/0973-7847.99898. 16.
Perrino, E.V.; Valerio, F.; Jallali, S.;
Trani, A.; Mezzapesa, G.N. Ecological and biological properties of Satureja
cuneifolia Ten. and Thymus spinulosus ten, two wild officinal
species of conservation concern in Apulia (Italy), A preliminary survey plants.
Nat. Lib. Med. 2021, 10, 1952. doi: 10.3390/plants10091952.17.
Taskin, T.; Dogan, M.; Cam, M.E.; Sahin,
T.; Senkardes, I. In vitro anti-urease, antioxidant, anticholinesterase,
cytotoxic and in vivo anti-inflammatory potential of Satureja cuneifolia. Ten.
Not. Sci. Biol. 2020, 12, 222-232. doi: 10.15835/nsb12210687. 18.
Taslimi, P. Anti-Alzheimer, antidiabetic
and antioxidant potential of Satureja cuneifolia and analysis of its phenolic
contents by LC-MS/MS. Arab. J. Chem. 2020, 13, 4528-4537. doi:
10.1016/j.arabjc.2019.10.002.19.
AbdRani, N.Z.; Husain, K.; Kumolosasi,
E. Moringa Genus: A review of phytochemistry and pharmacology. Front.
Pharmacol. 2018, 9, 108. doi: 10.3389/fphar.2018.00108. 20.
Anzano, A.; Ammar, M.; Papaianni, M.;
Grausol, L.; Sabbah, M.; Capparelli, R.; Lanzotti, V.; et al. Moringa
oleifera Lam.: A phytochemical and pharmacological overview. Horticulturae.
2021, 7, 409.doi: 10.3390/horticulturae7100409.21.
Magaji, U.; Sacan, O.; Yanardag, R. Antilipase,
antiacetylcholinesterase and antioxidant activities of Moringa oleifera
extracts. Rom. Biotechnol. Lett. 2022, 27, 3208-3214. doi:
10.25083/rbl/27.1/3208-3214. 22.
Mao, X.; Wu, L.F.; Guo, H.L.; Chen,
W.J.; Cui, Y.P.; Qi Qi.; Li, S.; Liang, W.Y.; et al. The genus Phyllanthus:
An ethnopharmacological, phytochemical, and pharmacological review. ECAM. 2016, 2016, 7584952. doi:
10.1155/2016/7584952.23.
Uddin, M.S.; Mamun, A.A.; Hossain, M.S.;
Ashaduzzaman, M.; Noor, M.A.A.; Uddin, M.J.; et al. Neuroprotective effect
of Phyllanthus acidus L. on learning and memory impairment in
scopolamine-induced animal model of dementia and oxidative stress: Natural
Wonder for Regulating the Development and Progression of Alzheimer’s Disease. Adv.
Alzheimers Dis. 2016, 5, 53-72. doi: 10.4236/aad.2016.52005. 24.
Moniruzzaman, M.; Asaduzzaman, M.;
Hossain, M.S.; Sarker, J.; Rahman, S.M.A.; Rashid, M.; Rahman, M.M.; et al. In
vitro antioxidant and cholinesterase inhibitory activities of methanolic fruit
extract of Phyllanthus acidus. BMC Complement. Altr. Med. 2015,
15, 403. doi: 10.1186/s12906-015-0930-y.25.
Mukherjee, P.K.; Kumar, V.; Kumar, N.S.;
Heinrich, M. The ayurvedic medicine Clitoriaternatea—From traditional
use to scientific assessment. J. Ethnopharmacol. 2008, 120, 291-301. doi:
10.1016/j.jep.2008.09.009. 26.
Jiji, K.N.; Muralidharan, P. Neuropharmacological
potential of Clitoria ternatea Linn.-A review. Res. J. Pharm. Technol.
2020, 13, 5497-5502. doi: 10.5958/0974-360X.2020.00960.9.27.
Srivastava, R.; Ahmed, H.; Dixit, R.K.;
Dharamveer.; Saraf, S.A. Crocus sativus L.: A comprehensive review. Pharmacogn.
Rev. 2010, 4, 200-208. doi: 10.4103/0973-7847.70919. 28.
Maqbool, Z.; Arshad, M.S.; Ali, A.;
Aziz, A.; Khalid, W.; Afzal, M.F.; Bangar, S.P.; Addi, M.; et al. Potential role
of phytochemical extract from saffron in development of functional foods and
protection of brain-related disorders. Oxid. Med. Cell. Longev. 2022,
2022, 6480590. doi: 10.1155/2022/6480590.29.
Talebi, M.; Talebi, M.; Samarghandian,
S. Association of Crocus sativus with cognitive dysfunctions and Alzheimer’s
Disease: A systematic review. Biointerface Res. Appl. Chem. 2020, 11,
7468-7492. doi: 10.33263/BRIAC111.74687492. 30.
Kumar, M.; Changan, S.; Tomar, M.;
Prajapati, U.; Saurabh, B.; Hasan, M.; Sasi, M.; et al. Custard apple (Annona
squamosa L.) Leaves: nutritional composition, phytochemical profile and
health-promoting biological activities. Biomolecules. 2021, 11, 614. doi:
10.3390/biom11050614.31.
Leite, D.O.D.; et al. Chemical profile
and evaluation of the antioxidant and anti-acetylcholinesterase activities of Annona
squamosa L. (Annonaceae) extracts. Nat. Lib. Med. 2021, 10, 1010-2343. doi:
10.3390/foods10102343. 32.
Pastorino, G.; Cornara, L.; Soares, S.; Rodrigues,
F.; Oliveira, M.B.P.P. Liquorice (Glycyrrhiza glabra): A phytochemical
and pharmacological review. Phytother. Res. 2018, 32, 2323-2339. doi:
10.1002/ptr.6178.33.
Graebin, C.S. The pharmacological
activities of glycyrrhizinic acid (“glycyrrhizin”) and glycyrrhetinic acid. Sweeteners.
2017, 245-261. doi: 10.1007/978-3-319-27027-2_15. 34.
Chakravarthi, K.K.; Avadhani, R. Beneficial
effect of aqueous root extract of Glycyrrhiza glabra on learning and
memory using different behavioral models: An experimental study. J. Nat. Sci.
Biol. Med. 2013, 4, 420-425. doi: 10.4103/0976-9668.117025.35.
Singh, N.; Bhalla, M.; de Jager, P.; Gilca, M. An overview on Ashwagandha: A rasayana
(Rejuvenator) of Ayurveda. Afr. J. Trad. Complement. Altr. Med. 2011, 8,
208-213. doi: 10.4314/ajtcam.v8i5S.9. 36.
Rao, R.V.; Descamps, O.; John, V.;
Bredesen, D.E. Ayurvedic medicinal plants for Alzheimer’s disease: a review. Alzheimers
Res. Ther. 2012, 4, 22. doi: 10.1186/alzrt125.37.
Kuboyama, T.; Tohda, C.; Komatsu, K. Effects
of Ashwagandha (roots of Withania somnifera) on neurodegenerative
diseases. 2014, 37, 892-897. doi:
10.1248/bpb.b14-00022. 38.
Nagpal, K.; Garg, M.; Arora, D.; Dubey,
A.; Grewal, A.S. An extensive review on phytochemistry and pharmacological
activities of Indian medicinal plant Celastrus paniculatus willd. Phytother.
Res. 2022, 36, 1930-1951.doi: 10.1002/ptr.7424.39.
Choudhary, A.; Soni, P. Pharmacological activities
of Celastrus paniculatus Willd.: A Review. Int. J. Pharm. Sci. Rev. Res.
2021, 69, 139-144. doi: 10.47583/ijpsrr.2021.v69i01.021. 40.
Badrul, A.; Ekramul, H. Anti-Alzheimer
and antioxidant activity of Celastrus paniculatus seed. Iran. J. Pharm.
Sci. 2011, 7, 49-56.41.
Oken, B.S.; Storzbach, D.M.; Kaye, J.A. The
efficacy of Ginkgo biloba on cognitive function in Alzheimer's disease. Arch.
Neurol. 1998, 55, 1409-1415. doi: 10.1001/archneur.55.11.1409. 42.
Weinmann, S.; Roll, S.; Schwarzbach, C.;
Vauth, C.; Willich, S.N. Effects of Ginkgo biloba in dementia: systematic
review and meta-analysis. BMC Geriatr. 2010, 10, 14. doi:
10.1186/1471-2318-10-14.43.
Sharma, M.M.; Sharma, R.K. Coriandrum
sativum-an overview. sciencedirect. 2012, 1, 42-54. 44.
Msaada, K.; et al. Antioxidant activity
of methanolic extracts from three coriander (Coriandrumsativum L.) fruit
varieties. Arab. J. Chem. 2017, 10, S3176-S3183. doi:
10.1016/j.arabjc.2013.12.011.45.
Hosseini, M.; Boskabady, M.H.; Khazdair,
M.R. Neuroprotective effects of Coriandrumsativum and its constituent,
linalool: A review. Avicenna J. Phytom. 2021, 11, 436-450. doi:
10.22038/AJP.2021.55681.2786. 46.
Mukherjee, P.K.; Amit Kar. Bacopa
monnieri - an overview. ScienceDirect. 2022, 2, 1-41.47.
Chaudhari, K.S.; Tiwari, N.R.; Tiwari,
R.R.; Sharma, R.S. Neurocognitive effect of nootropic drug Brahmi (Bacopa
monnieri) in Alzheimer’s disease. Ann. Neurosci. 2017, 24, 111-122. doi:
10.1159/000475900. 48.
Gonzales, G.F. Ethnobiology and ethnopharmacology
of Lepidium meyenii (Maca), a plant from the Peruvian highlands. ECAM. 2012, 2012, 193496. doi:
10.1155/2012/193496.49.
Rubio, J.; et al. Aqueous extract
of black Maca (Lepidium meyenii) on memory impairment induced by
ovariectomy in mice. ECAM.
2011, 2011, 253958. doi: 10.1093/ecam/nen063. 50.
Da Silva Leitão Peres, N.; et al. Medicinal
effects of Peruvian maca (Lepidium meyenii): a review. Food Funct. 2020,
11, 83-92. doi: 10.1039/c9fo02732g.51.
Poltanov, E.; et al. Chemical and
antioxidant evaluation of Indian gooseberry (Emblica officinalis Gaertn.,
syn. Phyllanthus emblica L.) supplements. Phytother. Res. 2009,
23, 1309-1315. doi: 10.1002/ptr.2775. 52.
Baliga, M.S.; Dsouza, J.J. Amla (Emblica
officinalis Gaertn), a wonder berry in the treatment and prevention of
cancer. Eur. J. Cancer Prev. Off. J. Eur. Cancer Prev. Organ. ECP. 2011, 20, 225-239. doi:
10.1097/CEJ.0b013e32834473f4.53.
Golechha, M.; Bhatia, J.; Arya, D.S. Studies
on effects of Emblica officinalis (Amla) on oxidative stress and
cholinergic function in scopolamine induced amnesia in mice. J. Environ. Biol.
2012, 33, 95-100. 54.
Sharma, V.; Sharma, R.; Gautam, D.S.;
Kuca, K.; Nepovimova, E.; Martins, N. Role of Vacha (Acorus calamus
Linn.) in neurological and metabolic disorders: evidence from
ethnopharmacology, phytochemistry, pharmacology and clinical study. J. Clin.
Med. 2020, 9, 1176. doi: 10.3390/jcm9041176.55.
Mikami, M.; et al. Acorus calamus
extract and its component α-asarone attenuate murine hippocampal neuronal cell
death induced by l-glutamate and tunicamycin. Biosci. Biotechnol. Biochem.
2021, 85, 493-501. doi: 10.1093/bbb/zbaa071. 56.
Sharma, V.; Singh, I.; Chaudhary, P. Acorus
calamus (The Healing Plant): a review on its medicinal potential, micropropagation
and conservation. Nat. Prod. Res. 2014, 28, 1454-1466. doi:
10.1080/14786419.2014.915827.57.
Mehta, S.D.; Rathore, P.; Rai, G.; Mehta,
S.D.; Rathore, P.; Rai, G. Ginseng: Pharmacological
action and phytochemistry prospective in Ginseng-modern aspects of the
famed traditional medicine. Intech Open. 2021, 12, 399. doi:
10.5772/intechopen.99646. 58.
Kim, H.J.; et al. Panax ginseng
as an adjuvant treatment for Alzheimer’s disease. J. Ginseng Res. 2017, 42,
401-411. doi: 10.1016/j.jgr.2017.12.008.59.
Choi, S.H.; et al. Ginseng gintonin,
aging societies, and geriatric brain diseases. Integr. Med. Res. 2021, 10,
100450. doi: 10.1016/j.imr.2020.100450. 60.
Koriem, K.M.M. Cortex Uncariae: A
review on pharmacology, toxicology, precautions, and dosage. Biointerface Res.
Appl. Chem. 2022, 13, 334. doi: 10.33263/BRIAC134.334.61.
Xu, Q.Q.; et al. Comparison of the
chemical constituents and anti-Alzheimer’s disease effects of Uncaria
rhynchophylla and Uncaria tomentosa. Chin. Med. 2021, 16,
110. doi: 10.1186/s13020-021-00514-2. 62.
Chowdhury, S.; Shivani.; Kumar, S. In
vitro anti-acetylcholinesterase activity of an aqueous extract of Unicaria
tomentosa and in silico study of its active constituents. Bioinformation.
2016, 12, 112-118. doi: 10.6026/97320630012112.63.
Ansar, S.; et al. Curcuma longa:
A treasure of medicinal properties. cellmed. 2020, 10, 9.1-9.7. doi:
10.5667/CELLMED.2020.0009. 64.
Kim, J.; Lee, H.J.; Lee, K.W. Naturally
occurring phytochemicals for the prevention of Alzheimer’s disease. J.
Neurochem. 2010, 112, 1415-1430. doi: 10.1111/j.1471-4159.2009.06562.x.65.
Lin, Y.W.; Fang, C.H.; Yang, C.Y.; Liang,
Y.J.; Lin, F.H. Investigating a curcumin-loaded PLGA-PEG-PLGA thermo-sensitive
hydrogel for the prevention of Alzheimer’s Disease. Antioxidants. 2022, 11,
727. doi: 10.3390/antiox11040727. 66.
Gohil, K.J.; Patel, J.A.; Gajjar, A.K. Pharmacological
review on Centella asiatica: A potential herbal cure-all. Indian J.
Pharm. Sci. 2010, 72, 546-556. doi: 10.4103/0250-474X.78519.67.
Abbas, S.; et al. A review of
antioxidant and anti-acetylcholinesterase activities of Centella asiatica
(L.) Urb. for the treatment of Alzheimer’s disease. Food Res. 2021, 5,
1-17. doi: 10.26656/fr.2017.5(2).355. 68.
Gray, N.E.; et al. Centella asiatica–Phytochemistry
and mechanisms of neuroprotection and cognitive enhancement. Phytochem. Rev.
Proc. Phytochem. Soc. Eur. 2018, 17, 161-194. doi:
10.1007/s11101-017-9528-y.69.
Rahman, M.; Sayeed, M.S.B.; Haque, A.; Hassan,
M.; Islam, S.M.A. Phytochemical screening, antioxidant, anti-alzheimer and
anti-diabetic activities of Centella asiatica. Scholar research library.
2012, 2, 504-511. 70.
Dhanasekaran, M.; et al. Centella
asiatica extract selectively decreases amyloid beta levels in hippocampus
of Alzheimer’s disease animal model. Phytother. Res. 2009, 23, 14-19. doi: 10.1002/ptr.2405.71.
Satish, A.B.; Deepa, R.V.; Nikhil, C.T.;
Trikannad.; Ethnobotany, A.A. Phytochemistry and pharmacology of Convolvulus
pluricaulis, choisy. Res. J. Pharm. Biol. Chem. Sci. 2016, 5,
629-636. 72.
Chandel, U.; Kharoliwal, S. Phytochemical
estimation of convolvulus pluricaulis choisy. WJPPS. 2018, 6, 1074-1082.
doi: 10.20959/wjpps201710-10262.73.
Rachitha, P.; et al. Chemical
composition, antioxidant potential, macromolecule damage and neuroprotective
activity of Convolvulus pluricaulis. J. Tradit. Complement. Med. 2018,
8, 483-496. doi: 10.1016/j.jtcme.2017.11.002.74.
Gupta, G.L.; Fernandes, J. Protective
effect of Convolvulus pluricaulis against neuroinflammation associated
depressive behavior induced by chronic unpredictable mild stress in rat. Biomed.
Pharmacother. 2019, 109, 1698-1708. doi:
10.1016/j.biopha.2018.11.046. 75.
Mithun, N.M.; Shashidhara, S. Eclipta
alba (L.) A review on its phytochemical and pharmacological profile. PharmaTutor.
2011, 26, 136.76.
Jaglan, D. Pharmacological activity and
chemical constituents of EcliptaAlba. Global J. Inc. 2013, 13, 135. 77.
Chokotia, L.S.; Vashistha1, P.; Sironiya1,
R.; Matoli, H. Parmacological activities of eclipta alba.Int. J. Res.
Dev. Pharm. Life Sci. 2013, 2, 499-502.78.
Bhaskar, M.; Chintamaneni, M. Withania
somnifera and Eclipta alba ameliorate oxidative stress induced
mitochondrial dysfunction in an animal model of Alzheimer’s disease. AJPCT. 2014,
3, 140-152. 79.
Miraj, S.; Kiani, S. A review study of
therapeutic effects of Salvia officinalis L. Scholar Res. Lib. 2016, 8,
299-303.80.
Ghorbani, A.; Esmaeilizadeh, M. Pharmacological
properties of Salvia officinalis and its components. J. Trad. Comp. Med.
2017, 7, 433-440. doi: 10.1016/j.jtcme.2016.12.014. 81.
Fu, Z.; Wang, H.; Hu, X.; Sun, Z.; Han,
C. The Pharmacological properties of Salvia essential oils. J. Appl. Pharm.
Sci. 2013, 3, 122-127.82.
Ministry of ayush government of india.
Common medicinal plants and their use,2020. 83.
Kokate, C.K.; Purohit, C.; BGhokale, S. Pharmacognosy,
47th Edn.; Nirali prakashan: Pune, India, 2012.
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
Abstract
Alzheimer's disease (AD) is the
dominant source of dementia worldwide among numerous neurodegenerative
disorders, with no definitive and enduring cure to date despite prolonged
research efforts. AD is believed to be caused due to the accumulation of
extracellular Aβ plaques and intracellular neurofibrillary tangles consisting
of hyper phosphorylated τ-protein in various parts of the human brain, such as
the cortex and limbic system. The development of drugs has been
significantly benefited by Ayurveda medicinal plants. Presently, there are more
than a hundred new medicines already in clinical development. The primary
emphasis of the review is on exploring multiple medicinal plants as a means of
preventing symptoms associated with Alzheimer’s disease. A study of the
phytochemical properties in various parts of plants has revealed several
significant chemical compounds, including lignans, flavonoids, tannins,
polyphenols, triterpenes, sterols, and alkaloids. These agents exhibit a
diverse range of pharmacological functions that can aid in the prevention and
treatment of various conditions, such as inflammation, amyloid deposits,
cholinesterase inhibition, hyperlipidemia, and oxidative stress.
Abstract Keywords
Alzheimer’s disease, medicinal
plants, phytochemical constituents, neuroprotective
herbs, therapeutic effects, flavonoids.
This work is licensed under the
Creative Commons Attribution
4.0
License (CC BY-NC 4.0).
Editor-in-Chief
This work is licensed under the
Creative Commons Attribution 4.0
License.(CC BY-NC 4.0).


